Abstract
In vivo, leukocyte transendothelial migration (TEM) occurs at endothelial cell junctions (paracellular) and nonjunctional (transcellular) locations, whereas in vitro models report that TEM is mostly paracellular. The mechanisms that control the route of leukocyte TEM remain unknown. Here we tested the hypothesis that elevated intercellular adhesion molecule-1 (ICAM-1) expression regulates the location of polymorphonuclear leukocyte (PMN) TEM. We used an in vitro flow model of tumor necrosis factor-α (TNF-α)-activated human umbilical vein endothelium cells (HUVECs) or an HUVEC cell line transfected with ICAM-1GFP (green fluorescent protein) and live-cell fluorescence microscopy to quantify the location of PMN adhesion and TEM. We observed robust transcellular TEM with TNF-α-activated HUVECs and ICAM-1GFP immortalized HUVECS (iHUVECs). In contrast, primary CD3+ T lymphocytes exclusively used a paracellular route. Endothelial ICAM-1 was identified as essential for both paracellular and transcellular PMN transmigration, and interfering with ICAM-1 cytoplasmic tail function preferentially reduced transcellular TEM. We also found that ICAM-1 surface density and distribution as well as endothelial cell shape contributed to transcellular TEM. In summary, ICAM-1 promotes junctional and nonjunctional TEM across inflamed vascular endothelium via distinct cytoplasmic tail associations. (Blood. 2005;106:584-592)
Introduction
Leukocyte transendothelial migration (TEM) is a key step in their recruitment to sites of inflammation, injury, and immune reactions. Leukocyte recruitment involves a multistep cascade consisting of leukocyte rolling, firm adhesion, and, ultimately, transmigration.1 These steps are regulated by shear flow, apical chemokines, and inducible adhesion molecules expressed by endothelium.2 The actual path of leukocyte egress (paracellular versus transcellular) has been examined in several models (reviewed in Muller,3 Kvietys and Sandig,4 and Engelhardt and Wolburg5 ). Experiments carried out in several animal models of inflammation found that leukocyte TEM can occur at both paracellular and transcellular locations.6-12 In contrast, most in vitro studies have reported that leukocyte TEM occurs primarily at paracellular locations.3,13-15 As a result, in vitro models of polymorphonuclear leukocyte (PMN) transcellular TEM do not exist, and the factors that dictate whether leukocytes use a transcellular or paracellular route and the mechanisms underlying transcellular TEM are unknown.
Intercellular adhesion molecule-1 (ICAM-1) interacting with its leukocyte counterreceptors lymphocyte function-associated antigen-1 (LFA-1) and macrophage antigen-1 (Mac-1) is key for leukocyte adhesion and TEM.16-19 ICAM-1 is a transmembrane glycoprotein with 5 extracellular immunoglobulin G (IgG)-like domains and a short cytoplasmic tail that associates with multiple cytoskeletal linker proteins (reviewed in Springer1 ). Vascular endothelium expresses low levels of ICAM-1, and inflammatory stimuli can markedly increase ICAM-1 surface expression.20,21 In acute and chronic inflammatory diseases, endothelial cells become activated and express high levels of ICAM-1, in addition to vascular cell adhesion molecule-1 (VCAM-1) and E-selectin.22-25
ICAM-1 has been shown to signal in endothelium during leukocyte adhesion or when cross-linked by anti-ICAM-1 antibodies. ICAM-1 occupancy triggers elevations in intracellular free Ca2+ and myosin contractility,26 activation of p38 kinase,27 and the small guanosine triphosphatases (GTPases), in particular, members of the Rho family28,29 and the tyrosine kinase p60Src.30 Activation of these signaling pathways results in extensive cytoskeletal remodeling events that alter endothelial cell contractility and function,27 possibly facilitating leukocyte diapedesis. Recently, we have shown that neutrophil LFA-1 rapidly redistributes to form a ringlike structure that coclusters with endothelial ICAM-1 during PMN transmigration at cell junctions.31 Others have reported that ICAM-1-enriched projections engulf leukocytes during their firm adhesion and locomotion32 and TEM.33,34 We, therefore, hypothesized that leukocyte LFA-1 differentially engages ICAM-1 at junctional versus nonjunctional regions during adhesion and transmigration steps. This prompted us to test the involvement of ICAM-1 in distinct TEM routes for leukocytes.
Here, we report that ICAM-1 density and distribution as well as endothelial cell shape influence the location of PMN adhesion and transmigration under physiologic shear flow. PMNs, but not primary CD3+ T lymphocytes, undergo transcellular TEM via a mechanism involving high-occupancy levels of ICAM-1 on the endothelial surface. Furthermore, the ICAM-1 cytoplasmic tail is necessary to translate ICAM-1 clustering into cytoplasmic association(s), which enhances PMN transcellular TEM.
Materials and methods
Materials
Human recombinant tumor necrosis factor-α (TNF-α) was from Genzyme (Cambridge, MA). Stromal cell-derived factor-1α (SDF-1α, CXCL12 [CXC chemokine ligand 12]) and interleukin 8 (IL-8, CXCL8) were from Peprotech (Rocky Hill, NJ). Murine monoclonal antibody (mAb) was used as purified IgG. Both TEA1/31 (Immunotech, Westbrook, ME) and Hec-1 (gift of Dr William Muller, Weill Medical College, New York, NY) are nonblocking mAbs to human vascular endothelial (VE)-cadherin and were conjugated to Alexa 568 (Molecular Probes, Eugene, OR) for detection of human umbilical vein endothelium cell (HUVEC) junctions as described.35 Other murine mAbs used at 20 μg/mL were function-blocking anti-ICAM-1 mAb (282; a gift from Dr Mary Gerritsen, Genentech, South San Francisco, CA) and nonblocking anti-ICAM-1 mAb (CL23.4; a gift from Dr Ferrone, Roswell Park Cancer Institute, Buffalo, NY),36 function-blocking anti-LFA-1 mAb (TS1/22; American Type Culture Collection [ATCC], Manassas, VA), function-blocking anti-Mac-1 mAb (44, ATCC; and LPM19c, gift of Dr Dan Simon, Brigham and Women's Hospital, Boston, MA), and nonblocking anti-JAM-A (junctional adhesion molecule-A) mAb 12A931,37 served as the binding control. Nonbinding control mAb K16/1638 was used in fluorescence-activated cell sorting (FACS).
Cells and cell lines
HUVECs were isolated and cultured as described.35 Human papilloma virus (HPV) immortalized HUVECs (iHUVECs) were provided by Dr Ashley Moses.39,40 HUVECs (passage 2) and iHUVECs (passage 7-10) were seeded on 25-mm diameter glass coverslips (Carolina Biological Supply, Burlington, NC) or glass-bottom 35-mm culture plates (MatTek, Ashland, MA) coated with 5 μg/mL fibronectin (Sigma, St Louis, MO). Human PMNs (> 95% pure) and CD3+ T lymphocytes (> 90% pure and 50%-60% CD45RO+) were isolated from sodium citrated whole blood drawn from healthy volunteers as detailed.35 Informed consent for blood donations was provided according to the Declaration of Helsinki and according to the Brigham and Women's Hospital Institutional Review Board (IRB)-approved protocols for protection of human subjects.
ICAM-1 constructs
ICAM-1GFP (green fluorescent protein)/Hygro was generated by cloning ICAM-1GFP HindIII/NotI (a gift from Dr Sanchez-Madrid, Madrid, Spain34 ) into pcDNA3.1/Hygro(+) (Invitrogen, Carlsbad, CA). Tailless ICAM-1GFP/Hygro was generated by amplification of extracellular and transmembrane domains of ICAM-1 via polymerase chain reaction (PCR) from ICAM-1GFP (sense, gagctcaagcttctcagcctcgc; antisense, ggaattcgcggttatagaggtacgtgctgaggc) and cloning both ICAM-1 tailless (HindIII/EcoRI) and GFP (EcoRI/NotI) into pcDNA3.1/Hygro(+). GFP/Hygro was generated by cloning EGFP-N2 (Clontech, Palo Alto, CA) NheI/NotI into the pcDNA3.1/Hygro(+). The DNA sequences of the above constructs were confirmed by High Throughput Sequencing Service Core, Brigham and Women's Hospital, Boston, MA.
Generation of stable transfection iHUVEC cell line
Stably transfected cell lines were established by transfection of pcDNA3.1/Hygro containing ICAM-1GFP, tailless ICAM-1GFP, or GFP into iHUVECs exactly as described previously.40
Immunoprecipitation and SDS-PAGE
iHUVECs or ICAM-1GFP iHUVECs cell membranes were biotinylated using an electrochemiluminescence (ECL) Protein Biotinylation kit (Amersham Biosciences, Piscataway, NJ). Endothelial cells were lysed, and cell extracts were incubated with protein G-Sepharose (Sigma) and 5 μg anti-ICAM-1 mAb (HU5/3)41 overnight at 4°C as previously reported.13 Following resuspension in lysis buffer, isolated proteins were separated by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) and transferred to polyvinylidene difluoride membranes. Biotinylated ICAM-1 or ICAM-1GFP was detected by Streptavidin-horseradish peroxidase (Amersham Biosciences).
Penetratin-ICAM-1 tail peptides
Penetratin-ICAM-1 peptides were N-terminally labeled with the fluorescent tag nitrobenzoxadiazole (NBD) and consisted of 16 amino acids (aa) of penetratin followed by the 13 C-terminal amino acids of human ICAM-1 (QRKIKKYRLQQAQ), similar to a previous report.26 The control NBD-tagged peptide was an irrelevant sequence from the soluble part of rat rodopsin (CKPMSNFRFGENH).26 Peptides were synthesized and purified by high-performance liquid chromatography (85%) (GeneMed, San Francisco, CA). We optimized conditions so that both ICAM-1 tail and control penetratin peptides (100 μg/mL, 2 hours at 37°C) were internalized with similar efficiency and distribution in greater than 90% of the iHUVECs as assessed by confocal microscopy (data not shown).
Leukocyte adhesion and transmigration assay
The live-cell fluorescence microscopy flow model has been described.31,35 Confluent HUVECs were stimulated with TNF-α (25 ng/mL) for 4 or 24 hours and were inserted into the flow chamber. Where indicated, HUVECs were preincubated with Alexa 568-tagged anti-VE-cadherin mAb (10 minutes, 0.7 μg/mL).35 PMNs or T lymphocytes (1 × 106/mL) suspended in flow buffer31,35 (Dulbecco phosphate-buffered saline/0.1% human serum albumin [DPBS/0.1% HSA]) were drawn across HUVECs at 1.0 dyne/cm2 for 3 minutes, followed by buffer alone for 10 minutes. For T-lymphocyte studies, 50 ng/mL SDF-1α was preincubated with HUVEC monolayers for 5 minutes prior to assay to promote T-lymphocyte TEM.2,35
Laser scanning confocal microscopy
Confluent iHUVECs or ICAM-1GFP iHUVECs were treated for 4 hours with TNF-α to induce ICAM-1. The surface distribution pattern of ICAM was detected by live-cell staining using anti-ICAM-1 Alexa 488-conjugated mAb CL23.4 as described,31 and ICAM-1GFP was detected by monitoring GFP signal (green channel). Endothelial monolayers were observed using a BioRad Radiance 2100 laser scanning confocal head attached to an inverted microscope (Nikon TE2000, Melville, NY) through a 60 × oil immersion objective. The confocal iris was set to provide a 0.5-μm slice thickness. ICAM-1 fluorescence was scanned in the Z-axis in 0.5-μm steps. Images were analyzed using Lasersharp2000 v4.3 (BioRad, Hercules, CA) and ImageJ (National Institutes of Health, Bethesda, MD), and figures were generated in Photoshop v.7.0 (Adobe, San Jose, CA).
Image acquisition and analysis
Live-cell imaging of leukocyte TEM was performed using a digital imaging system coupled to a Nikon TE2000 inverted microscope as detailed previously.31 An ORCA-ER camera (Hamamatsu, Bridgewater, NJ) acquired images through MetaMorph software (v.5.0; Universal Imaging, Downingtown, PA). For TEM experiments, sequential images of VE-cadherin (red) and differential interference contrast (DIC) were taken every 15 seconds for 10 minutes in a representative field from 3 minutes after the start of leukocyte perfusion. The total number of accumulated leukocytes was determined by counting total adherent and transmigrated cells in 5 fields. The percentage of TEM = total transmigrated leukocytes/[total adhered + transmigrated leukocytes] × 100. The image in Figure 4A was prepared by overlaying locations of adhesion and subsequent PMN TEM onto the image of VE-cadherin staining. Leukocytes that transmigrated more than 5 μm from junctions, as identified by VE-cadherin staining, and did not disrupt the VE-cadherin staining were considered “transcellular TEM.”
Electron microscopy
Confluent 4-hour TNF-α-stimulated ICAM-1GFP iHUVECs on 25-mm diameter glass coverslips were coincubated with 6 × 106 PMNs for 5minutes at 37°C. Nonadhered PMNs were removed by 3 washes with PBS. HUVECs were fixed and processed for electron microscopy (CM-10; Philips, Einhoven, The Netherlands) as described.9
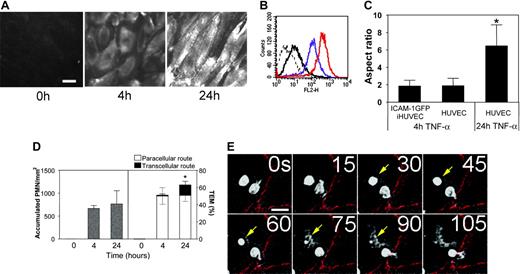
HUVECs expressing high levels of ICAM-1 support PMN transcellular TEM. (A) Fluorescent micrographs of live HUVEC monolayers treated with media containing TNF-α for 4 hours or 24 hours or media alone (0 hours), as indicated, and then stained with the nonfunction-blocking ICAM-1 mAb CL23.4 conjugated with Alexa 488. The images were captured using identical settings and were processed identically as detailed in “Materials and methods” to create the images shown. Bar, 20 μm, × 40 objective, 0.75 NA. (B) The surface expression of ICAM-1 on 4-hour (purple line) or 24-hour (red line) TNF-α-activated HUVECs was assessed by indirect immunofluorescence staining and flow cytometry as previously described.37 Dashed line indicates nonbinding mAb; solid black line, media alone (0 hours after TNF-α treatment). (C) Confluent primary HUVECs or ICAM-1GFP iHUVECs were treated with TNF-α for 4 hours or 24 hours, as indicated. Endothelial cell monolayers were stained with Alexa 568-conjugated anti-VE-cadherin mAb (Alexa568/hec-1) to identify cell-cell lateral junctions.13 The aspect ratio (length to width) was determined by measuring the maximal length and width of the cell outlined by VE-cadherin staining. *P ≤ .05. (D) PMNs (5 × 105/mL) in flow buffer (DPBS plus 0.1% HSA) were drawn across control, 4- or 24-hour TNF-α-activated HUVEC monolayers that had been stained with fluorescently labeled VE-cadherin mAb as described in “Materials and methods.” PMN accumulation and location of TEM were determined as follows: sequential DIC and corresponding VE-cadherin red fluorescence images were overlaid in register to create a time-lapse movie using MetaMorph software. The VE-cadherin fluorescence identified the endothelial cell-cell junctions. Neutrophils that arrested or transmigrated at locations greater than 5 μm from cell-cell junctions, as identified by VE-cadherin staining, were considered nonjunctional events. n = 6 from 3 independent experiments. *P < .05. The black and gray error bars correspond to paracellular and transcellular route, respectively. All data are mean ± SD. (E) Sequential images recorded by live-cell imaging under high power (× 40 objective), depict a PMN (arrow) that arrested and then transmigrated at a nonjunctional site on 24-hour TNF-α-activated HUVECs. No change in VE-cadherin staining was observed during TEM of this PMN. Bar, 10 μm.
HUVECs expressing high levels of ICAM-1 support PMN transcellular TEM. (A) Fluorescent micrographs of live HUVEC monolayers treated with media containing TNF-α for 4 hours or 24 hours or media alone (0 hours), as indicated, and then stained with the nonfunction-blocking ICAM-1 mAb CL23.4 conjugated with Alexa 488. The images were captured using identical settings and were processed identically as detailed in “Materials and methods” to create the images shown. Bar, 20 μm, × 40 objective, 0.75 NA. (B) The surface expression of ICAM-1 on 4-hour (purple line) or 24-hour (red line) TNF-α-activated HUVECs was assessed by indirect immunofluorescence staining and flow cytometry as previously described.37 Dashed line indicates nonbinding mAb; solid black line, media alone (0 hours after TNF-α treatment). (C) Confluent primary HUVECs or ICAM-1GFP iHUVECs were treated with TNF-α for 4 hours or 24 hours, as indicated. Endothelial cell monolayers were stained with Alexa 568-conjugated anti-VE-cadherin mAb (Alexa568/hec-1) to identify cell-cell lateral junctions.13 The aspect ratio (length to width) was determined by measuring the maximal length and width of the cell outlined by VE-cadherin staining. *P ≤ .05. (D) PMNs (5 × 105/mL) in flow buffer (DPBS plus 0.1% HSA) were drawn across control, 4- or 24-hour TNF-α-activated HUVEC monolayers that had been stained with fluorescently labeled VE-cadherin mAb as described in “Materials and methods.” PMN accumulation and location of TEM were determined as follows: sequential DIC and corresponding VE-cadherin red fluorescence images were overlaid in register to create a time-lapse movie using MetaMorph software. The VE-cadherin fluorescence identified the endothelial cell-cell junctions. Neutrophils that arrested or transmigrated at locations greater than 5 μm from cell-cell junctions, as identified by VE-cadherin staining, were considered nonjunctional events. n = 6 from 3 independent experiments. *P < .05. The black and gray error bars correspond to paracellular and transcellular route, respectively. All data are mean ± SD. (E) Sequential images recorded by live-cell imaging under high power (× 40 objective), depict a PMN (arrow) that arrested and then transmigrated at a nonjunctional site on 24-hour TNF-α-activated HUVECs. No change in VE-cadherin staining was observed during TEM of this PMN. Bar, 10 μm.
Statistics
Data are mean plus or minus SD for n separate experiments. P of .05 or less was considered statistically significant using paired t test or one-way ANOVA for multiple groups.
Results
Primary HUVECs activated by TNF-α for 24 hours support transcellular neutrophil TEM
To define stimulatory conditions in which endothelial cells express elevated levels of ICAM-1, we treated HUVECs, a well-established endothelial model of large vessel endothelium, with the prototypic inflammatory cytokine TNF-α for 4 or 24 hours. ICAM-1 expression was increased at 4 hours and peaked after 24 hours of TNF-α stimulation as assessed by live-cell epifluorescence microscopy (Figure 1A) or flow cytometry (Figure 1B). At 24 hours, ICAM-1 staining showed some preferential density at junctions, and cells exhibited a dramatic shape change from polygonal to an elongated shape. This apparent shape change was quantified by determining the ratio of maximal length to width of endothelial cells (termed the aspect ratio42 ) from the junctional outlines of immunolabeled VE-cadherin (Figure 1C) using MetaMorph software.
We next determined the extent of paracellular and transcellular location of PMN transmigration of 4- or 24-hour-activated HUVEC monolayers using an in vitro flow model. To identify cell junctions, HUVEC monolayers were labeled with a nonblocking VE-cadherin mAb conjugated with Alexa 568 fluorescent tag.35 Our previous studies35 indicated that this staining procedure identifies leukocyte transmigration at junctions (transient gaps in VE-cadherin form and reseal13 ) but does not alter HUVEC permeability or leukocyte TEM. TNF-α treatment of HUVECs for 4 hours promoted robust PMN accumulation and TEM, and 24-hour treatment further increased TEM without altering accumulation (Figure 1D). Strikingly, we noted a significant number of PMNs undergoing transcellular TEM through 24-hour TNF-α-activated HUVECs, whereas nearly all PMNs transmigrated via paracellular route of 4-hour-activated HUVECs. Figure 1E depicts video frames from a live-cell experiment of a PMN that transmigrates through the endothelial cell by a transcellular route as evidenced by no disruption of VE-cadherin staining (Video S1, available on the Blood website; see the Supplemental Videos link at the top of the online article). These data suggest that the ICAM-1 expression level influences the route of TEM.
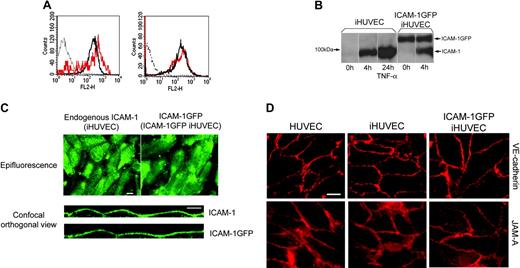
Characterization of ICAM-1GFP iHUVECs. (A) Surface expression of ICAM-1 on 4-hour TNF-α-activated ICAM-1GFP iHUVECs (red lines) was assessed by indirect immunofluorescence staining and flow cytometry and compared with that of 4-hour (left) and 24-hour TNF-α-activated iHUVECs (right; black solid line). Dotted line indicates nonbinding mAb. (B) iHUVECs or ICAM-1GFP iHUVECs were treated with TNF-α for 4 hours or 24 hours or media alone (0 hours), as indicated. Expression of endogenous ICAM-1 and ICAM-1GFP was determined by immunoprecipitation of biotinylated ICAM-1 on cell surface, as described in “Materials and methods.” (C) Surface distribution pattern of ICAM-1 on 4-hour TNF-α-activated iHUVECs and ICAM-1GFP iHUVECs was detected by live-cell epifluorescence microscopy (× 40 objective; top) and confocal microscopy (× 60 objective; bottom) as described in “Materials and methods.” Orthogonal views of endothelium were reproduced from serial z-sections horizontally scanned using confocal microscopy (0.5-μm increments) and manipulated with Lasersharp 2000 V4.3. To visualize ICAM-1, live iHUVECs were stimulated with TNF-α for 4 hours and incubated with Alexa 488-labeled anti-ICAM-1 mAb CL23.4, washed, and observed by confocal microscopy. ICAM-1GFP on 4-hour TNF-α-activated ICAM-1GFP iHUVECs was detected by GFP signal (not Alexa 488 anti-ICAM-1 mAb). (D) Live confluent HUVECs, iHUVECs, and ICAM-1GFP iHUVECs were stained with Alexa 568-labeled anti-JAM-A mAb 1H2A9 or anti-VE-cadherin TEA1/31. Images were obtained by the live-cell epifluorescence imaging as detailed in “Materials and methods.”
Characterization of ICAM-1GFP iHUVECs. (A) Surface expression of ICAM-1 on 4-hour TNF-α-activated ICAM-1GFP iHUVECs (red lines) was assessed by indirect immunofluorescence staining and flow cytometry and compared with that of 4-hour (left) and 24-hour TNF-α-activated iHUVECs (right; black solid line). Dotted line indicates nonbinding mAb. (B) iHUVECs or ICAM-1GFP iHUVECs were treated with TNF-α for 4 hours or 24 hours or media alone (0 hours), as indicated. Expression of endogenous ICAM-1 and ICAM-1GFP was determined by immunoprecipitation of biotinylated ICAM-1 on cell surface, as described in “Materials and methods.” (C) Surface distribution pattern of ICAM-1 on 4-hour TNF-α-activated iHUVECs and ICAM-1GFP iHUVECs was detected by live-cell epifluorescence microscopy (× 40 objective; top) and confocal microscopy (× 60 objective; bottom) as described in “Materials and methods.” Orthogonal views of endothelium were reproduced from serial z-sections horizontally scanned using confocal microscopy (0.5-μm increments) and manipulated with Lasersharp 2000 V4.3. To visualize ICAM-1, live iHUVECs were stimulated with TNF-α for 4 hours and incubated with Alexa 488-labeled anti-ICAM-1 mAb CL23.4, washed, and observed by confocal microscopy. ICAM-1GFP on 4-hour TNF-α-activated ICAM-1GFP iHUVECs was detected by GFP signal (not Alexa 488 anti-ICAM-1 mAb). (D) Live confluent HUVECs, iHUVECs, and ICAM-1GFP iHUVECs were stained with Alexa 568-labeled anti-JAM-A mAb 1H2A9 or anti-VE-cadherin TEA1/31. Images were obtained by the live-cell epifluorescence imaging as detailed in “Materials and methods.”
Generation of endothelial monolayers with enhanced ICAM-1 surface expression
To further investigate the role of ICAM-1 density in regulating location of TEM, we used the immortalized iHUVECs, which we had characterized earlier,40 to generate stably transfected endothelial cells overexpressing ICAM-1GFP or GFP alone. Flow cytometric analysis showed that stable transfection of iHUVECs with ICAM-1GFP significantly increased ICAM-1 expression levels both on resting (data not shown) and 4-hour TNF-α monolayers (1.8-fold increase over GFP iHUVECs treated with TNF-α for 4 hours; Figure 2A, left). The overall level of ICAM-1 expressed on 4-hour TNF-α-activated ICAM-1GFP iHUVECs was similar to that seen in 24-hour TNF-α-activated iHUVECs (Figure 2A, right). Biochemical analysis also confirmed expression of ICAM-1 and ICAM-1GFP (Figure 2B). Live-cell epifluorescence microscopy showed that the surface distribution pattern of ICAM-1GFP was similar to endogenous ICAM-1 on 4-hour TNF-α-stimulated iHUVECs (Figure 2C, top) and that ICAM-1GFP iHUVECs treated 4 hours with TNF-α maintained their polygonal shape (Figure 1C).
Further comparison of the surface distribution of ICAM-1GFP to endogenous ICAM-1 was done using confocal microscopy of live cells. Orthogonal images of these iHUVEC monolayers were reconstructed from a stack of confocal sections and revealed that the surface distribution of ICAM-1GFP was uniform and was essentially the same as endogenous ICAM-1 (Figure 2 C, bottom). Furthermore, the introduction of the ICAM-1GFP into iHUVECs did not alter the expression or distribution of junctional molecules such as VE-cadherin and JAM-A (Figure 2D).
Enhanced expression of ICAM-1 on endothelial surface promotes PMN transcellular TEM
The enhanced expression of ICAM-1GFP resulted in a variable increase in PMN accumulation on 4-hour TNF-α-stimulated iHUVECs (Figure 3A, left), but did not increase the total fraction of adherent PMNs that transmigrated. Strikingly, we observed that HUVECs stably expressing ICAM-1GFP supported robust transcellular TEM (Figure 3A, right). In contrast, 4-hour TNF-α-stimulated iHUVECs or GFP iHUVECs supported few transcellular TEM events (Figure 3A, right), consistent with data collected on 4-hour TNF-α-stimulated primary HUVECs (Figure 1D). The increase in transcellular migration of PMNs could not be explained by increased numbers of adherent PMNs, because in comparison to the concentration of PMNs used in Figure 3A (1 × 106/mL) doubling the concentration of PMNs or reducing that number by 50% did not change the distribution of transcellular or paracellular TEM (Figure 3B). This finding suggests that transcellular TEM is not dictated by PMN-PMN interactions and is not due to saturation of putative junctional TEM sites.
We next evaluated whether shear flow was required for transcellular TEM. Under static conditions or at 1 dyne/cm2, extensive transcellular TEM was still observed, and higher shear stress (3 dynes/cm2) significantly enhanced TEM by both routes but did not change the ratio of paracellular to transcellular TEM (Figure 3C). We noted that TNF-α stimulation of the iHUVEC monolayers was required for PMN transmigration because resting ICAM-1GFP iHUVECs failed to support PMN adhesion or TEM (data not shown). These data suggested a key but not exclusive role for ICAM-1 in promoting transcellular TEM.
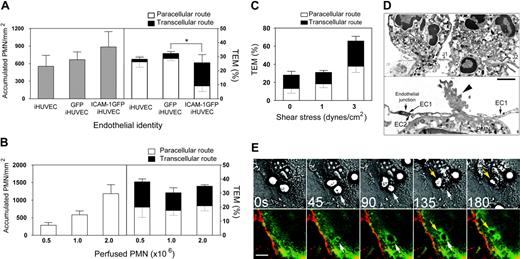
Enhanced ICAM-1 expression in HUVECs directs transcellular TEM. (A) PMN accumulation and TEM on 4-hour TNF-α-activated iHUVECs, GFP iHUVECs, or ICAM-1GFP iHUVECs were assessed as described in “Materials and methods” and Figure 1D legend. (B) Increasing numbers of PMNs (as indicated) were perfused across HUVEC monolayers under lower shear stress (1 dyne/cm2). Accumulation of PMNs (left) and the route of TEM (right) were determined. (C) PMNs (106/mL) were perfused across HUVEC monolayers at the estimated shear stress indicated. The route of TEM (right) was determined as in “Materials and methods.” n = 3 experiments. (D) Electron microscopy of PMNs undergoing transcellular TEM of 4-hour TNF-α-activated ICAM-1GFP iHUVECs under static conditions. In the top panel, 2 PMNs have extended granule-containing and, in one case, nuclear-lobe-containing cell processes, completely through the endothelial cell thickness from their apical to basilar surfaces. These endothelial cell holes are bound by endothelial cell plasma membranes and are located fewer then 5 μm (the cutoff used to group migrating cells in the “junctional route” category) from adjacent closed junctions (J1 and J2) and apposed lateral borders (arrows) of individual endothelial cells in the HUVEC monolayer. In lower panel, another PMN is nearly completely beneath the HUVEC layer after passing through an endothelial cell hole fewer than 5 μm from the adjacent closed junction (arrow) between 2 endothelial cells (EC1 and EC2). An organelle-free, actin-rich, cellular process (arrowhead) still spans the endothelial cell hole. Magnification top, × 18 000; lower, × 20 500. Bar, 1 μm. (E) Live-cell imaging of ICAM-1 GFP iHUVEC monolayers was performed as detailed35 to monitor redistribution of ICAM-1 during PMN transcellular TEM under shear flow (1 dyne/cm2). Arrows identify 2 PMNs that undergo transcellular TEM (see Video S2). Bars, 20 μm. *P < .05.
Enhanced ICAM-1 expression in HUVECs directs transcellular TEM. (A) PMN accumulation and TEM on 4-hour TNF-α-activated iHUVECs, GFP iHUVECs, or ICAM-1GFP iHUVECs were assessed as described in “Materials and methods” and Figure 1D legend. (B) Increasing numbers of PMNs (as indicated) were perfused across HUVEC monolayers under lower shear stress (1 dyne/cm2). Accumulation of PMNs (left) and the route of TEM (right) were determined. (C) PMNs (106/mL) were perfused across HUVEC monolayers at the estimated shear stress indicated. The route of TEM (right) was determined as in “Materials and methods.” n = 3 experiments. (D) Electron microscopy of PMNs undergoing transcellular TEM of 4-hour TNF-α-activated ICAM-1GFP iHUVECs under static conditions. In the top panel, 2 PMNs have extended granule-containing and, in one case, nuclear-lobe-containing cell processes, completely through the endothelial cell thickness from their apical to basilar surfaces. These endothelial cell holes are bound by endothelial cell plasma membranes and are located fewer then 5 μm (the cutoff used to group migrating cells in the “junctional route” category) from adjacent closed junctions (J1 and J2) and apposed lateral borders (arrows) of individual endothelial cells in the HUVEC monolayer. In lower panel, another PMN is nearly completely beneath the HUVEC layer after passing through an endothelial cell hole fewer than 5 μm from the adjacent closed junction (arrow) between 2 endothelial cells (EC1 and EC2). An organelle-free, actin-rich, cellular process (arrowhead) still spans the endothelial cell hole. Magnification top, × 18 000; lower, × 20 500. Bar, 1 μm. (E) Live-cell imaging of ICAM-1 GFP iHUVEC monolayers was performed as detailed35 to monitor redistribution of ICAM-1 during PMN transcellular TEM under shear flow (1 dyne/cm2). Arrows identify 2 PMNs that undergo transcellular TEM (see Video S2). Bars, 20 μm. *P < .05.
Parallel studies at the ultrastructural level using transmission electron microscopy were carried out to observe transcellular transmigration in detail. All PMNs interacting with the ICAM-1GFP iHUVEC layer were photographed in 10 samples. The endothelium was clearly oriented to show entry and exit borders for PMN transmigration and closely apposed lateral borders with visible focal junctional elements. Endothelial cells and migrating PMNs showed no ultrastructural criteria of injury. Attached and migrating PMNs developed cellular processes with multiple or single contacts with the underlying endothelium. Transmigrating PMNs passed through endothelial monolayers via a transcellular or paracellular route. Many of these events were remote from EC lateral borders with their contained junctional elements. Interestingly, some cells undergoing transcellular TEM, as defined by the 5-μm cutoff used in the light microscopic studies, were also seen to pass through and not between endothelial cells with the additional resolution afforded by transmission electron microscopy (Figure 3D). This route classification was aided by careful identification of the location(s) of individual junctional components in the lateral membrane borders of endothelial cells by transmission electron microscopy.
Because we have shown that ICAM-1 clusters around PMNs actively transmigrate at junctions,31 we imaged ICAM-1GFP iHUVECs during PMN transcellular TEM to demonstrate that its behavior is similar to endogenous ICAM-1. Interestingly, as PMNs transmigrated via a transcellular route, ICAM-1GFP was recruited to and clustered around actively transmigrating PMNs (Figure 3E; Figure 3 movie). We subsequently used the 4-hour TNF-α-activated ICAM-1GFP iHUVECs to further explore PMN transcellular TEM, because this cell type supported the most robust transcellular TEM, did not undergo dramatic shape change (Figure 1C) or decreased barrier function,37 or loss of endothelial cells as compared with 24 hours of TNF-α treatment.
High physiologic levels of ICAM-1 enhance PMN nonjunctional adhesion and promote transcellular TEM
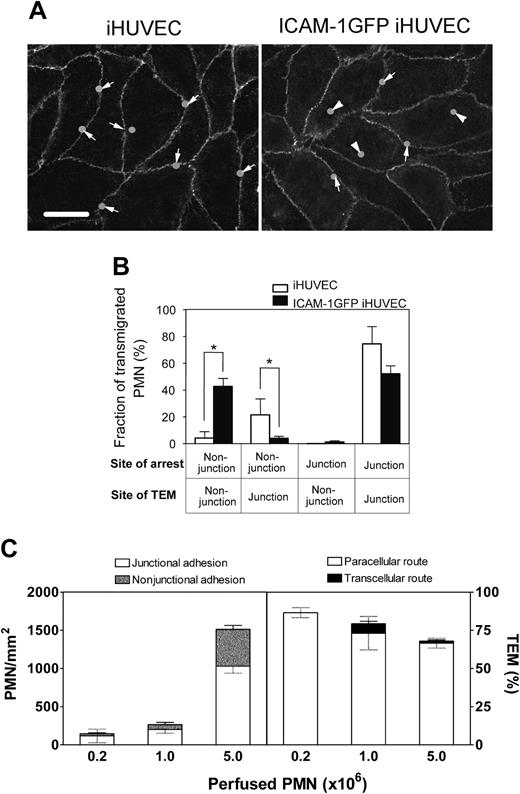
We have previous reported that approximately 70% of PMNs adhere at or adjacent to junctions of 4-hour TNF-α-activated HUVECs and undergo paracellular TEM.31 This high correlation of adhesion location to TEM route prompted us to investigate whether the location of PMN adhesion on ICAM-1GFP iHUVEC monolayers differed from that of iHUVECs. For each PMN that transmigrated, we identified the location of adhesion (solid circles) and transmigration, and this information was transferred in register to the VE-cadherin fluorescence image (Figure 4A; arrow indicates, paracellular; arrowhead, transcellular). Although both endothelial monolayers have a polygonal shape with a similar aspect ratio (Figure 1C), which provides similar surface area for PMNs to arrest at nonjunctional sites, PMNs adhered and subsequently transmigrated more frequently at nonjunctional locations of ICAM-1GFP iHUVECs (Figure 4B; n = 30 cells for each group, n = 3 experiments). Most PMNs (∼ 70%) adhered at junctions of iHUVECs. Some PMNs arrested at junctional sites and then crawled to the cell junctions to transmigrate iHUVEC monolayers. These data suggest that elevated but physiologically relevant levels of ICAM-1 and a polygonal shape of endothelium promote nonjunctional arrest and transcellular TEM.
The location of PMN adhesion and subsequent transmigration on ICAM-1GFP iHUVEC monolayers. (A) The initial adhesion sites of PMNs and the sites of TEM were determined from review of individual images using ImageJ, and these positions were overlaid onto the VE-cadherin staining pattern graph obtained prior to introduction of PMNs into the chamber. Bar, 20 μm. (B) The transmigrated PMNs were grouped according to their sites of initial arrest and TEM at junction or nonjunction (> 5 μm from any junction). The fraction of PMNs in each group was calculated. *P < .05. The black and gray error bars correspond to paracellular and transcellular routes, respectively. (C) Increasing numbers of PMNs (as indicated) were drawn across 4-hour TNF-α iHUVEC monolayers at 0.5 dyne/cm2. Initial location of arrested PMNs (left) and the route of TEM (right) were determined as detailed in “Materials and methods.”
The location of PMN adhesion and subsequent transmigration on ICAM-1GFP iHUVEC monolayers. (A) The initial adhesion sites of PMNs and the sites of TEM were determined from review of individual images using ImageJ, and these positions were overlaid onto the VE-cadherin staining pattern graph obtained prior to introduction of PMNs into the chamber. Bar, 20 μm. (B) The transmigrated PMNs were grouped according to their sites of initial arrest and TEM at junction or nonjunction (> 5 μm from any junction). The fraction of PMNs in each group was calculated. *P < .05. The black and gray error bars correspond to paracellular and transcellular routes, respectively. (C) Increasing numbers of PMNs (as indicated) were drawn across 4-hour TNF-α iHUVEC monolayers at 0.5 dyne/cm2. Initial location of arrested PMNs (left) and the route of TEM (right) were determined as detailed in “Materials and methods.”
We next determined whether PMN adhesion at nonjunctional locations of 4-hour TNF-α-activated sham-transfected iHUVECs would favor transcellular TEM. Perfusing with a 5-fold higher number of PMNs led to significant PMN adhesion at nonjunctional locations but no transcellular TEM (Figure 4C). Thus, stable β2 integrin-dependent PMN adhesion at nonjunctional sites, when coupled to high ICAM-1 occupancy and clustering (Figure 3E), appears to favor transcellular TEM.
Transcellular migration of PMNs requires ICAM-1 cytoplasmic tail
ICAM-1 is not only essential for leukocyte TEM but is the main receptor for PMN arrest under shear flow. Thus, the dissection of its role in postaccumulation transmigration events as opposed to adhesion strengthening is difficult. Indeed, blocking the extracellular domain of ICAM-1 with a function-blocking mAb resulted in more than 50% decrease in adhesion of PMNs to the ICAM-1GFP iHUVECs at 2 or 3 dynes/cm2. To differentiate the contribution of ICAM-1 to TEM steps that occur after PMNs have arrested, we used low shear flow conditions (1 dyne/cm2). Treatment of ICAM-1GFP iHUVECs with blocking anti-ICAM-1 mAb did not affect PMN accumulation but abrogated both routes of TEM (data not shown). This finding suggests that direct occupancy of ICAM-1 by adherent PMNs is required for both transcellular and paracellular TEM.
Because the association of ICAM-1 with the cytoskeleton via linker proteins and p38 kinase has been reported to regulate both adhesion and retention of PMNs at junctions,27,43 we investigated the role of the ICAM-1 cytoplasmic domain in regulating accumulation and route of TEM. First, a cell-permeant ICAM-1 cytoplasmic domain-derived peptide was used to antagonize the cytoplasmic domain of ICAM-1 without interfering with ligand recognition by the extracellular domain.26 The NBD fluorescent-tagged peptide consisted of the membrane-proximal 13-residue region of the cytoplasmic ICAM-1 tail fused to penetratin. This same ICAM-1 peptide has been shown previously to enter cultured rat endothelium and block T-lymphocyte TEM but not adhesion.26 The ICAM-1 tail peptide did not affect PMN accumulation on ICAM-1GFP iHUVECs (Figure 5A, left) or GFP iHUVECs (data not shown), or location of adhesion (data not shown), but did significantly reduce TEM across both ICAM-1GFP iHUVECs (Figure 5A, right) and iHUVECs (data not shown). Strikingly, transcellular neutrophil TEM was much more sensitive to inhibition by the ICAM-1 tail peptide than to paracellular TEM (Figure 5A, right).
We next generated iHUVECs stably expressing an ICAM-1GFP mutant lacking the entire cytoplasmic tail (herein tailless ICAM-1GFP). The expression level of tailless ICAM-1GFP and wild-type ICAM-1GFP were similar, as assessed by flow cytometry (data not shown). PMN accumulation on the various 4-hour TNF-α iHUVEC monolayers was similar, and increased nonjunctional adhesion was seen with wild-type or tailless ICAM-1GFP (Figure 5B, left). A reduction in TEM, however, was seen only in the tailless ICAM-1GFP iHUVECs (Figure 5B, right). This was the result of a marked inhibition of transcellular TEM. Taken together, both approaches suggest that transcellular TEM depends on ICAM-1 tail associations with cytoskeletal components to a greater extent than paracellular TEM.
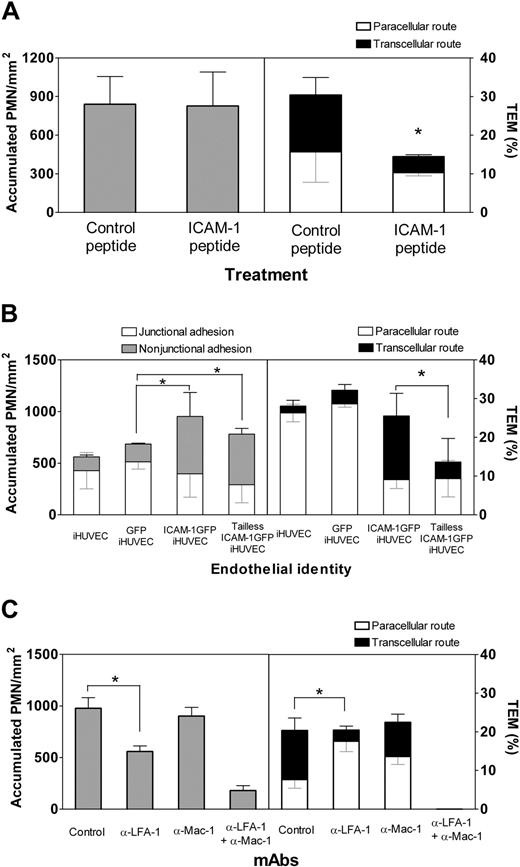
The role of LFA-1 and Mac-1 in transcellular TEM was examined using function-blocking mAb. Although the shear stress was kept at 1 dyne/cm2 as in previous experiments to minimize loss of PMN adhesion, blocking LFA-1 reduced PMN accumulation, while Mac-1 blocking had no significant effect (Figure 5C, left). Blocking of either LFA-1 or Mac-1 reduced transcellular TEM; however, LFA-1 blockade was more effective (Figure 5C, right). Taken together, these data suggest that the LFA-1/ICAM-1 pair plays a greater role than the Mac-1/ICAM-1 pair in transcellular TEM and that paracellular TEM can be triggered by either LFA-1/ICAM-1 or Mac-1/ICAM-1. As expected, blocking both LFA-1 and Mac-1 not only abrogated TEM, but also reduced accumulation.
Enhanced ICAM-1 does not trigger transcellular TEM of T lymphocytes
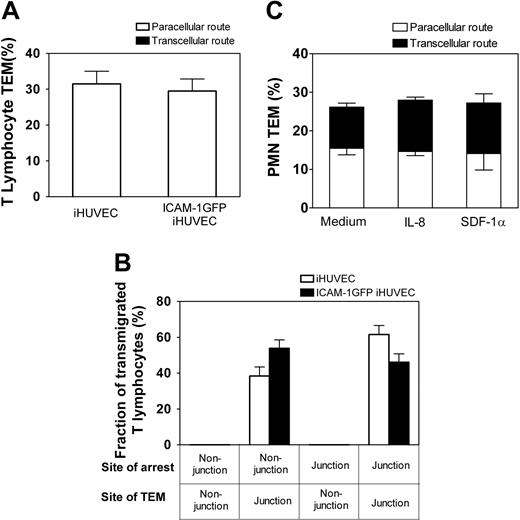
T lymphocytes express high levels of LFA-1 that can be efficiently activated by immobilized apical chemokines and mediate robust TEM.2 We, therefore, asked whether the ICAM-1GFP iHUVECs could support transcellular TEM of primary T lymphocytes. Live-cell imaging of T lymphocytes revealed that TEM occurred only at endothelial junctions of both iHUVECs and ICAM-1GFP iHUVECs treated with SDF-1α (Figure 6A). Strikingly, T lymphocytes migrated at high velocity (range, 9-16 μm/min) and considerable distances from their original site of arrest (mean distance migrated, 35 ± 10 μm) before they transmigrated. Notably, an equivalent proportion of T lymphocytes arrested at junctional and nonjunctional sites, but all T lymphocytes crawled to junctions to transmigrate (Figure 6B). In light of the migratory behavior of T lymphocytes in the presence of SDF-1α, we examined whether CXCL8 (IL-8) or SDF-1α applied to ICAM-1GFP iHUVECs altered TEM behavior of PMNs. Interestingly, these chemokines had no effect on PMN locomotion and did not alter PMN route of TEM (Figure 6C).
Transcellular TEM requires intact ICAM-1. (A) ICAM-1GFP iHUVECs were incubated with penatratin-ICAM-1 peptide or control penetratin peptide (100 μg/mL, 2 hours). PMN accumulation and the route of TEM were assessed at 1 dyne/cm2 as described in Figure 1D legend. (B) Initial location of PMN adhesion and TEM at 1 dyne/cm2 were assessed for iHUVECs, GFP iHUVECs, ICAM-1GFP iHUVECs, and tailless ICAM-1GFP iHUVECs. (C) PMN accumulation and TEM were assessed for the 4-hour TNF-α-stimulated endothelial monolayers after preincubation of PMNs with function-blocking mAb to LFA-1 (TS 1/22) or Mac-1 (mAb 44), or both. A second mAb to Mac-1 (LPM19c, data not shown) gave similar results. *P < .05.
Transcellular TEM requires intact ICAM-1. (A) ICAM-1GFP iHUVECs were incubated with penatratin-ICAM-1 peptide or control penetratin peptide (100 μg/mL, 2 hours). PMN accumulation and the route of TEM were assessed at 1 dyne/cm2 as described in Figure 1D legend. (B) Initial location of PMN adhesion and TEM at 1 dyne/cm2 were assessed for iHUVECs, GFP iHUVECs, ICAM-1GFP iHUVECs, and tailless ICAM-1GFP iHUVECs. (C) PMN accumulation and TEM were assessed for the 4-hour TNF-α-stimulated endothelial monolayers after preincubation of PMNs with function-blocking mAb to LFA-1 (TS 1/22) or Mac-1 (mAb 44), or both. A second mAb to Mac-1 (LPM19c, data not shown) gave similar results. *P < .05.
Discussion
Previous studies have reported that blood leukocytes can undergo transcellular TEM in a variety of in vivo animal models of inflammation, which suggests that this route of leukocyte transmigration is of physiologic importance.9-12 The molecular components and factors controlling transcellular TEM, however, have not been identified. To address these issues, we used an in vitro live-cell fluorescence imaging approach to visualize the endothelial cell junction marker VE-cadherin to accurately determine the site of PMN arrest and TEM (paracellular or transcellular) under physiologically relevant shear flow. We confirm recent reports33,44 that leukocytes undergo transcellular TEM and extend these findings to identify ICAM-1 as a key mediator in PMN transcellular TEM. Elevated levels of apical ICAM-1, identical to cytokine-activated HUVECs, promote an increase in PMN arrest at nonjunctional locations where high-occupancy levels of ICAM-1 by LFA-1, and to a lesser degree by Mac-1, trigger PMN transcellular TEM. In contrast, elevated levels of ICAM-1 did not trigger primary T-lymphocyte TEM by the transcellular route. These data indicate a clear difference in T-lymphocyte and PMN TEM behavior and differential signaling downstream of ICAM-1 ligation in these leukocytes.
Elevated ICAM-1 expression does not trigger T-lymphocyte transcellular TEM. The site of T-lymphocyte arrest and subsequent TEM, or PMN location of TEM across 4-hour TNF-α-activated iHUVECs or ICAM-1 GFP iHUVECs was determined by live-cell imaging as in the legend of Figure 4. (A) The percentage of T lymphocytes that transmigrated by paracellular or transcellular routes was determined. (B) The fraction of transmigrated T lymphocytes with different sites of arrest and TEM of ICAM-1GFP iHUVECs, grouped as in Figure 4A, were determined. (C) Four-hour TNF-α-activated ICAM-1GFP iHUVECs were incubated for 15 minutes with 100 ng/mL recombinant human IL-8, 10 μg/mL SDF-1α, or buffer alone and then assayed for PMN TEM as detailed in the legend of Figure 4.
Elevated ICAM-1 expression does not trigger T-lymphocyte transcellular TEM. The site of T-lymphocyte arrest and subsequent TEM, or PMN location of TEM across 4-hour TNF-α-activated iHUVECs or ICAM-1 GFP iHUVECs was determined by live-cell imaging as in the legend of Figure 4. (A) The percentage of T lymphocytes that transmigrated by paracellular or transcellular routes was determined. (B) The fraction of transmigrated T lymphocytes with different sites of arrest and TEM of ICAM-1GFP iHUVECs, grouped as in Figure 4A, were determined. (C) Four-hour TNF-α-activated ICAM-1GFP iHUVECs were incubated for 15 minutes with 100 ng/mL recombinant human IL-8, 10 μg/mL SDF-1α, or buffer alone and then assayed for PMN TEM as detailed in the legend of Figure 4.
In our study, ICAM-1GFP iHUVECs with 4-hour TNF-α activation expressed ICAM-1 at levels comparable to that observed in 24-hour TNF-α-stimulated primary HUVECs (Figures 1-2), indicating the level of ICAM-1 expression is physiologically relevant. The staining patterns of ICAM-1GFP and endogenous ICAM-1 are comparable and relatively uniform across the apical surfaces of iHUVECs activated for 4 to 6 hours. ICAM-1GFP clustered around transmigrating PMNs (Figure 3E), which is similar to endogenous ICAM-1.31,33 These data suggest no artifactual alterations in surface distribution and behavior of ICAM-1GFP. Additionally, ICAM-1GFP transfection did not affect expression or distribution of junction molecules VE-cadherin or JAM-A (Figure 2D).
There are several findings that provide important insight into the mechanisms that promote ICAM-1-dependent transcellular TEM. The expression level and distribution of ICAM-1 on endothelium strongly influence the route of PMN transmigration. We compared the PMN transmigration route though endothelium expressing different levels of ICAM-1 (4-hour versus 24-hour TNF-α-activated HUVECs, Figure 1D; mock versus ICAM-1GFP-transfected iHUVECs activated by TNF-α for 4 hours, Figures 3-4) and found that high ICAM-1 expression correlated with robust PMN transmigration at nonjunctional locations. We postulate that high levels and uniform distribution of ICAM-1 provide high occupancy of LFA-1, which promotes PMN nonjunctional adhesion and subsequent transcellular TEM.
Endothelial cell shape is the second factor that contributes to PMN transcellular TEM in this system. TNF-α treatment for 24 hours induced a pronounced increase in ICAM-1 levels and increased PMN transcellular TEM (Figure 1). However, we noted that the level of transcellular TEM in this model was lower than that in 4-hour TNF-α-activated ICAM-1GFP iHUVECs, even though both endothelial monolayers expressed essentially the same level of ICAM-1. We suggest that the difference is due to the change in cell shape (from polygonal to elongated shape with an increased aspect ratio, Figure 1C), which limits the surface area available for nonjunctional adhesion and thus transcellular TEM. Interestingly, polygonal-shaped endothelium with high-density adhesion molecules (eg, VCAM-1 and ICAM-1) have been identified in regions of the aorta of rabbits and mouse that developed atherosclerosis.23,25 Although blood monocyte TEM was not examined here, it is tempting to speculate that monocyte transcellular TEM occurs in these regions and may further contribute to endothelial cell dysfunction and vessel disease.
We were surprised to find that the tailless ICAM-1 was as effective as the ICAM-1 peptide in blocking TEM. One explanation is because surface-expressed ICAM-1 is a dimer,45 the dimers could be composed of mixtures of wild and tailless such that 3 to 4 molecules would contain at least one truncated tail version (tailless ICAM-1GFP and wild-type ICAM-1GFP were expressed at similar levels as determined by FACS). If this distribution is true, then the ratio of tailless to wild-type ICAM-1 would be higher and have less signaling capabilities, hence the (unexpected) higher effect on TEM. In addition, the preferential reduction of transcellular TEM by peptide or the tailless ICAM-1 could be due to a total dependency on ICAM-1 for TEM, whereas the paracellular route could use junctional adhesion molecules (eg, PECAM-1 [platelet endothelial cell adhesion molecule-1], CD99, JAMs).46,47
The striking result that primary isolated blood T lymphocytes transmigrate exclusively via the paracellular route as compared with PMNs strongly indicate additional factors, beyond adhesion molecule density and cell shape, regulate the transmigration route. T lymphocytes migrate extensively on the apical surface of primary HUVECs, iHUVECs, and ICAM-1GFP iHUVECs at speeds up to 16 μm/min before transmigrating exclusively by paracellular TEM. We speculate that these differences could be due to requirements for production and/or location of chemokines specific for T lymphocytes, other than SDF-1α, or differences in intracellular signaling pathways, including small GTPases Rap1 (replication-associated protein 1), Rap-L (regulator of cell adhesion and polarization enriched in lymphoid tissues), vav-1 (Vav proto-oncogene) (reviewed in Hogg et al48 ). Of note, the requirement for exogenous chemokine appears to be T lymphocyte specific because pretreatment of ICAM-1GFP iHUVECs with apical IL-8 or SDF-1α did not alter the robust PMN transcellular TEM.
Recent reports of transcellular TEM have speculated on factors that regulate this route of transmigration. Cinamon et al44 reported that low β2 integrin occupancy and appropriate chemotactic signals under shear flow promoted a low percentage of PMNs to undergo transcellular TEM via a mechanisms involving PMNs extending invaginations into the endothelial surface for transcellular TEM. A second report found that TEM occurs predominately at paracellular locations but that about 10% of PMNs, monocytes, and cultured lymphoblasts undergo transcellular TEM.33 The investigators suggest that the formation of an ICAM-1 transmigration cup structure was essential for either pathway of TEM, but the cellular mechanisms that promote transcellular TEM were not examined in detail. Hence, further studies are needed to understand the cellular mechanisms that allow for PMN egress through the endothelial cell.
In summary, our initial hypothesis was that elevated levels of ICAM-1 provide high occupancy of LFA-1, which, in turn, initiates rapid arrest and triggers transmigration at nonjunctional sites. The in vitro model described herein supports this hypothesis and provides a new assay to characterize transcellular TEM. Future studies are necessary to address how β2-integrins in subsets of leukocytes initiate apical locomotion versus rapid arrest and transcellular rather than junctional routes, and to identify specific inflammatory signal(s) that prime ICAM-1 occupancy and clustering to promote transcellular TEM. It is also of interest to determine whether endothelial cell molecules that mediate an adhesive function during junctional TEM, such as PECAM-1, CD99, and the JAM family members47,49 (JAM-A, -B, -C) also contribute to transcellular TEM. Addressing these questions may provide new targets for therapeutic approaches to interfere with pathologic TEM through junctional and nonjunctional sites.
Prepublished online as Blood First Edition Paper, April 5, 2005; DOI 10.1182/blood-2004-12-4942.
Supported by the National Institutes of Health (grants HL53993, HL36028, and HL56985 [F.W.L.] and AI33372 [A.M.D.]).
The online version of the article contains a data supplement.
An Inside Blood analysis of this article appears in the front of this issue.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 U.S.C. section 1734.
We thank the Center for Newborn at the Brigham and Women's Hospital for providing umbilical cords; Kay Case, Vannessa Davis, and Deanna Lamont for well-characterized cultures of HUVECs; and our colleagues Drs Michael Kim, Tanya Mayadas, Michael Gimbrone, and Daniel Simon at the Brigham and Women's Hospital for thoughtful and lively discussions.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal