We investigated the clinical significance of a minor population of paroxysmal nocturnal hemoglobinuria (PNH)-type blood cells in patients with acquired aplastic anemia (AA). We quantified CD55-CD59- granulocytes and red blood cells (RBCs) in peripheral blood from 122 patients with recently diagnosed AA and correlated numbers of PNH-type cells and responses to immunosuppressive therapy (IST). Flow cytometry detected 0.005% to 23.1% of GPI-AP- cells in 68% of patients with AA. Sixty-eight of 83 (91%) patients with an increased proportion of PNH-type cells (PNH+) responded to antithymocyte globulin (ATG) + cyclosporin (CsA) therapy, whereas 18 of 39 (48%) without such an increase (PNH-) responded. Failure-free survival rates were significantly higher (64%) among patients with PNH+ than patients with PNH- (12%) at 5 years, although overall survival rates were comparable between the groups. Numbers of PNH-type and normal-type cells increased in parallel among most patients with PNH+ who responded to IST, suggesting that these cells are equally sensitive to immune attack. These results indicate that a minor population of PNH-type cells represents a reliable marker of a positive IST response and a favorable prognosis among patients with AA. Furthermore, immune attack against hematopoietic stem cells that allows PNH clonal expansion might occur only at the onset of AA.
Introduction
Immunosuppressive therapy (IST) with antithymocyte globulin (ATG) plus cyclosporin (CsA) is the standard approach to treating acquired aplastic anemia (AA).1-5 Approximately 70% of patients respond to this therapy and achieve remission. However, for the remaining 30%, IST might even be harmful because of an increased risk of opportunistic infections, particularly in the absence of any remission. The immune pathophysiology of patients should thus be understood at diagnosis, and IST should be applied only to those with immune-mediated AA. Several factors have been proposed as good markers that appear to reflect the immune pathophysiology of AA. These factors include an increased ratio of activated T cells,6 increased interferon-γ expression in bone marrow,7 and peripheral-blood T cells,8 as well as increased expression of heat-shock protein 70.9 Although these markers are useful in predicting responses to IST, few patients with AA have been tested, and the assays applied to detect these abnormalities are vulnerable to the effects of artifacts and the transportation of test samples. Consequently, none of the markers have been practically applied to predict responses to IST. Because of this, patients with AA are placed on IST without understanding the underlying pathophysiology.
One marker closely associated with immune pathophysiology in bone marrow failure is a small number of cells that are glycosylphosphatidylinositol-anchored membrane protein-deficient (GPI-AP-), namely paroxysmal nocturnal hemoglobinuria (PNH)-type cells.10-14 Dunn et al11 have demonstrated that an increase in CD15- CD66b-CD16+ granulocytes is associated with a good response to ATG among patients with myelodysplastic syndrome (MDS). Using 2-color flow cytometry that can distinguish proportions of CD55-CD59-CD11b+ granulocytes and CD55-CD59- glycophorin A+ red blood cells (RBCs) below 0.1%, we also demonstrated that a population of 0.01% to 6% PNH-type cells among granulocytes and red blood cells predicts a response to CsA in patients with MDS.15 Although one study group did not find a correlation between PNH-type cells and response to ATG in patients with AA,14 an increase in the proportion of PNH-type cells was correlated with a good response to IST among our patients with AA16 as well as those in another report.12 However, the significance of a minor population of PNH-type cells in the management of patients with AA has remained obscure because the number of patients with recently diagnosed AA has been small and follow-up periods have not been long enough. Our sensitive flow cytometric protocol has not become popular despite its potential clinical usefulness, perhaps because of the lower cut-off values (0.003% for granulocytes and 0.005% for RBCs) than previous assays.11,12,17,18
The outcome of IST in patients with AA is negatively affected by the length of time from diagnosis to treatment.19 To clarify the role of a marker that would predict a good response to IST, the marker should be tested on patients who have been recently diagnosed with AA and before they receive therapy, and then the marker should be correlated with the subsequent response to IST. Since 1999, we have been studying the presence of PNH-type cells in peripheral blood using flow cytometry in 241 patients who had not yet undergone therapy and who were diagnosed with AA. The present study focuses on 122 patients who were treated with ATG and CsA within 1 year of the diagnosis of AA and compares the response rates to IST and subsequent survival between patients with (PNH+) and without (PNH-) an increased proportion of PNH-type cells. We also examined changes in the number of PNH-type cells after successful IST to characterize the immune system attack against hematopoietic stem cells that confers a survival advantage on PNH-type stem cells in immune-mediated AA.
Patients, materials, and methods
Patients
We evaluated PNH-type cells in peripheral-blood samples from 122 Japanese patients (55 men and 67 women; median age, 56 years) with idiopathic AA (75 severe and 47 moderate AA) before they received IST. The patients were diagnosed with AA at Kanazawa University Hospital, hospitals participating in a cooperative study led by the Intractable Disease Study Group of Japan, and other referring institutions. The severity of AA was classified according to the criteria proposed by Camitta et al.20 All patients were treated with ATG Lymphoglobuline (Aventis Behring, King of Prussia, PA) 15 mg/kg/d, 5 days; plus CsA (Novartis, Basel, Switzerland) 6 mg/kg/d; within 1 year of diagnosis between April 1999 and December 2004. The dose of CsA was adjusted to maintain trough levels between 150 and 250 ng/mL, and the appropriate dose was administered for at least 6 months. Granulocyte colony-stimulating factor (G-CSF; filgrastim, 300 μg/m2 or lenograstim, 5 μg/kg) was administered to some patients. Response to IST was evaluated according to the response criteria described by Camitta.21 Complete response (CR) was defined as hemoglobin normal for age, neutrophil count more than 1.5 × 109/L, and platelet count more than 150 × 109/L. Partial response (PR) was defined as transfusion independent and no longer meeting criteria for severe disease in patients with severe AA, and it was defined as transfusion independence (if previously dependent) or doubling or normalization of at least one cell line or increase in baseline hemoglobin of more than 30 g/L (if initially less than 60 g/L), neutrophil count of more than 0.5 × 109/L (if initially less than 0.5 × 109/L), and platelet count of more than 10 × 109/L (if initially less than 20 × 109/L) in patients with moderate AA. The patients provided written, informed consent to participate in all procedures associated with the study, which was reviewed and approved by the ethical committee of Kanazawa University Hospital (study no. 46). The study also conforms to the recently revised tenets of the Helsinki protocol.
High-resolution 2-color flow cytometry
We improved the 2-color flow cytometry developed by Araten et al22 as follows. Briefly, 3 to 5 mL heparinized blood was drawn from each patient. To detect PNH-type granulocytes, RBCs were lysed in NH4Cl 8.26 g/L, KHCO3 1.0 g/L, and EDTA · E4Na 0.037 g/L (lysis buffer). After a saline wash, 50 μL leukocyte suspension was incubated with 4 μL phycoerythrin (PE)-labeled anti-CD11b monoclonal antibodies (mAbs; Becton Dickinson, Franklin Lakes, NJ), fluorescein-isothiocyanate (FITC)-labeled anti-CD55 mAbs (clone IA10, mouse IgG2a; Pharmingen, San Diego, CA), and FITC-labeled anti-CD59 mAbs (clone p282, mouse IgG2a; Pharmingen) on ice for 30 minutes.13 To detect PNH-type RBCs, PE-labeled anti-glycophorin A mAbs (clone JC159; DAKO, Glostrup, Denmark) were included instead of anti-CD11b mAbs.15 Fresh blood was diluted to 3% in phosphate-buffered saline (PBS), and then 50 μL was incubated with 4 μL PE-labeled anti-glycophorin A mAbs, FITC-labeled anti-CD55, and anti-CD59 mAbs on ice for 30 minutes. A total of at least 1 × 105 CD11b+ granulocytes and glycophorin A+ RBCs within each corresponding gate were analyzed using a FACScan (Becton Dickinson, Franklin Lakes, NJ) flow cytometry. To exclude damaged cells that often produce false-positive results, all samples were treated for flow cytometry within 24 hours after collection, and SSCdim and CD11bdim granulocytes and glycophorin Adim RBCs on the histograms were excluded from the analyses by careful gating as shown in Figure 1A. On the basis of analytic results from 68 healthy individuals, the presence of greater than 0.003% CD11b+ granulocytes and 0.005% glycophorin A+ RBCs was considered abnormal. Both thresholds greatly exceeded the mean + 4 SDs for GPI-AP- granulocytes (0.0025%) and RBCs (0.0032%) determined in healthy individuals.13,15 When PNH-type cells were increased in only 1 of the 2 cell lineages, another sample was collected, and the patient was deemed PNH+ only when the second sample produced similar results.
We compared the sensitivity of detecting a few PNH-type cells in this manner with that of a low-resolution method23 by analyzing the blood of some patients by 2-color flow cytometry using both PE-labeled anti-CD55 and FITC-labeled anti-CD59 mAbs. This assay defines the presence of 1% or more PNH-type cells as a significant increase.
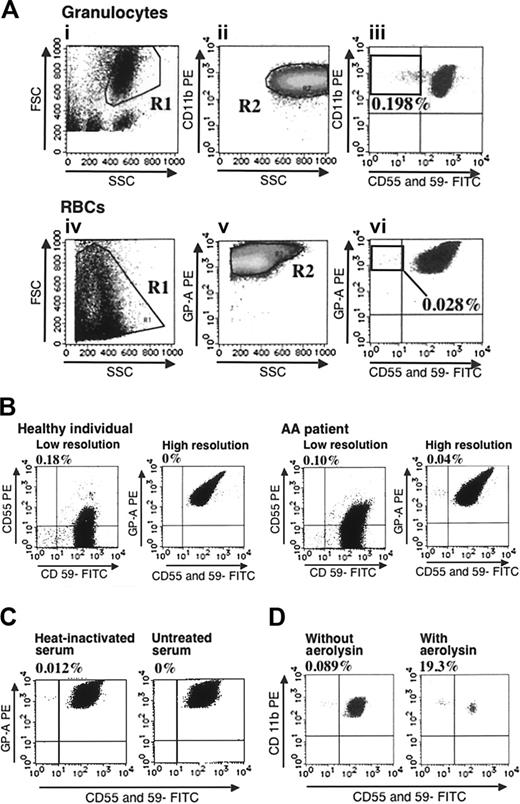
Validity of high-resolution flow cytometry. (A) An example of analysis on a patient with PNH+ AA is shown. Gates were set up to exclude SSCdim (i) and CD11bdim granulocytes and glycophorin Adim RBCs (ii,v). Cells within rectangles showing horizontal distribution represent PNH-type cells. (B) RBCs from a healthy individual and a patient with AA were examined using a low-resolution assay and the high-resolution assay. Numbers on histograms denote the percentages of CD55-CD59- cells in total RBCs for the low-resolution assay, and in glycophorin A+ RBCs for the high-resolution assay. (C) RBCs from a patient with PNH+ AA were incubated in acidified saline containing heat-inactivated or untreated serum. CD55-CD59- RBCs were then quantified. (D) PNH+ AA WBCs were incubated with or without 0.5 × 10-8 M aerolysin and analyzed by flow cytometry.
Validity of high-resolution flow cytometry. (A) An example of analysis on a patient with PNH+ AA is shown. Gates were set up to exclude SSCdim (i) and CD11bdim granulocytes and glycophorin Adim RBCs (ii,v). Cells within rectangles showing horizontal distribution represent PNH-type cells. (B) RBCs from a healthy individual and a patient with AA were examined using a low-resolution assay and the high-resolution assay. Numbers on histograms denote the percentages of CD55-CD59- cells in total RBCs for the low-resolution assay, and in glycophorin A+ RBCs for the high-resolution assay. (C) RBCs from a patient with PNH+ AA were incubated in acidified saline containing heat-inactivated or untreated serum. CD55-CD59- RBCs were then quantified. (D) PNH+ AA WBCs were incubated with or without 0.5 × 10-8 M aerolysin and analyzed by flow cytometry.
Modified Ham test
Peripheral blood of patients with AA with a low proportion (< 0.1%) of CD55-CD59- RBCs was washed with saline and suspended in saline at a hematocrit of 50%. The RBC suspension (15 μL) was incubated with 80 μL heat-inactivated fetal calf serum (FCS) for 10 minutes at 4°C for sensitization by anti-human heteroantibodies and then washed with saline. Human AB serum as a source of complement (0.5 mL) and 55 μL 0.2 N HCl were then added to the cell suspension. The negative control included heat-inactivated human AB serum instead of untreated human AB serum. These RBC suspensions were incubated for 60 minutes at 37°C and washed with PBS, and then the RBCs were analyzed by flow cytometry as described in “High resolution 2-color flow cytometry.”
Aerolysin treatment of granulocytes
Peripheral blood from patients with AA with a low proportion of PNH-type granulocytes was lysed as described in “High resolution 2-color flow cytometry,” and suspended in PBS at a density of 2 × 105 cells/mL. The leukocyte suspension was split into 2 portions; one was incubated for 15 minutes with and the other without 0.5 × 10-8 M aerolysin at 37°C.24 Before and after the incubation with aerolysin, the suspension was examined by flow cytometry to detect CD55-CD59-CD11b+ granulocytes as described in “High resolution 2-color flow cytometry.”
Statistics
The Mann-Whitney test compared clinical characteristics between patients with PNH+ and patients with PNH-. Fisher exact test and logistic regression modelling25 analyzed associations between individual pretreatment variables with response to IST. Kaplan-Meier methods graphically compared the cumulative incidence of the response with IST and time to event, and differences between patients with PNH+ and patients with PNH- were assessed by the log-rank test. A paired t test analyzed changes in the proportions of PNH-type cells associated with IST. All statistical analyses were performed using JMP version 5.0.1J software (SAS Institute, Cary, NC).
Results
Validity of high-resolution flow cytometry
Figure 1B shows that a low-resolution assay using PE-labeled anti-CD55 and FITC-labeled anti-CD59 mAbs detected greater than 0.1% PNH-type RBCs in the peripheral blood of a healthy individual, whereas our assay of the same sample detected 0% PNH-type cells. Thus, the low-resolution assay could not discriminate a patient with AA with 0.1% PNH-type cells from a healthy individual, whereas our method revealed 0.04% PNH-type RBCs in the same patient, indicating a diagnosis of PNH+ AA. When the sensitivity of RBCs to complement-mediated lysis was examined using the modified Ham test, almost all RBCs in the glycophorin A+CD55-CD59- fraction disappeared after an incubation in acidified saline containing human AB serum, verifying the reliability of our method for detecting PNH-type RBCs (Figure 1C). Conversely, when granulocytes from a patient with PNH+ AA were treated with aerolysin, approximately 99% of granulocytes in the CD11b+CD55+CD59+ fraction disappeared, whereas almost all cells in the CD11b+CD55-CD59- fraction remained unchanged (Figure 1D), indicating that the few granulocytes in the CD11b+CD55-CD59- fraction had the properties of PNH-type cells.
Proportions of PNH-type cells in patients with AA
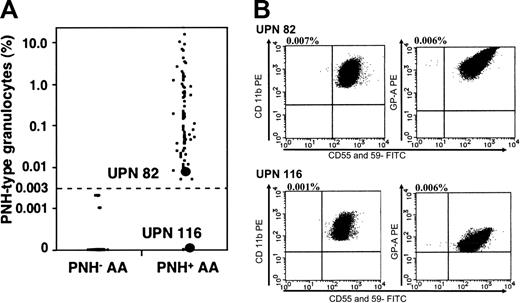
The proportion of PNH-type cells was increased in 83 (68%) patients. Among these patients with PNH+, the number of PNH-type cells was increased in both the granulocytes and RBCs of 69 (83%) of them, in only the granulocytes of 12 (15%), and in only the RBCs of 2 (2%). Figure 2A shows the proportions of PNH-type granulocytes and histograms from 2 patients with PNH+. Notably, the proportions of PNH-type granulocytes were below 0.1% in greater than 40% of patients with PNH+. Table 1 compares the clinical characteristics between patients with PNH+ and PNH-. Although the PNH+ group tended to be older and have higher WBC and MCV values than the PNH- group, the clinical and hematologic parameters did not significantly differ between them.
Clinical characteristics of PNH+ and PNH- patients
. | PNH+ . | PNH- . | P . |
|---|---|---|---|
| No. of patients | 83 | 39 | NA |
| Median age, y (range) | 57 (13-83) | 54 (12-83) | .16 |
| Sex, M/F | 36/47 | 19/20 | .58 |
| Severity, severe/moderate | 53/30 | 22/17 | .43 |
| Chromosome abnormality, no. of patients | 7 | 3 | .88 |
| –7 | 0 | 1 | |
| +8 | 2 | 1 | |
| –Y | 3 | 0 | |
| Others | 2 | 1 | |
| Median WBC count, × 109 (range) | 2.1 (0.5-4.3) | 1.9 (0.7-3.2) | .15 |
| Median neutrophil count, × 109/L (range) | 0.53 (0.02-2.2) | 0.49 (0.01-2.7) | .65 |
| Median hemoglobin level, g/L (range) | 67 (32-140) | 67 (40-108) | .92 |
| Mean corpuscular volume, fL (range) | 101.5 (84.2-123.5) | 98.5 (77.2-118.0) | .13 |
| Median platelet count, × 109/L (range) | 14.0 (2.0-60.0) | 16.0 (1.0-87.0) | .65 |
| Median reticulocyte count, × 109/L (range) | 19.0 (3.0-90.0) | 24.0 (2.0-106.0) | .50 |
| Median time from diagnosis to IST, d (range) | 30 (1-334) | 33 (2-268) | .46 |
| No. of patients who received G-CSF during IST | 25 | 12 | .94 |
. | PNH+ . | PNH- . | P . |
|---|---|---|---|
| No. of patients | 83 | 39 | NA |
| Median age, y (range) | 57 (13-83) | 54 (12-83) | .16 |
| Sex, M/F | 36/47 | 19/20 | .58 |
| Severity, severe/moderate | 53/30 | 22/17 | .43 |
| Chromosome abnormality, no. of patients | 7 | 3 | .88 |
| –7 | 0 | 1 | |
| +8 | 2 | 1 | |
| –Y | 3 | 0 | |
| Others | 2 | 1 | |
| Median WBC count, × 109 (range) | 2.1 (0.5-4.3) | 1.9 (0.7-3.2) | .15 |
| Median neutrophil count, × 109/L (range) | 0.53 (0.02-2.2) | 0.49 (0.01-2.7) | .65 |
| Median hemoglobin level, g/L (range) | 67 (32-140) | 67 (40-108) | .92 |
| Mean corpuscular volume, fL (range) | 101.5 (84.2-123.5) | 98.5 (77.2-118.0) | .13 |
| Median platelet count, × 109/L (range) | 14.0 (2.0-60.0) | 16.0 (1.0-87.0) | .65 |
| Median reticulocyte count, × 109/L (range) | 19.0 (3.0-90.0) | 24.0 (2.0-106.0) | .50 |
| Median time from diagnosis to IST, d (range) | 30 (1-334) | 33 (2-268) | .46 |
| No. of patients who received G-CSF during IST | 25 | 12 | .94 |
NA indicates not applicable.
Response to ATG and CsA therapy
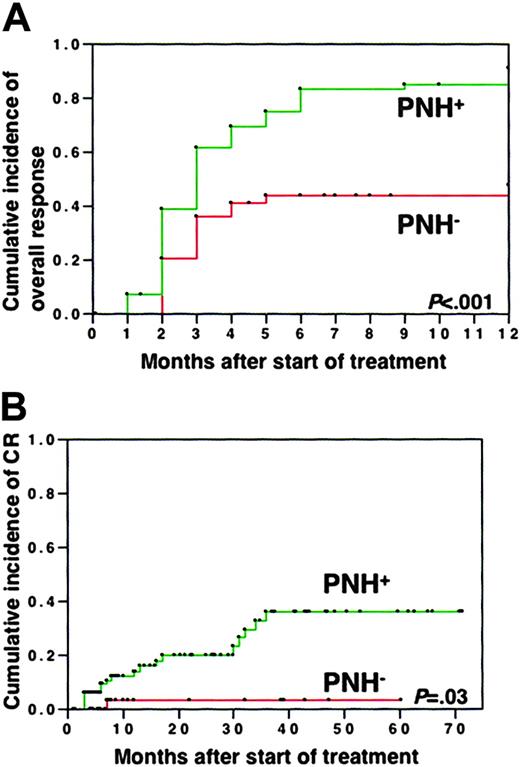
Sixty-eight of 83 (91%) patients with PNH+ improved with IST and achieved PR or CR at 12 months. However, only 18 of 39 (48%) patients with PNH- responded to IST. Kaplan-Meier analysis showed that the chance of achieving PR was significantly better among patients with PNH+ than among patients with PNH- (Figure 3A). The rate of obtaining CR at 5 years was also significantly higher in patients with PNH+ (36%) than in patients with PNH- (3%) (Figure 3B). Multivariate analysis showed that among sex (male or female), age (older or younger than 40 years), severity (severe or moderate), presence or absence of chromosomal abnormalities, and presence or absence of increased PNH-type cells, only the presence of increased PNH-type granulocytes was a significant factor associated with good response to IST (P < .001). When patients with PNH+ were classified into 5 subgroups according to the proportions of PNH-type granulocytes (0.003%-0.01% in 7, 0.01%-0.1% in 21, 0.1%-1.0% in 22, 1.0%-10.0% in 13, 10.0%-23.1% in 3), the response rates to IST at 6 months did not significantly differ (88%, 74%, 90%, 81%, and 100%, respectively) among these subgroups. The responses of all of these subpopulations were significantly better than that of patients with PNH-.
Prognosis after IST
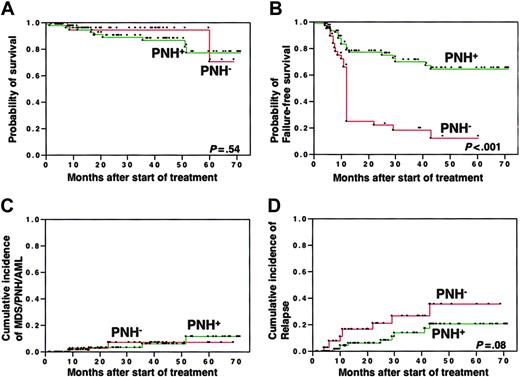
The median follow-up period was 26.4 months (range, 0.1 to 71.4 months). In contrast to the response rates, the rates of overall survival at 5 years were comparable between patients with PNH+ (77%) and with PNH- (71%) (Figure 4A). However, the probability of surviving failure free at 5 years was significantly higher in patients with PNH+ (64%) than in patients with PNH- (12%) when failure-free survival was calculated based on time to treatment failure. This was defined as whichever came first among time from the first day of treatment until salvage treatment for nonresponse, relapse, development of a clonal hematologic disease (PNH, MDS, leukemia), solid tumor, or disease- or treatment-related death (Figure 4B). Although the probability of evolution into florid PNH or MDS at 5 years after IST did not significantly differ between patients with PNH+ (6% and 3%) and patients with PNH- (0% and 4%) (Figure 4C), the probability of relapse tended to be higher in patients with PNH- (36%) than in patients with PNH+ (21%) (Figure 4D). Two (2%) patients with PNH+ and 7 (18%) with PNH- underwent allogeneic bone marrow transplantation (BMT) from related (n = 6) or unrelated (n = 3) donors because of failure to respond to IST (n = 6) and relapse of AA (n = 3). Rates of survival after BMT did not significantly differ between the 2 groups (data not shown).
Proportions of PNH-type granulocytes. (A) Proportions of CD55-CD59- granulocytes in each patient. (B) Histograms from one patient with PNH+ (UPN 82) with minimal PNH-type cells and from another patient with increased PNH-type cells only in RBCs (UPN 116).
Proportions of PNH-type granulocytes. (A) Proportions of CD55-CD59- granulocytes in each patient. (B) Histograms from one patient with PNH+ (UPN 82) with minimal PNH-type cells and from another patient with increased PNH-type cells only in RBCs (UPN 116).
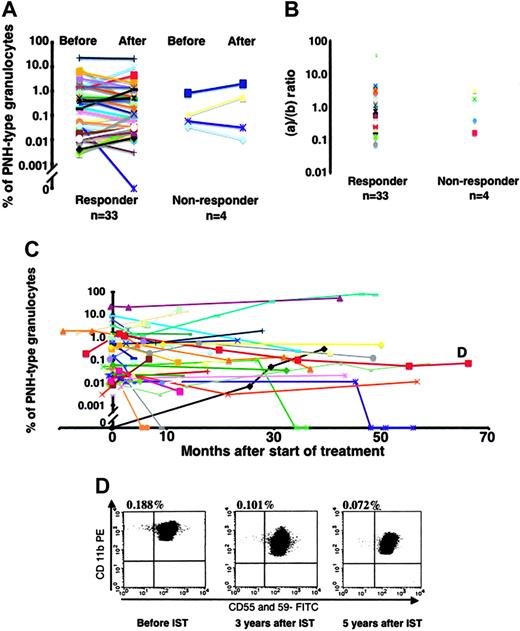
Changes in PNH-type granulocytes after IST
The presence of PNH-type cells after IST was serially tested in the peripheral blood of 53 of 122 patients. To characterize immune attack against hematopoietic stem cells that favors PNH-type cell clonal expansion, we examined the numbers of PNH-type cells in responsive patients. Figure 5A shows that the proportions of PNH-type granulocytes remained almost constant in 32 of 33 patients with PNH+ who responded to IST and decreased from 0.045% to 0% in only 1 patient (UPN 25). This indicates that the absolute number of PNH-type as well as of normal-type granulocytes increased in most responsive patients after IST. We compared the ratio of the degree of the increase in the absolute count between PNH-type (a) and normal-type (b) granulocytes before IST. The PNH-type granulocyte-to-normal-type granulocyte ratio in 32 patients ranged from 0.07 to 38.1 with a median of 1.06 (Figure 5B). The proportions of PNH-type cells did not change in 4 patients with PNH+ who were refractory to IST (Figure 5A-B). Sixteen patients with PNH- were also tested after 6 to 24 months of IST. Only one patient who had achieved PR became PNH+ at 24 months and then relapsed with AA at 29 months after IST.
The proportions of PNH-type granulocytes were repeatedly determined in 23 patients for more than 24 months after IST. Figure 5C shows that the proportions remained constant over a long period in most patients including one (UPN 106) who had 0.1% PNH-type granulocytes (Figure 5D). The proportion of PNH-type granulocytes significantly increased from 3.31% to 76.0% in only one patient during the 4-year observation period.
Discussion
An increase in the proportion of PNH-type cells in peripheral blood has been implicated in the immune pathophysiology of bone marrow failure.10 Several studies including our previous investigation found a correlation between an increase in the proportion of PNH-type cells and a favorable response to IST among patients with MDS11,12,15 and with AA.16,26 However, the clinical application of these findings has been hampered. Small patient cohorts and the relatively low prevalence of an increased number of PNH-type cells in these studies have led to concerns about unreliability of the correlation. The present study based on a larger number of patients with recently diagnosed AA conclusively demonstrated that a minor population of PNH-type cells predicts a good response to IST as well as good prognosis for patients with AA after IST. The reliability of our high-resolution flow cytometry, which was verified by the modified Ham test and by aerolysin treatment, revealed an increase in the number of PNH-type cells in 68% of the patients with AA. This was considerably higher than the reported prevalence.
Response to immunosuppressive therapy. Incidence of overall (A) and complete (B) responses in patients with PNH+ and PNH-.
Response to immunosuppressive therapy. Incidence of overall (A) and complete (B) responses in patients with PNH+ and PNH-.
Prognosis after IST compared between patients with PNH+ and with PNH-. (A) Overall survival; (B) failure-free survival; (C) incidence of clonal hematologic disorders, including PNH, myelodysplastic syndrome, and acute myelogenous leukemia; and (D) incidence of relapse.
Prognosis after IST compared between patients with PNH+ and with PNH-. (A) Overall survival; (B) failure-free survival; (C) incidence of clonal hematologic disorders, including PNH, myelodysplastic syndrome, and acute myelogenous leukemia; and (D) incidence of relapse.
The clinical features and overall survival rates did not significantly differ between patients with PNH+ and patients with PNH- in the present study. However, failure-free survival was obviously better among patients with PNH+ than patients with PNH-. This indicated that, although patients with PNH- can survive as long as patients with PNH+ after IST, they often require salvage or supportive treatment such as allogeneic stem cell transplantation and blood transfusions, because of a partial response to IST or a high rate of relapse. Contrary to the expectation based on the presence of abnormal hematopoietic clones such as PNH-type cells, the probability of evolving into clinical PNH or MDS in patients with PNH+ was comparable to that in patients with PNH-. The proportions of PNH-type granulocytes remained stable over a period of 1 to 66 months in most patients with PNH+, a finding consistent with previous reports.26,27 These findings indicate that the presence of an increased proportion of PNH-type cells predicts not only a positive response but also a good quality of response to IST among patients with AA.
Changes in proportions of PNH-type granulocytes associated with responses to IST. (A) Change in responders and nonresponders. (B) Proportions of granulocyte counts after and before IST determined for PNH-type (a) and normal-type (b) granulocytes and ratios of PNH-type granulocytes (a) to normal-type granulocytes (b) were plotted. (C) Longitudinal analysis of PNH-type granulocytes. Proportions of PNH-type granulocytes of 37 patients with PNH+ and 1 patient with PNH- who became PNH+ (black line) were displayed. (D) Changes in proportions of PNH-type granulocytes over 5 years in patient UPN 106 with AA (shown as D in Figure 5C).
Changes in proportions of PNH-type granulocytes associated with responses to IST. (A) Change in responders and nonresponders. (B) Proportions of granulocyte counts after and before IST determined for PNH-type (a) and normal-type (b) granulocytes and ratios of PNH-type granulocytes (a) to normal-type granulocytes (b) were plotted. (C) Longitudinal analysis of PNH-type granulocytes. Proportions of PNH-type granulocytes of 37 patients with PNH+ and 1 patient with PNH- who became PNH+ (black line) were displayed. (D) Changes in proportions of PNH-type granulocytes over 5 years in patient UPN 106 with AA (shown as D in Figure 5C).
The significantly high response rate to IST among patients with PNH+ AA suggests that PNH+ AA is an authentic type of immune-mediated marrow failure. In line with this hypothesis, patients with PNH+ AA often have a specific HLA-DR allele (HLA-DR15) and antigen-driven T-cell proliferation in the bone marrow.12,28 Furthermore, antibodies against diazepam-binding inhibitor-related sequence-1 (DRS-1), a peroxisomal protein abundantly expressed by hematopoietic progenitor cells, are frequently detected in sera from patients with PNH+ AA.29 However, the relatively low response rate to IST among patients with PNH- AA indicates that a heterogeneous pathophysiology might underlie this subset of AA. In line with this notion as described in our previous study,16 clonal hematopoiesis arose more frequently in patients with PNH- AA than in patients with PNH+ AA. Even among patients who responded to IST, patients with PNH- AA rarely achieved complete recovery of hematopoiesis and were susceptible to AA relapse. Immune mechanisms that are not associated with an increase in the proportion of PNH-type cells might damage hematopoietic stem cells more profoundly than those in PNH+ AA.
PNH-type stem cells might acquire a survival advantage over normal-type stem cells when T or natural killer (NK) cells attack hematopoietic stem cells.30-32 The high response rate to IST in patients with PNH+ AA indicates that such an immune mechanism is functional in this subset of AA. If the immune mechanisms were responsible for bone marrow failure, IST would more efficiently induce expansion of normal-type than of PNH-type stem cells. However, in most patients with PNH+, successful IST resulted in a similar increase in the number of both PNH-type and normal-type granulocytes, which contradicts the immune escape theory. A similar finding has been reported by Maciejewski et al26 for patients with AA with 1% or more CD15+CD66b-CD16- granulocytes. One possible explanation for this discrepancy is as follows. An immune attack against hematopoietic stem cells at the onset of AA that allows PNH-type stem cells to survive does not contribute to the subsequent progression of bone marrow failure, which is caused by different immune mechanisms targeting epitopes other than those that induce disease. Such epitope spreading occurs in the development of other immune diseases such as multiple sclerosis.33 Alternatively, the suppression of hematopoiesis after the clonal expansion of PNH-type cells might be caused by myelosuppressive cytokines rather than antigen-specific T cells.
The presence of a few PNH-type cells has profound significance for the management of patients with recently diagnosed AA. Although those who have PNH- AA can improve with IST, the maximal response rate is 50% and the rate of failure-free survival at 5 years is below 20%. Therefore, allogeneic BMT is recommended more often than IST for young patients with PNH- who have HLA-compatible sibling donors. Conversely, IST is more frequently recommended than BMT for patients with PNH+, particularly when the likelihood of BMT-related mortality is high. Among patients with AA who are unresponsive to the initial ATG and CsA therapy, those who benefit from a second IST might be PNH+. Conventional flow cytometry capable of detecting 1% or more PNH-type cells would also be clinically useful in predicting response to IST because the response to IST does not change according to the proportion of PNH-type cells. The predictive value of an increased proportion of PNH-type cells for a favorable prognosis in AA identified here warrants a further worldwide prospective study on non-Japanese patients with AA.
Prepublished online as Blood First Edition Paper, September 22, 2005; DOI 10.1182/blood-2005-06-2485.
Supported in part by a Grant-in-Aid for Scientific Research from the Ministry of Education, Science, Technology, Sports, and Culture of Japan (KAKENHI 15390298) and grants from the Research Committee for the Idiopathic Hematopoietic Disorders, Ministry of Health, Labor, and Welfare, Japan.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 U.S.C. section 1734.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal