Abstract
Mutations in fibrillin-1 (FBN1) result in Marfan syndrome, demonstrating a critical requirement for microfibrils in vessel structure and function. However, the identity and function of many microfibril-associated molecules essential for vascular development and function have yet to be characterized. In our morpholino-based screen for members of the secretome required for vascular development, we identified a key player in microfibril formation in zebrafish embryogenesis. Microfibril-associated glycoprotein-1 (MAGP1) is a conserved protein found in mammalian and zebrafish microfibrils. Expression of magp1 mRNA is detected in microfibril-producing cells. Analysis of a functional Magp1-mRFP fusion protein reveals localization along the midline and in the vasculature during embryogenesis. Underexpression and overexpression analyses demonstrate that specific Magp1 protein levels are critical for vascular development. Integrin function is compromised in magp1 morphant embryos, suggesting that reduced integrin–matrix interaction is the main mechanism for the vascular defects in magp1 morphants. We further show that Magp1 and fibrillin-1 interact in vivo. This study implicates MAGP1 as a key player in microfibril formation and integrity during development. The essential role for MAGP1 in vascular morphogenesis and function also supports a wide range of clinical applications, including therapeutic targets in vascular disease and cardiovascular tissue engineering.
Introduction
Extensive studies have shown that proper vascular development requires complex interactions among hematopoietic cells, endothelial cells, and supporting cells such as smooth muscle cells.1-4 Advances have been made in elucidating the role of extracellular matrix (ECM) components, such as the integrins, the laminins, and the proteoglycans, in modulating the behaviors of endothelial cells and vascular smooth muscle cells, among them migration, contractility, proliferation, and apoptosis.5-8 However, increasing evidence suggests that the structural elements of ECM components are also critical players in orchestrating proper vascular morphogenesis and function. For example, fibrillin-1 is a component of the microfibrils, which represent a major fibrillar network in the connective tissues within the cardiovascular system.9 Dissecting aortic aneurysm is a major vascular abnormality of Marfan syndrome, a clinical entity associated with mutations in the fibrillin-1 gene (FBN1).10 The idea that fibrillin-1 is more than a structural protein has been substantiated by studies demonstrating fibrillin-1 participation in TGF-β signaling in lung development and mitral valve disease.11,12 Fibrillin-1 has also been implicated in vascular matrix remodeling by modulating the release of matrix metalloproteases in patients with bicuspid aortic valve malformations.13 In spite of the increasing evidence that has begun to shed light on the diverse roles played by some major structural elements of the ECM in vascular morphogenesis and function, many molecular players that render the dynamic role of the ECM in vascular biology remain to be identified.
Microfibrils represent 1 critical component in the ECM of the cardiovascular system and are present in the elastic fibers of the vessel wall. Elastic fibers, which are composed of an elastin core surrounded by microfibrils, lend elasticity and resilience to the tissues. It is proposed that microfibrils regulate elastic fiber formation by guiding the deposition of the elastic precursor molecule during embryogenesis and postnatal life.14 Biochemical analyses have revealed 2 large extracellular glycoproteins, fibrillin-1 and fibrillin-2, as the major components that constitute the backbone of the microfibrils.14,15
Several molecules have been shown to physically associate with microfibrils.16-18 Immunolocalization studies have shown that microfibril-associated glycoprotein-1 (MAGP1, also known as MFAP2), a small–molecular weight glycoprotein, is localized to the “beads” of microfibrils.19 MAGP1, along with its structural relative, MAGP2, constitutes a small, unique family of microfibrillar proteins.20,21 The mammalian genes for MAGP1 have been isolated in human,22,23 mouse,24 and bovine.25 Previous in vitro studies have shown the ability of MAGP1 to bind tropoelastin (the elastin precursor protein), and antibodies directed against a domain near the amino-terminus of MAGP1 are able to block the incorporation of tropoelastin into elastic fibers by cultured chondrocytes.26,27 MAGP1 has also been shown to directly bind fibrillin-1 in vitro.28 However, the role of MAGP1 in microfibril assembly and its in vivo function were previously unknown.
As part of a morpholino-based vascular screen for members of the secretome required for vascular development, we have identified Magp1 as a key player in microfibril and elastic fiber formation during zebrafish embryogenesis. The conservation of vascular development in vertebrates29 and the capacity to effectively target this process using morpholino knockdowns30 are the basis for our morpholino-based screen for novel genes essential for vascular development. Morpholinos were designed based on 5′ leader sequences from a collection of zebrafish expression sequence tags (ESTs) with high homology to a human, a Drosophila, or a Fugu gene. Bioinformatics methods to enrich for partial sequences of cell-surface or secreted proteins based on full-length reference protein data sets have been determined, as previously described.31,32 As part of this ongoing effort, Syndecan-2, a cell-surface heparan sulfate proteoglycan, has previously been shown to play an essential role in angiogenesis during vascular development in zebrafish.33 We report here the characterization of another gene, magp1, identified from the morpholino-based vascular screen.
In this study, we show that a critical balance of Magp1 protein levels is required for proper vascular morphogenesis because underexpression and overexpression of magp1 perturb vascular development. Histologic analysis reveals altered architecture of the vessel wall and disrupted elastic fiber formation in magp1 morphants. Zebrafish magp1 mRNA marks microfibril-producing cells, as revealed by in situ hybridization. Using a functional Magp1-mRFP fusion protein, the enrichment of MAGP-1 protein is detected in elastic fiber–containing structures. MAGP-1 likely participates in integrin-mediated cell–matrix interaction during vascular development, as evidenced by the synergistic induction of vascular defects by the magp1 MO and the integrin inhibitor, Echistatin (Sigma, St Louis, MO). We further show that magp1 morphant embryos and fbn1 morphant embryos display overlapping vascular phenotypes, supporting the essential role for microfibrils in vascular development. MAGP1 represents a key molecule involved in microfibril formation during vertebrate vascular development. Its essential role in vascular morphogenesis and function promises a variety of clinical applications such as therapeutic targets in vascular disease and cardiovascular tissue engineering.
Materials and methods
Morpholino sequences and injections
The morpholino phosphorodiamidate oligonucleotides (morpholinos), from Gene Tools (Philomath, OR), have the following sequences: magp1 MO1, 5′-GGTGTCTCTGACTCAGTCCTCTGTT-3′; magp1 MO2, 5′-GCAGAAGCAGGTAACAGAGTCTCAT-3′; fibrillin-1 (fbn1) splice site MO1, 5′-GGAAACCACCTCACACTCATTG-3′; fbn1 splice site MO2, 5′-CTGGACTCACCTGTGCAGTTGCGTT-3′; VE-cadherin MO1, 5′-CTGGCACACTGTTTCATCATCAGAG-3′; and VE-cadherin MO2, 5′-ATATCCTCCTCAGTTTCAGGTCAG-3′.
All magp1 morpholinos, along with other morpholinos for the vascular screen, were designed against the 5′ leader sequences of a zebrafish gene target set (E. Klee, M. Pickart, L. Ellis, and S.C.E., unpublished observations, June 2003) enriched for predicted secreted or transmembrane proteins by bioinformatics algorithms.31,32 The fbn1 splice site MO was designed based on the sequence including a splice-donor site, which was identified by aligning the cDNA sequence with the zebrafish genomic sequence available at the Wellcome Trust Sanger Institute ensemble database (http://www.ensembl.org/Danio_rerio). All morpholinos were diluted to working concentrations using 0.3 × Danieau buffer. Danieau buffer at 1 × concentration consists of 58 mM NaCl, 0.7 mM KCl, 0.4 mM MgSO4, 0.6 mM Ca(NO3)2, and 5.0 mM HEPES pH 7.6. Morpholino solution was injected into zebrafish embryos at 1 to 2 cell stages, as described.30
For DNA-based rescue of morpholino-generated phenotypes, a solution of zebrafish magp1 and DsRed reporter DNA expression cassettes was injected into the blastomere–yolk interface of the embryos at cell stage 1 to ensure uniform mosaicism. The DsRed reporter construct was included as a lineage tracer to facilitate identification of successfully injected embryos. For coinjection experiments, magp1 MO and the mixture of magp1 DNA and DsRed reporter DNA were injected independently into embryos at the first cell stage. The expression of DsRed was visualized using a rhodamine filter set, as previously described.34
Raising and staging embryos
Embryos were raised at 29°C. Zebrafish embryos were staged using standard morphologic criteria35 before fixation and analysis to compensate for any observed developmental delay.
Cloning of zebrafish magp1
Magp1 was identified as a member of the zebrafish secretome using the procedure of Klee et al31 (and E. Klee, M. Pickart, L. Ellis, and S.C.E., unpublished data, June 2003). A full-length magp1 coding sequence was contained within the EST sequence available at the TIGR zebrafish EST database (TC192646; TIGR Gene Index 14.0). The coding cDNA was amplified from a 24-hour cDNA library using primers 5′-GTCAGAGACACCATGAGAC-3′ and 5′-TTTAGTAGGATCCACAACTC-3′ and was cloned into the TOPO vector (Invitrogen, Carlsbad, CA). A “touchdown” polymerase chain reaction (PCR) protocol was performed with the starting annealing temperature set at 69°C for 38 cycles, with 0.5°C subtracted from each of the first 29 cycles. The annealing temperature was set at 55°C for the last 9 cycles. The DNA fragment containing magp1 cDNA was isolated using EcoRI digest and was subcloned into the FRM expression construct.36 The magp1 cDNA sequence was submitted to GenBank (accession no. AY704364).
Magp1-mRFP fusion expression construct
A mixture of 3 primers (2 outer primers covering the coding regions of magp1 and monomeric RFP (mRFP) and one bridging primer covering the overlap region) was used in an asymmetric PCR protocol to generate the Magp1-mRFP fusion molecule. The monomeric RFP DNA construct was kindly provided by Roger Tsien.37 High-fidelity polymerase (Roche, Nutley, NJ) was used to amplify the fusion PCR product under the following conditions: 92°C for 2 minutes, 30 cycles at 94°C for 1 minute, 55 °C for 1 minute, and 72 °C for 1 minute, 72 °C for 7 minutes in a volume of 20 μL with 5 ng DNA template, 1 pmol each of the outer primers, 0.4 pmol of the bridging primer, 0.2 mM dNTP, and 1 U high-fidelity enzyme. The fusion PCR product was gel purified and cloned into the TOPO vector (Invitrogen) for sequence verification. The DNA fragment containing the fusion protein was subsequently isolated by EcoRI restriction digest and subcloned into the EcoRI site of the FRM expression construct. Expression of the mRFP was visualized using the rhodamine filter set as described.34
RT-PCR analysis
Reverse transcription (RT) by the MMLV reverse transcriptase (Gibco, Grand Island, NY) was performed on total RNA isolated from 20 to 25 36-hour wild-type or fbn1-MO–injected embryos to obtain cDNA. To assess endogenous fbn1 transcript levels, the primers used for PCR were 5′-CAATGAGTGTGAGGTGTTTC-3′ and 5′-CAGAGCTGAGTGTACTCC-3′. Transcript levels of zebrafish EF1α were used as a PCR control with the primers 5′-GGACACAGAGACTTCATCA-3′ and 5′-GGCATCAAGAAGAGTAGT-3′. The touchdown PCR program (see “Cloning of zebrafish magp1”) was used for PCR reactions.
Microangiography
Fluorescein isothiocyanate (FITC) dextran dye or tetramethylrhodamine isothiocyanate–dextran (TRITC) dye at 20 mg/mL in 0.3 × Danieau buffer was microinjected into the common cardinal vein of zebrafish embryos 44 to 48 hours after fertilization, as described.33 Microangiograms were visualized using the bandpass FITC and the rhodamine filter sets, as previously described.34
In situ hybridization
cDNA for smooth muscle 22α (sm22a38 ; GB: AAP79113) was amplified from the cDNA library of a zebrafish 24 hours after fertilization using primers 5′-ACTACTGAGCAGGGTTTCT-3′ and 5′-CAACTGGTCAGTCTTTCTG-3′. The PCR fragment was cloned into the TOPO vector (Invitrogen). The plasmid was then linearized with NotI. DIG-labeled antisense RNA was synthesized using the T3 polymerase in conjunction with the in vitro DIG labeling kit (Roche). Zebrafish in situ hybridization was performed as previously described.39
For sectioning, embryos were embedded in molten paraplast (Sigma). Tissue sections measuring 7 μm each were placed on superfrost microscope slides (Fisher Scientific, Hanover Park, IL), cleared with toluene, and mounted using Permount (Sigma).
Histologic analyses
Zebrafish embryos at 44 to 48 hours after fertilization were fixed in 4% paraformaldehye in PBS overnight, were washed twice in toluene (5-10 minutes each wash), and were embedded in paraffin. Tissue sections at 7 μM were obtained for histologic processing. Visualization of elastin staining was performed using a protocol adapted from the manufacturer (modified Verhoeff van Gieson Elastin Stain Kit, HT25A; Sigma). Briefly, once the deparaffinized tissues were rehydrated, the slides were placed in the working elastin solution (20 mL hematoxylin, 3 mL ferric chloride, 8 mL Weigert iodine solution, and 5 mL deionized water for 36 mL working solution) for 5 to 10 minutes. After a rinse in tap water, tissues were differentiated in working ferric chloride solution (3 mL ferric chloride solution and 37 mL deionized water for 40 mL working solution) for approximately 2 minutes. Slides were then rinsed in tap water and 95% ethanol, followed by staining in van Gieson solution for approximately 1 minute. Tissues were dehydrated in 95% ethanol, cleared in toluene, and mounted in Permount (Sigma). Paraffinized embryo sections at 7 μm were obtained and processed for hematoxylin and eosin with the use of standard procedures.33
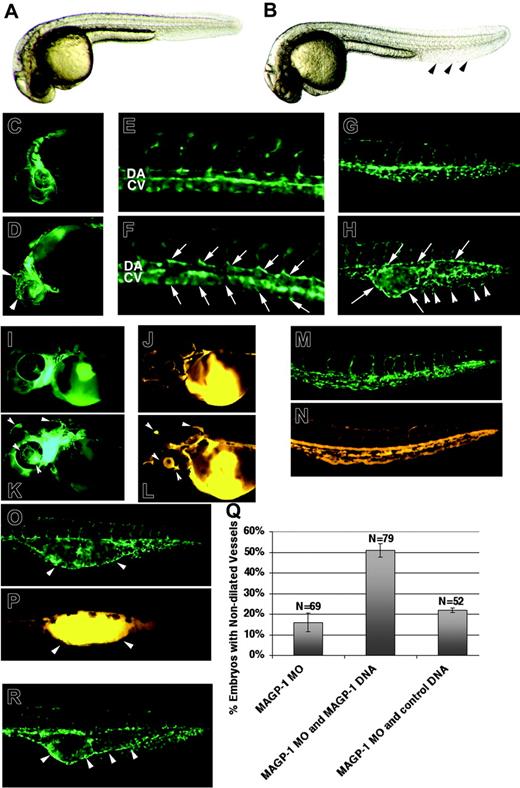
Underexpression and overexpression of magp1 result in vascular defects in zebrafish embryos. Bright-field images of an uninjected Tg(fli1/EGFP) embryo (A) and an magp1 MO–injected embryo (B) at 30 hours after fertilization. magp1 MO–injected embryos display normal morphology, except for the expanded tail fin (arrowheads). Compared with the uninjected Tg(fli1/EGFP) embryo (C, E, G), magp1 MO–injected Tg(fli1/EGFP) embryos exhibit distended vessels in the brain and the eye (D, arrowheads; 47% ± 16%; N = 60), axial vessels with irregular lumens (F, arrows; 70% ± 3%; N = 36), and dilated vascular channel in the venous plexus (H, arrows) with only a few branches of venous microvessels (H, arrowheads; 96% ±;N = 84). DA indicates dorsal aorta; CV, cardinal vein. To assess functionality of the circulation, microangiography was conducted on Tg(fli1/EGFP) embryos (I, M, uninjected embryos; K, O, magp1 MO–injected embryos) at 48 hours after fertilization using TRITC dextran. Compared with uninjected Tg(fli-1/EGFP) embryos (J, N), magp1 MO–injected Tg(fli1/EGFP) embryos (L, P) display lumenized vasculature. However, pooling of TRITC dextran in dilated regions of the blood vessels is observed in magp1 MO–injected Tg(fli1/EGFP) embryos (L, P, arrowheads). The corresponding region of dilated vessels for panel P is shown in panel O as indicated by arrowheads. (Q) Summary of 3 independent experiments showing alleviation of 3 ng magp1 MO–induced vascular defects with the introduction of Magp1 protein through injection of 10 pg magp1 expression construct (compare columns 2 and 1; P = .01). N indicates the number of embryos scored. In contrast, coinjection of 10 pg control expression construct, FRM-DsRED, does not affect the frequency of the magp1 MO–induced vascular defects (column 3). Error bars indicate SEM. (R) Overexpression of magp1 generates a range of vascular defects, including the dilation of caudal vein (arrowheads). Original magnifications: (A-D, G-H, I-P, R) 5 ×; (E-F) 10 ×.
Underexpression and overexpression of magp1 result in vascular defects in zebrafish embryos. Bright-field images of an uninjected Tg(fli1/EGFP) embryo (A) and an magp1 MO–injected embryo (B) at 30 hours after fertilization. magp1 MO–injected embryos display normal morphology, except for the expanded tail fin (arrowheads). Compared with the uninjected Tg(fli1/EGFP) embryo (C, E, G), magp1 MO–injected Tg(fli1/EGFP) embryos exhibit distended vessels in the brain and the eye (D, arrowheads; 47% ± 16%; N = 60), axial vessels with irregular lumens (F, arrows; 70% ± 3%; N = 36), and dilated vascular channel in the venous plexus (H, arrows) with only a few branches of venous microvessels (H, arrowheads; 96% ±;N = 84). DA indicates dorsal aorta; CV, cardinal vein. To assess functionality of the circulation, microangiography was conducted on Tg(fli1/EGFP) embryos (I, M, uninjected embryos; K, O, magp1 MO–injected embryos) at 48 hours after fertilization using TRITC dextran. Compared with uninjected Tg(fli-1/EGFP) embryos (J, N), magp1 MO–injected Tg(fli1/EGFP) embryos (L, P) display lumenized vasculature. However, pooling of TRITC dextran in dilated regions of the blood vessels is observed in magp1 MO–injected Tg(fli1/EGFP) embryos (L, P, arrowheads). The corresponding region of dilated vessels for panel P is shown in panel O as indicated by arrowheads. (Q) Summary of 3 independent experiments showing alleviation of 3 ng magp1 MO–induced vascular defects with the introduction of Magp1 protein through injection of 10 pg magp1 expression construct (compare columns 2 and 1; P = .01). N indicates the number of embryos scored. In contrast, coinjection of 10 pg control expression construct, FRM-DsRED, does not affect the frequency of the magp1 MO–induced vascular defects (column 3). Error bars indicate SEM. (R) Overexpression of magp1 generates a range of vascular defects, including the dilation of caudal vein (arrowheads). Original magnifications: (A-D, G-H, I-P, R) 5 ×; (E-F) 10 ×.
Sequence analysis
Phylogenetic analysis of MAGP proteins was conducted using the CLUSTAL W algorithm in the Macvector 7.2.3 software (Accelrys, San Diego, CA). The value at each branch denotes the mean number of differences per residue along each branch.40
Injection of the integrin antagonist Echistatin
The native Echistatin peptide was purchased from Sigma and reconstituted in PBS, pH 7.5. Echistatin was first mixed with 50 mM cascade blue dextran (molecular weight [MW], 10 000; Molecular Probes, Eugene, OR) to achieve a final concentration of 20 μM. The heat-attenuated form of Echistatin (Sigma) was obtained by incubating the Echistatin solution at 100°C for 20 minutes. The Echistatin (Sigma)/dextran solution (9 nL) was injected into the common cardinal vein of the embryo at 24 to 25 hours after fertilization. Embryos successfully injected with this solution, as visualized using the blue GFP bandpass filter,34 were used for phenotype analysis at 30 hours after fertilization.
Image acquisition and manipulation
All bright-field and fluorescent images were captured using a Zeiss Axiocam HR camera (early version, r1.4; Carl Zeiss, Oberkochen, Germany) mounted on a Zeiss Axioskop2 microscope equipped with a 5 ×/0.25 numeric aperture (NA) or a 10 ×/0.45 NA objective and using Zeiss Axiovision 4.2 software. For Figure 5D-G, images were obtained using an Axioskop2 adapted with a Zeiss apotome deconvolution slider, equipped with a 10 ×/0.45 NA or a 20 ×/0.75 NA objective. In situ hybridization and histologic images were captured using a Nikon Coolpix 990 camera (Nikon, Tokyo, Japan) with a Zeiss Axioplan2 microscope equipped with a 10 ×/0.45 NA or a 20 ×/0.75 objective, and were rendered with Adobe Photoshop 7.0 (Adobe Systems, San Jose, CA).
Results
Underexpression and overexpression of magp1 in zebrafish embryos result in vascular defects
In transgenic (fli1/EGFP) embryos, the fli1 promoter drives the expression of the EGFP reporter gene in the endothelium that lines the vessel walls of the developing vasculature.41 In our ongoing morpholino (MO)–based vascular screen, morpholinos were injected into Tg(fli1/EGFP) embryos to facilitate the identification of novel genes essential for vascular development. magp1 MO–injected Tg(fli1/EGFP) embryos showed normal overall morphology except for the enlarged caudal fins (Figure 1B). However, an examination of magp1 MO–injected Tg(fli1/EGFP) embryos under the GFP filter set revealed vascular defects, including dilated vessels in the brain and the eyes (Figure 1D, arrowheads), irregular lumens of axial vessels (Figure 1F, arrows), and a dilated caudal vein (Figure 1H, arrows) with altered venous plexus formation (Figure 1H, arrowheads). Microangiography was conducted to assess the integrity of the circulation in magp1 MO–injected embryos. Lumenized vessels were observed throughout magp1 MO–injected embryos (Figure 1P). However, pooling of the microangiography dye was observed in severely dilated vessels (arrowheads in Figure 1L,P), suggesting compromised vessel wall function. No extravasation of red blood cells was observed (Figure S1; see the Supplemental Figure link at the top of the online article, at the Blood website).
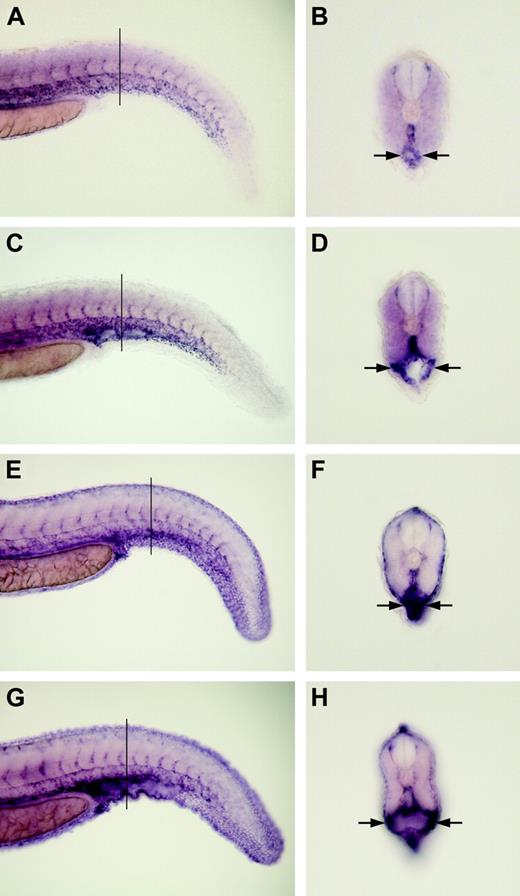
Magp1 is not required for specifying or maintaining the expression of markers for vascular endothelium and smooth muscles. In situ hybridization was conducted to analyze the expression of vascular markers. Expression of flk1 in a wild-type embryo (A-B) and an magp1 MO–injected embryo (C-D) at 31 hours after fertilization. Cross-sections of panels B and D revealed normal expression levels of flk1 in the endothelium lining the dilated caudal vein of magp1 morphant embryos (frequency with dilated caudal vein: 68% ± 7%; N = 47). Expression of sm22a in a wild-type embryo (E-F) and an magp1 MO–injected embryo (G-H) at 31 hours after fertilization. Cross-sections of panels F and H revealed that magp1 morphant embryos displayed normal expression levels of sm22a surrounding the dilated caudal vein (frequency with dilated caudal vein: 78% ± 10%; N = 34). Arrows indicate the dilated caudal veins in magp1 morphant embryos. Lines in panels A, C, E, and G indicate levels of sections in panels B, D, F, and H, respectively. (A-H) Original magnifications, 10 ×.
Magp1 is not required for specifying or maintaining the expression of markers for vascular endothelium and smooth muscles. In situ hybridization was conducted to analyze the expression of vascular markers. Expression of flk1 in a wild-type embryo (A-B) and an magp1 MO–injected embryo (C-D) at 31 hours after fertilization. Cross-sections of panels B and D revealed normal expression levels of flk1 in the endothelium lining the dilated caudal vein of magp1 morphant embryos (frequency with dilated caudal vein: 68% ± 7%; N = 47). Expression of sm22a in a wild-type embryo (E-F) and an magp1 MO–injected embryo (G-H) at 31 hours after fertilization. Cross-sections of panels F and H revealed that magp1 morphant embryos displayed normal expression levels of sm22a surrounding the dilated caudal vein (frequency with dilated caudal vein: 78% ± 10%; N = 34). Arrows indicate the dilated caudal veins in magp1 morphant embryos. Lines in panels A, C, E, and G indicate levels of sections in panels B, D, F, and H, respectively. (A-H) Original magnifications, 10 ×.
To investigate the consequence on vascular development of overexpressing magp1, magp1 expression construct was injected into zebrafish embryos. magp1 DNA–injected embryos showed a range of vascular defects, including a reduction in the caudal vein plexus formation and the dilation of the caudal vein (Figure 1R, arrowheads; data not shown). Underexpression and overexpression studies of Magp1 function indicated overlapping vascular phenotypes, namely vessel dilation (Table 1). Overexpression of magp1 DNA (25 ng) resulted in dilated CV in 24% ± 1% of embryos, and reduced CV in 24% ± 1% of embryos (N = 34; 2 independent experiments), at 30 hours after fertilization.
Summary of vascular defects in magp1 morphant embryos at 30 hours after fertilization
Axial vessels . | Head vessels . | Venous plexus . | MO1, % . | MO2, % . |
|---|---|---|---|---|
| - | - | + | 23 ± 6 | 16 ± 8 |
| - | + | - | 6 ± 5 | 4 ± 3 |
| + | - | - | 3 ± 2 | 4 ± 3 |
| - | + | + | 18 ± 11 | 8 ± 1 |
| + | + | + | 32 ± 7 | 19 ± 4 |
Axial vessels . | Head vessels . | Venous plexus . | MO1, % . | MO2, % . |
|---|---|---|---|---|
| - | - | + | 23 ± 6 | 16 ± 8 |
| - | + | - | 6 ± 5 | 4 ± 3 |
| + | - | - | 3 ± 2 | 4 ± 3 |
| - | + | + | 18 ± 11 | 8 ± 1 |
| + | + | + | 32 ± 7 | 19 ± 4 |
Two independent experiments were performed. MO1 (N = 35) was injected at 6 ng with the percentage of the total vascular defects at 80% ± 8%. MO2 (N = 26) was injected at 3 ng with the percentage of the total vascular defects at 58% ± 4%. Plus-minus values indicate SD.
- indicates absence of the phenotype; +, presence of the phenotype.
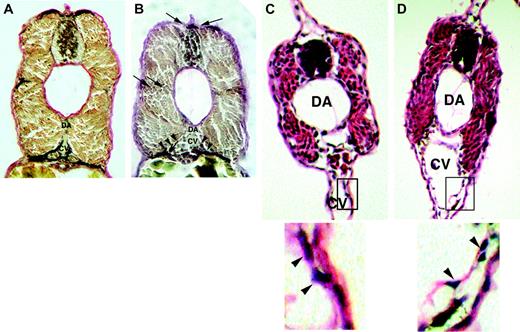
Histologic analysis reveals altered vessel wall architecture in magp1 morphant embryos. Elastin (van Gieson) stain was conducted on fixed sections from 48-hour wild-type and MO–injected embryos. Compared with wild-type embryos (A), magp1 MO–injected embryos (B) showed reduced elastin stain around the vasculature (arrowheads) in the intersomitic region and the connective tissues attached to the dorsal neural tube (arrows). Hematoxylin and eosin staining was performed at 40 hours after fertilization to assess vessel wall structure in magp1 morphant embryos. Compared with wild-type embryos (C, inset in the bottom panel), the vascular cells in the magp1 morphant embryos show a loose association with the surrounding tissue. Cells detaching from the underlying tissue can be observed (D, arrowheads in the inset, bottom panel). Original magnification 20 ×. Bottom panels of C and D are enlarged versions of the boxed sections of the top panels.
Histologic analysis reveals altered vessel wall architecture in magp1 morphant embryos. Elastin (van Gieson) stain was conducted on fixed sections from 48-hour wild-type and MO–injected embryos. Compared with wild-type embryos (A), magp1 MO–injected embryos (B) showed reduced elastin stain around the vasculature (arrowheads) in the intersomitic region and the connective tissues attached to the dorsal neural tube (arrows). Hematoxylin and eosin staining was performed at 40 hours after fertilization to assess vessel wall structure in magp1 morphant embryos. Compared with wild-type embryos (C, inset in the bottom panel), the vascular cells in the magp1 morphant embryos show a loose association with the surrounding tissue. Cells detaching from the underlying tissue can be observed (D, arrowheads in the inset, bottom panel). Original magnification 20 ×. Bottom panels of C and D are enlarged versions of the boxed sections of the top panels.
Specificity of the vascular phenotypes in magp1 morphant embryos
Of 208 gene targets tested using 296 distinct morpholino oligos, only the morpholinos against magp1 generated vascular phenotypes, as described in “Underexpression and overexpression of magp1 in zebrafish embryos result in vascular defects.” To confirm the specificity of the magp1 MO, a second nonoverlapping magp1 MO was injected and generated a similar range of vascular phenotypes (85% ± 4% showing dilated vessels at 1.8 ng; N = 40). To assess whether the presence of the Magp1 protein would ameliorate the vascular phenotypes observed in magp1 MO–injected embryos, an magp1 expression construct was coinjected with magp1 MO into embryos. The magp1 MO used in the injection experiments was designed against the 5′ untranslated region (UTR) of the magp1 cDNA to avoid inhibition of the magp1 expression construct. The dose of the magp1 expression construct used did not elicit overexpression phenotypes when it was introduced into zebrafish embryos alone. Compared with embryos injected with the magp1 MO alone, a higher frequency of coinjected embryos showed nondilated vessels (Figure 1Q; compare columns 2 and 1; P = .01). In contrast, coinjection with a control FRM-DsRed reporter construct did not significantly affect the frequency of embryos with vascular defects (Figure 1Q; compare columns 3 and 1; P = .25). These results suggest that the vascular phenotypes observed in magp1 MO–injected embryos are specific to the loss of the Magp1 protein.
Magp1 is not required for the specification and the maintenance of vascular cell fate. To understand the molecular basis of the vascular phenotypes in magp1 morphant embryos, in situ hybridization was conducted to analyze the expression of several markers of different vascular cell types. flk1, a vascular endothelial growth factor receptor-2 (VEGFR-2), is expressed in the endothelium of axial vessels, the dorsal aorta, the cardinal vein, and the nascent sprouts of the intersegmental vessels from the dorsal aorta.42 The expression of flk1 was normal in magp1 morphant embryos at 24 hours after fertilization (N = 34; data not shown). Endothelial cells that line the dilated vessels in magp1 morphant embryos retained the expression of flk1 at 30 to 31 hours after fertilization (Figure 2E-F).
During embryogenesis, mature vasculature is characterized by association of the endothelium with the smooth muscle cells. The expression of the vascular smooth muscle marker, smooth muscle 22α (sm22a), was normal in magp1 morphant embryos at 24 hours after fertilization (n = 52; data not shown). Normal expression of sm22a was also detected around the dilated vessels in the caudal region of magp1 morphant embryos at 30 to 31 hours (Figure 2G-H). These results suggest that Magp1 is not required for specifying and maintaining the fate of vascular cell types.
MAGP1 is required for maintaining the structural integrity of the vessel wall and proper elastic fiber formation. It has been proposed that MAGP1 binds to tropoelastin, the precursor of elastin, so as to facilitate proper alignment of elastic fibers.26 The formation of elastic fibers was examined using a histologic stain for elastin (van Gieson stain) in magp1 morphant embryos. Wild-type embryos did not show van Gieson–stained elastin before 40 hours after fertilization, suggesting that maturation of the elastic fibers does not occur until relatively late in embryonic development. Compared with wild-type embryos, the elastic fibers were fragmented around the region where dilated vessels were observed in magp1 morphant embryos (Figure 3B, arrowheads) at approximately 48 hours after fertilization. This result suggests an important role played by Magp1 in elastic fiber assembly during vascular development.
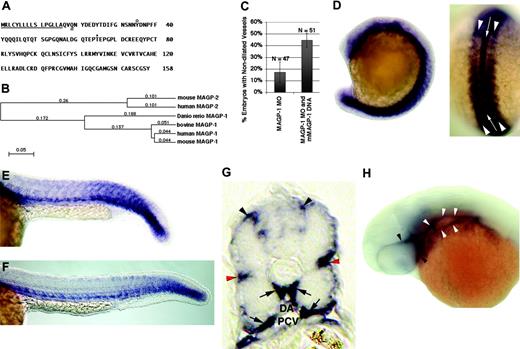
Sequence of zebrafish Magp1 protein and expression pattern of zebrafish magp1 mRNA during embryonic development. (A) Predicted amino acid sequence of zebrafish Magp1 protein. The predicted amino-terminal signal sequence is underlined. The predicted site of transglutamination at a glutamine residue is indicated by a double underline. The predicted tyrosine sulfation site is indicated by a circle. Predicted O-glycosylation site at a threonine residue. Sites of modifications are predicted based on the amino acid sequence analysis of the bovine MAGP1 protein.27 (B) Phylogenetic tree of vertebrate MAGP proteins. Zebrafish Magp1 clusters with vertebrate MAGP1 proteins. Mouse MAGP1 GenBank accession no. P55002; human MAGP1, P55001; bovine MAGP1, P27424; human MAGP2 (also known as MFAP5), NP_003471; mouse MAGP2, NP_056591. The value at each branch denotes the mean number of differences per residue along each branch. (C) Summary of 2 independent experiments showing the alleviation of zebrafish magp1 MO–generated vascular phenotypes by the coinjection of mouse magp1 expression construct (compare columns 2 and 1). N denotes the number of embryos scored. Error bars indicate SEM. (D) At 16-somite stage (dorsal view shown on the right), magp1 is expressed in the notochord (white arrow in the dorsal view) and the somites (white arrowheads in the dorsal view). (E) At 22 hours after fertilization or 26-somite stage, expression of magp1 in the somites begins to be down-regulated. Expression of magp1 is also detected in the mesoderm. (F) At 24 hours after fertilization (end of somitogenesis), expression of magp1 is detected in the caudal somites and the trunk mesenchyme. (G) A cross-section of a 24-hour embryo showing the expression of magp1 in the connective tissue between the neural tube, around somites (red arrowheads), ventral medial somites, and cells surrounding the dorsal aorta (DA) and the posterior cardinal vein (PCV) (arrows). (H) Expression of magp1 in the blood vessels posterior to the eyes (white arrowheads) and around the eyes (black arrowheads) is also detected at 33 hours after fertilization. For panels E, F, and H, anterior is to the left. Original magnifications: (D-F, H) 10 ×; (G) 20 ×.
Sequence of zebrafish Magp1 protein and expression pattern of zebrafish magp1 mRNA during embryonic development. (A) Predicted amino acid sequence of zebrafish Magp1 protein. The predicted amino-terminal signal sequence is underlined. The predicted site of transglutamination at a glutamine residue is indicated by a double underline. The predicted tyrosine sulfation site is indicated by a circle. Predicted O-glycosylation site at a threonine residue. Sites of modifications are predicted based on the amino acid sequence analysis of the bovine MAGP1 protein.27 (B) Phylogenetic tree of vertebrate MAGP proteins. Zebrafish Magp1 clusters with vertebrate MAGP1 proteins. Mouse MAGP1 GenBank accession no. P55002; human MAGP1, P55001; bovine MAGP1, P27424; human MAGP2 (also known as MFAP5), NP_003471; mouse MAGP2, NP_056591. The value at each branch denotes the mean number of differences per residue along each branch. (C) Summary of 2 independent experiments showing the alleviation of zebrafish magp1 MO–generated vascular phenotypes by the coinjection of mouse magp1 expression construct (compare columns 2 and 1). N denotes the number of embryos scored. Error bars indicate SEM. (D) At 16-somite stage (dorsal view shown on the right), magp1 is expressed in the notochord (white arrow in the dorsal view) and the somites (white arrowheads in the dorsal view). (E) At 22 hours after fertilization or 26-somite stage, expression of magp1 in the somites begins to be down-regulated. Expression of magp1 is also detected in the mesoderm. (F) At 24 hours after fertilization (end of somitogenesis), expression of magp1 is detected in the caudal somites and the trunk mesenchyme. (G) A cross-section of a 24-hour embryo showing the expression of magp1 in the connective tissue between the neural tube, around somites (red arrowheads), ventral medial somites, and cells surrounding the dorsal aorta (DA) and the posterior cardinal vein (PCV) (arrows). (H) Expression of magp1 in the blood vessels posterior to the eyes (white arrowheads) and around the eyes (black arrowheads) is also detected at 33 hours after fertilization. For panels E, F, and H, anterior is to the left. Original magnifications: (D-F, H) 10 ×; (G) 20 ×.
To assess the integrity of the vessel wall architecture in magp1 morphant embryos, hematoxylin and eosin staining was applied. The association among cells lining the dilated vessels was looser in morphant embryos than in wild-type embryos. Detachment of the cells from the underlying tissue was observed (Figure 3D, bottom arrowheads). A reduction in or a lack of venous plexus formation in magp1 morphant embryos was indicated by the enlarged lumen of a cavernous vessel in the caudal vein region (Figure 3D). These results suggested that Magp1 is required for maintaining the structural integrity of the vessel wall and venous plexus morphogenesis.
Identification and mRNA expression analysis of magp1
The zebrafish magp1 gene was originally identified from our ongoing morpholino-based screen. The morpholino targeted against the magp1 gene was designed based on the 5′ UTR identified in the cDNA for the magp1 gene. Full-length zebrafish magp1 cDNA was subsequently isolated from the cDNA library of an embryo 24 hours after fertilization (Figure 3A; see “Materials and methods”). Based on amino acid sequence comparisons, zebrafish Magp1 clustered to the vertebrate MAGP1 family (Figure 4B) with 61% identity to mouse MAGP1 (MFAP2) and 61% identity to human MAGP1 (MFAP2). The function of MAGP1 protein was likely conserved because the introduction of mouse MAGP1 by the injection of mouse MAGP1 expression construct alleviated the vascular defects in zebrafish magp1 morphant embryos (Figure 4C; compare columns 2 and 1; P = .04).
In situ hybridization was conducted to determine the expression pattern of zebrafish magp1 mRNA during embryonic development. At the 16-somite stage, magp1 is expressed in the somitic mesoderm (Figure 4D, arrowheads in the dorsal view) and the notochord (Figure 4D, white arrow in dorsal view). At 22 hours after fertilization (26-somite stage), magp1 expression in the mesendoderm persisted while its expression in the somites began to be down-regulated because it was detected only in the caudal somites at this stage (Figure 4E). At 24 hours after fertilization, the expression of magp1 was detected in the caudal-most somites, the ventromedial somites, and the cells surrounding the dorsal aorta and the cardinal vein (Figure 4F-G, arrows). Examination of cross-sections from an embryo 24 hours after fertilization also revealed magp1 expression in the connective tissues between the somites and the neural tube (Figure 4G, top arrowheads) and the intersomitic region (Figure 4G, red arrowheads). This localized expression of magp1 marked the regions of elastic fiber formation (compare Figures 4G and 3C). Expression of magp1 along the trunk persisted at 33 hours after fertilization (data not shown) and was also detected in vessels around the eye and in those posterior to the eye (Figure 4H, white and black arrowheads, respectively), where dilation was observed in magp1 morphants.
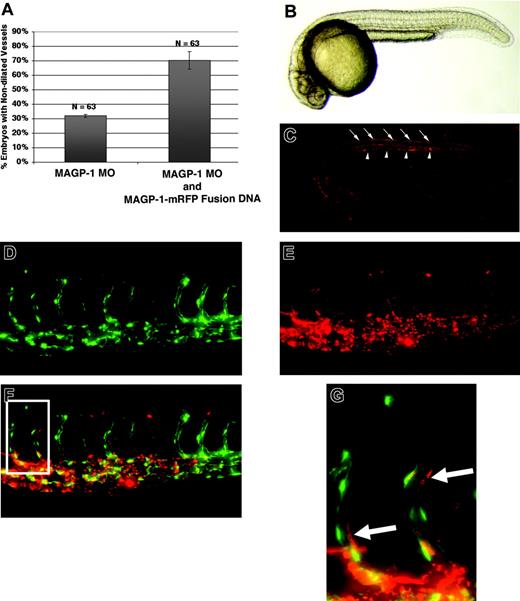
Distribution of functional Magp1-mRFP protein during early development. DNA encoding the Magp1-mRFP fusion construct was injected into wild-type embryos, yielding a mosaic distribution of the fusion protein as observed at 12 hours after fertilization (data not shown). By 24 hours, the Magp1-mRFP fusion protein exhibited localized tissue distribution. (A) Coinjection of the Magp1-mRFP fusion construct alleviated the vascular defects in magp1 morphant embryos (compare columns 2 and 1). Error bars indicate SEM. (B) Bright-field image of a wild-type embryo at 24 hours after fertilization. (C) Wild-type embryo injected with 12 pg Magp1-mRFP fusion construct under rhodamine illumination, showing enrichment of the Magp1 protein around the notochord (arrowheads) and in the dorsal connective tissues (arrows). (D) Tg(fli1/EGFP) embryo injected with 12 pg Magp1-mRFP fusion construct, showing the endothelium-lined vasculature under FITC illumination. (E) Distribution of Magp1-mRFP fusion protein is visualized under the rhodamine filter. (F) Overlay of panels D and E, showing the enrichment of the Magp1-mRFP fusion protein around the developing vasculature. (G) Inset from panel F, as indicated by the rectangle in panel F, showing the Magp1-mRFP fusion protein to be in close association with the endothelial cells (arrows). Anterior is to the left for all panels. (B-G) Original magnifications, 5 ×.
Distribution of functional Magp1-mRFP protein during early development. DNA encoding the Magp1-mRFP fusion construct was injected into wild-type embryos, yielding a mosaic distribution of the fusion protein as observed at 12 hours after fertilization (data not shown). By 24 hours, the Magp1-mRFP fusion protein exhibited localized tissue distribution. (A) Coinjection of the Magp1-mRFP fusion construct alleviated the vascular defects in magp1 morphant embryos (compare columns 2 and 1). Error bars indicate SEM. (B) Bright-field image of a wild-type embryo at 24 hours after fertilization. (C) Wild-type embryo injected with 12 pg Magp1-mRFP fusion construct under rhodamine illumination, showing enrichment of the Magp1 protein around the notochord (arrowheads) and in the dorsal connective tissues (arrows). (D) Tg(fli1/EGFP) embryo injected with 12 pg Magp1-mRFP fusion construct, showing the endothelium-lined vasculature under FITC illumination. (E) Distribution of Magp1-mRFP fusion protein is visualized under the rhodamine filter. (F) Overlay of panels D and E, showing the enrichment of the Magp1-mRFP fusion protein around the developing vasculature. (G) Inset from panel F, as indicated by the rectangle in panel F, showing the Magp1-mRFP fusion protein to be in close association with the endothelial cells (arrows). Anterior is to the left for all panels. (B-G) Original magnifications, 5 ×.
Distribution of a functional Magp1–mRFP protein during embryonic development
To determine the distribution of Magp1 protein during development, an Magp1 mRFP fusion expression construct was used. The Magp1-mRFP fusion protein retained the native activity of the Magp1 protein—introduction of the Magp1-mRFP expression construct into magp1 morphant embryos alleviated the vascular phenotypes observed (Figure 5A; P = .04). To visualize the distribution of Magp1 protein during development, the Magp1-mRFP fusion construct was injected into zebrafish embryos. DNA injection resulted in mosaic distribution of Magp1-mRFP fusion protein, as observed at 12 hours after fertilization (data not shown). However, localized expression of the fusion protein was observed at 24 hours after fertilization. The locations of Magp1 protein distribution coincided with endogenous magp1 mRNA–expressing cells, such as in the notochord (arrowheads in Figure 5C). Later, at 48 hours after fertilization, localized distribution of Magp1-mRFP fusion protein around the developing vasculature was observed (Figure 5D-G), supporting the critical function of Magp1 in vascular development. The localized distribution of Magp1 protein likely resulted from incorporation in localized microfibril complexes and likely reflected the endogenous expression pattern of this protein.
Integrins and Magp1 interact in vivo during vascular development
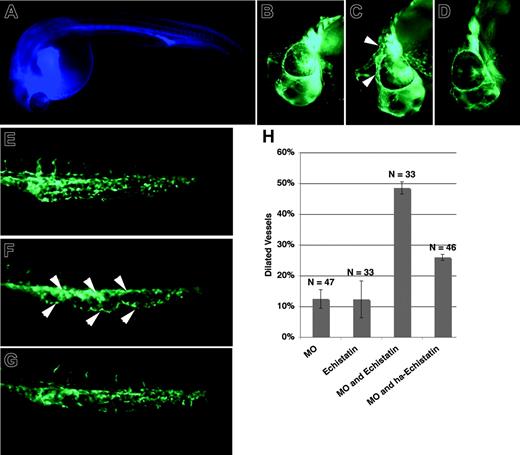
Localized distribution of Magp1-mRFP protein around the developing vasculature was observed, prompting us to question whether Magp1 influenced cell–cell or cell–matrix interaction in the developing vasculature. The integrins are heterodimeric receptors to ECM molecules, and α5β1 and αvβ3 integrins have been shown by in vitro studies to be essential for cell adhesion to microfibrils.43,44 To assess whether integrin and Magp1 were capable of functionally interacting in vivo, we introduced the integrin antagonist Echistatin (Sigma) into embryos injected with magp1 MO at limiting doses, where little effect on vascular development was noted with the injection of either antagonist alone. Echistatin (Sigma) is a disintegrin isolated from snake venom and has been previously shown to interfere with integrin function during the growth and pathfinding of ventral nerves in zebrafish embryos.45 At 24 to 25 hours after fertilization, when circulation began in zebrafish embryos, Echistatin (Sigma) was injected into the common cardinal vein by microangiography. The dose of Echistatin (Sigma) or magp1 MO individually injected into embryos resulted in little or no vascular defects (Figure 6H, columns 1-2). In contrast, embryos coinjected with these doses of magp1 MO and Echistatin (Sigma) showed a synergistic increase in embryos with vessel dilation (Figure 6C, F, H column 3). Coinjection of heat-attenuated Echistatin (Sigma) abolished this synergy with magp1 MO (Figure 6D, G, H column 4), indicating that the inhibitory effect of Echistatin (Sigma) was specific. This suggested that the likely mechanism of the vascular defects noted in magp1-morphant embryos included a contribution of integrin-mediated cell–matrix interactions.
Magp1 and integrin functionally interact in vivo during vascular development. A mixture of the disintegrin Echistatin and blue dextran was injected into the common cardinal veins of wild-type and magp1 MO–injected Tg(fli1/EGFP) embryos at 24 to 25 hours after fertilization by microangiography. Embryos successfully injected, as indicated by the blue fluorescence in the circulation (A), were selected for phenotypic analysis at 30 hours after fertilization. The head (B) and the tail (E) of a Tg(fli1/EGFP) embryo at 30 hours after fertilization. The head (C) and the tail (F) of a Tg(fli1/EGFP) embryo injected with 1 ng magp1 MO and Echistatin showing dilation of vessels in the midbrain–hindbrain region, around the eye (C, arrowheads) and the caudal vein (F, arrowheads). The head (D) and the tail (G) of a Tg(fli1/EGFP) embryo injected with 1 ng magp1 MO and heat-attenuated Echistatin, exhibiting no significant change in the vasculature compared with wild-type embryos. (H) Summary of 2 independent injection experiments; 1 ng magp1 morpholino and 9 nL 20 μM Echistatin solution were injected (see “Injection of the integrin antagonist Echistatin”). HA indicates heat-attenuated; N, number of embryos scored. Error bars indicate SEM. (A-G) Original magnifications, 10 ×.
Magp1 and integrin functionally interact in vivo during vascular development. A mixture of the disintegrin Echistatin and blue dextran was injected into the common cardinal veins of wild-type and magp1 MO–injected Tg(fli1/EGFP) embryos at 24 to 25 hours after fertilization by microangiography. Embryos successfully injected, as indicated by the blue fluorescence in the circulation (A), were selected for phenotypic analysis at 30 hours after fertilization. The head (B) and the tail (E) of a Tg(fli1/EGFP) embryo at 30 hours after fertilization. The head (C) and the tail (F) of a Tg(fli1/EGFP) embryo injected with 1 ng magp1 MO and Echistatin showing dilation of vessels in the midbrain–hindbrain region, around the eye (C, arrowheads) and the caudal vein (F, arrowheads). The head (D) and the tail (G) of a Tg(fli1/EGFP) embryo injected with 1 ng magp1 MO and heat-attenuated Echistatin, exhibiting no significant change in the vasculature compared with wild-type embryos. (H) Summary of 2 independent injection experiments; 1 ng magp1 morpholino and 9 nL 20 μM Echistatin solution were injected (see “Injection of the integrin antagonist Echistatin”). HA indicates heat-attenuated; N, number of embryos scored. Error bars indicate SEM. (A-G) Original magnifications, 10 ×.
To assess whether Magp1 influences cell–cell adhesion interaction in the developing vasculature, morpholinos against the vascular cadherin gene VE-cadherin46 and magp1 MO were coinjected into embryos. In contrast to work with the integrin antagonist, no synergy was noted when VE-cadherin and Magp1 function were partially compromised (data not shown). This suggests that a deficiency in cadherin-based adhesion is not a primary mechanism underlying the vascular phenotypes observed in magp1 morphant embryos. However, the possibility of functional redundancy among the cadherins cannot be excluded from these analyses.
Comparison of zebrafish fibrillin-1 (fbn1) and magp1 morphant phenotypes
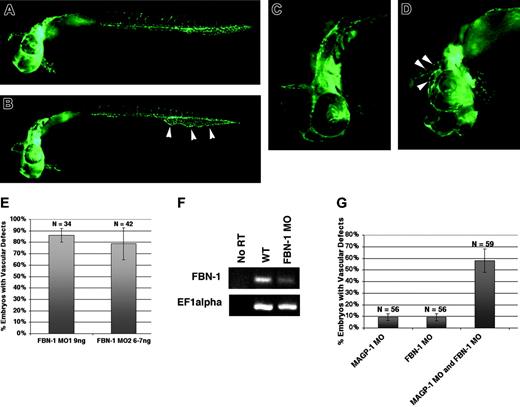
MAGP1 is one of the proteins that associate with fibrillin-containing microfibrils.19 Direct physical interaction between FBN1 and MAGP1 has also been demonstrated using in vitro coimmunoprecipitation assay.28 This raises the possibility that FBN1 and MAGP1 may play a collaborative role during vascular development. To test this hypothesis, the phenotypes of fbn1 MO–injected embryos were analyzed and compared with those of magp1 morphant embryos. Because the 5′ UTR sequence of zebrafish fibrillin-1 is unavailable in the zebrafish database, 2 independent splice-site morpholinos against the zebrafish fbn1 gene were used to generate knockdown embryos. To investigate the role of fibrillin-1 on zebrafish vascular development, fbn1 MO was injected into Tg(fli1/EGFP) embryos. Similar to magp1 MO–injected Tg(fli1/EGFP) embryos, fbn1 MO–injected Tg(fli1/EGFP) embryos displayed dilated caudal veins with reduced or no venous plexus formation (Figure 7B, arrowheads) and dilated vessels in the head and the eyes (Figure 7D, arrowheads) at 30 to 31 hours after fertilization. The 2 splice-site fbn1 MOs generated similar vascular defects at high frequency (Figure 7E).
The efficacy of gene targeting by the splice-site fbn1 MO was assessed by examining the levels of endogenous fbn1 transcript in fbn1 MO–injected embryos using RT-PCR. Compared with the uninjected wild-type embryos, fbn1 MO–injected embryos showed depletion of the wild-type fbn1 transcript by RT-PCR analysis (Figure 7F). The second splice-site fbn1 MO, targeted against another exon–intron boundary of the fbn1 gene, generated a truncated fibrillin-1 protein, as evidenced by the presence of the aberrantly spliced transcript (data not shown). These results suggest that the splice-site fbn1 MOs effectively inhibited normal splicing of the wild-type transcript.
To assess whether Magp1 and fibrillin-1 interact in vivo during vascular development, low doses of magp1 MO and fbn1 MO were coinjected into Tg(fli-1/EGFP) embryos. Compared with embryos injected with magp1 MO or fbn1 MO alone, coinjected embryos showed a synergistic increase in the frequency of embryos with vascular defects (Figure 7G), suggesting that Magp1 and Fbn1 interact in vivo during vascular development.
Phenotypic analysis of fibrillin-1 (fbn1) morphant embryos. (A) Uninjected Tg(fli1/EGFP) embryo at 31 hours after fertilization. (B) fbn1 MO–injected Tg(fli1/EGFP) embryo at 31 hours after fertilization, showing dilated caudal vein with reduced venous plexus formation (arrowheads). Frequency, 86% ± 4%; N = 45. (C) Anterior segment of an uninjected Tg(fli1/EGFP) embryo at 31 hours after fertilization. (D) fbn1 MO–injected Tg(fli1/EGFP) embryo at 31 hours after fertilization showing dilated vessels around the eye and in the head (arrowheads). (E) Two splice-site morpholinos against the fbn1 gene generate similar vascular defects at high frequency. (F) RT-PCR analysis demonstrating depletion of the endogenous fbn1 transcript in fbn1 MO–injected embryos. (G) Summary of 2 independent injection experiments. Synergy between magp1 MO and fbn1 MO in generating embryos with dilated vessels (compare column 3 with columns 1 and 2). In these experiments, 1 ng magp1 MO and 2 ng fbn1 MO were used. N indicates number of embryos scored. Original magnifications: (A-B) 5 ×; (C-D) 10 ×. (E,G) Error bars indicate SEM.
Phenotypic analysis of fibrillin-1 (fbn1) morphant embryos. (A) Uninjected Tg(fli1/EGFP) embryo at 31 hours after fertilization. (B) fbn1 MO–injected Tg(fli1/EGFP) embryo at 31 hours after fertilization, showing dilated caudal vein with reduced venous plexus formation (arrowheads). Frequency, 86% ± 4%; N = 45. (C) Anterior segment of an uninjected Tg(fli1/EGFP) embryo at 31 hours after fertilization. (D) fbn1 MO–injected Tg(fli1/EGFP) embryo at 31 hours after fertilization showing dilated vessels around the eye and in the head (arrowheads). (E) Two splice-site morpholinos against the fbn1 gene generate similar vascular defects at high frequency. (F) RT-PCR analysis demonstrating depletion of the endogenous fbn1 transcript in fbn1 MO–injected embryos. (G) Summary of 2 independent injection experiments. Synergy between magp1 MO and fbn1 MO in generating embryos with dilated vessels (compare column 3 with columns 1 and 2). In these experiments, 1 ng magp1 MO and 2 ng fbn1 MO were used. N indicates number of embryos scored. Original magnifications: (A-B) 5 ×; (C-D) 10 ×. (E,G) Error bars indicate SEM.
Discussion
Microfibrils are a major ECM component of the cardiovascular system. Although the role of the endothelium in vascular development has been extensively characterized, the contribution of ECM components to vascular development and function is not as well understood. This study presents the characterization of the role for Magp1, a vascular ECM component, in zebrafish vascular development and function.
Role for Magp1 in vascular function and morphogenesis
magp1 morphant embryos exhibit vessel dilation and altered vessel wall architecture, as shown by histologic analysis. Perturbation of integrin function can be one underlying mechanism leading to the observed phenotypes. The integrins are heterodimeric receptors in the ECM, and they act as integrators of a variety of extracellular signals that control cell behavior during development.47-49 In this study, the function of integrins in the developing vasculature was inhibited by introduction of the disintegrin Echistatin (Sigma). A synergistic interaction between magp1 MO and Echistatin (Sigma) was observed. A previous in vitro study has shown that α5β1 and αvβ3 integrins are specific receptors for fibrillin ligands and the microfibrils, and they influence fibrillin-mediated cell adhesion and spreading.43 Our in vivo study implicates that the integrins participated in Magp1–mediated cell adhesion to the microfibrils during vascular development. It has been shown that the interaction of integrins with ECM molecules leads to the activation of a variety of signal transduction pathways,48 but the nature of the signaling pathway(s) deregulated from the inhibition of Magp1 remains to be determined.
Reduced caudal vein branching in magp1 morphant embryos suggests that Magp1 also plays a role in vascular patterning. The caudal vein plexus is a vascular network that undergoes a complex remodeling process, including branching morphogenesis.50,51 Fibrillin-2, a major microfibrillar protein, has been shown to be essential for branching morphogenesis of embryonic lung in a rat embryonic lung explant model.52 Regulation of epithelial and mesenchymal interactions is a proposed mechanism by which fibrillin-2 modulates lung organogenesis. MAGP1 might also regulate vascular morphogenesis by a similar mechanism, alone or in conjunction with fibrillin-1 or -2. Alternatively, the defective formation of the venous plexus may be secondary to the compromised vessel wall function in magp1 morphant embryos. Further studies will provide insight into the mechanism by which MAGP1 regulates venous plexus remodeling.
Overexpression and underexpression of magp1 result in overlapping vascular defects, including vessel dilation and reduced venous plexus remodeling. An in vitro binding study has shown that MAGP1, fibrillin-1, and decorin, a small chondroitin sulfate proteoglycan, can form a ternary complex,28 suggesting that MAGP1 may also participate in the formation of molecular complexes in the microfibril network. It is possible that overexpression of magp1 perturbs the stoichiometric balance of the components forming the complexes by titration. As a result, a loss-of-function effect is elicited instead. A similar phenomenon has been observed in a study investigating the overexpression effect of suppressor of hairless (Su[H]), an intracellular transducer of the Notch signaling pathway, in Drosophila.53 Overexpression of Su[H] results in mixed phenotypes, including loss of Notch activity, indicating that phenotypic manifestations from Su[H] overexpression are dose sensitive.
Role for fibrillin-1 and the microfibrils in vascular development
Dominant-negative mutations of the fibrillin-1 gene are associated with one form of Marfan syndrome.54,55 Clinical features of the Marfan syndrome, such as malfunction of the mitral valve and dilatation/dissection of the aorta, are often manifested in adulthood.10,55 In an accepted model of Marfan syndrome, mutant mice homozygous for a partial loss-of-function allele die of aortic dissection and rupture 2 weeks after they are born, recapitulating the vascular phenotype of the Marfan syndrome.56 In this study, the morpholino knockdown approach was used in zebrafish to determine the loss-of-function consequence for the fibrillin-1 gene in embryonic development. fbn1 MO–injected embryos exhibited abnormal vessel dilation and no venous plexus remodeling, suggesting that fibrillin-1 also plays an essential role in maintaining vessel wall function and regulating vascular morphogenesis during embryonic development of zebrafish. The vascular function of fibrillin-1 in embryonic development is likely conserved, as supported by conserved expression patterns of chick and human fibrillin-1 proteins.57,58 Fibrillin-1 morphant embryos and magp1 morphant embryos exhibit overlapping vascular defects. The in vivo interaction between Magp1 and fibrillin-1 during vascular development is demonstrated by the synergistic effect of partial loss-of-function of Magp1 and fibrillin-1. Given that MAGP1 and fibrillin-1 are components of the microfibrils, our analysis of magp1 and fbn1 morphants suggests the dynamic role played by microfibrils in vascular development.
Clinical applications for MAGP-1 in cardiovascular diseases
This study suggests an essential role for MAGP1 in vascular morphogenesis and function, results with a potential implication for a variety of clinical applications in cardiovascular disease. For example, MAGP1, along with FBN1, becomes a novel therapeutic target in the management of patients with Marfan syndrome and those with vascular diseases such as congenital bicuspid valves.59,60 Cardiovascular tissue engineering is being explored as a solution for tissue repair and creation in diseases such as atherosclerosis and congestive heart failure. Clinical applications include biosynthetic vascular grafts used in cardiac or peripheral bypass surgery.61,62 Artificial ECM proteins derived from elastin and fibronectin have been used in testing as candidates for small-diameter vascular grafts.63,64 MAGP1 is thus an attractive component of vascular graft engineering approaches.
Prepublished online as Blood First Edition Paper, February 9, 2006; DOI 10.1182/blood-2005-02-0789.
Supported by National Institutes of Health grant GM63904 (S.C.E.) and by the combined MD/PhD program at the University of Minnesota (E.C.).
The online version of this article contains a data supplement.
An Inside Blood analysis of this article appears at the front of this issue.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 U.S.C. section 1734.
We thank Dr Michael Pickart, Dr Lynda Ellis, and Eric Klee for bioinformatics support and the morpholino screen team of Aubrey Nielsen, Brent Bill, Eric Mendenhall, Craig Eckfeldt, and Dr Sridhar Sivasubbu. We also thank Dr Nicole Kirchhof and the Cancer Center Histopathology Core at the University of Minnesota for performing hematoxylin and eosin staining.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal