Comment on Harman et al, page 886
The site of T-cell lineage commitment remains controversial. In this issue of Blood, Harman and colleagues elegantly redefine the site where Notch signaling determines lineage choice.
During development of the immune system, lymphoid progenitor cells must decide whether to become T or B cells. The debate continues over the timing and location of this lineage choice. While several studies have identified early T-cell progenitors from extrathymic sites, it is not clear whether precursors recruited into the thymus are multipotential hemopoietic stem cells, common lymphoid progenitors, or cells already restricted to the T-cell lineage.1,2 This confusion arises largely because studies have been carried out in adult mice where the recirculating B-cell–restricted progenitors will be present in the thymic vasculature.
Notch signaling is thought to be paramount for T-/B-cell lineage choice.3 This was first highlighted by studies showing that B cells develop in the thymus of mice colonized by Notch-1–deficient bone marrow precursors.4 It suggested that lymphoid progenitors can develop into either T or B cells depending on the Notch signaling in the thymus. However, it did not exclude the possibility that both T-restricted and B-restricted progenitors could already be present in the thymus and that the absence of Notch signaling merely allowed B-cell–restricted progenitors to develop.FIG1
T/B lineage choice in migrant thymic progenitors occurs prior to thymus entry and contact with Notch ligands on thymic epithelium. See the complete figure in the article beginning on page 886.
T/B lineage choice in migrant thymic progenitors occurs prior to thymus entry and contact with Notch ligands on thymic epithelium. See the complete figure in the article beginning on page 886.
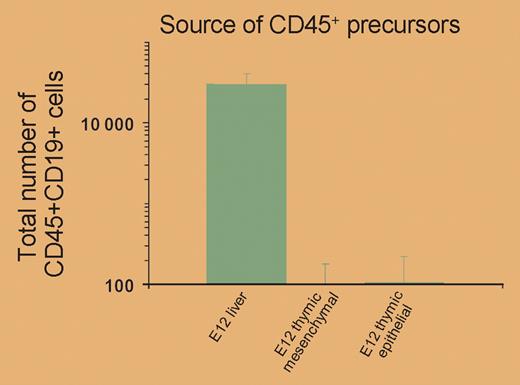
In this issue of Blood, Harman and colleagues elegantly redefine the site where Notch signaling determines lineage choice. By developing a technique that permits microdissection of fetal thymic rudiments, they were able to separate perithymic mesenchyme from the intraepithelial regions of the thymus. This allowed comparison of precursors both before and after contact with the Notch ligands expressed on thymic epithelial cells without the involvement of more mature cells from the circulation. The authors show that progenitors isolated from the epithelial region can give rise to only T cells. This is in marked contrast to progenitors from fetal liver, which could generate both T and B cells. In fact, only progenitor cells isolated from fetal liver were able to give rise to B cells (see the figure).
This shows that the decision to become a T cell occurs in fetal liver, prior to entry into the thymus. It does not exclude a role for Notch signaling in this decision; indeed, Harman et al show that Notch signaling occurs in fetal liver progenitors and suggest that further Notch signaling in the thymus reveals this choice.
In a further development, Dallas et al5 have recently shown that the density of the Notch ligand, Delta-like 1, determines the generation of B- and T-cell precursors from hematopoietic cells with low densities promoting both B- and T-cell precursor development and high densities promoting only T-cell precursor development. This correlates with the expression of Delta-like 1 shown by Harman et al. Delta-like 1 is the predominant Notch ligand expressed by fetal liver stromal cells. Thus, the density of Delta-like 1 expressed in extrathymic sites may be a key determinant in the T-/B-cell lineage choice. ▪

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal