Abstract
Keratinocyte growth factor (KGF) is a member of the fibroblast growth factor family that mediates epithelial cell proliferation and differentiation in a variety of tissues, including the thymus. We studied the role of KGF in T-cell development with KGF-/- mice and demonstrated that thymic cellularity and the distribution of thymocyte subsets among KGF-/-, wildtype (WT), and KGF+/- mice were similar. However, KGF-/- mice are more vulnerable to sublethal irradiation (450 cGy), and a significant decrease was found in thymic cellularity after irradiation. Defective thymopoiesis and peripheral T-cell reconstitution were found in KGF-/- recipients of syngeneic or allogeneic bone marrow transplant, but using KGF-/- mice as a donor did not affect T-cell development after transplantation. Despite causing an early developmental block in the thymus, administration of KGF to young and old mice enhanced thymopoiesis. Exogenous KGF also accelerated thymic recovery after irradiation, cyclophosphamide, and dexamethasone treatment. Finally, we found that administering KGF before bone marrow transplantation (BMT) resulted in enhanced thymopoiesis and peripheral T-cell numbers in middle-aged recipients of an allogeneic BM transplant. We conclude that KGF plays a critical role in postnatal thymic regeneration and may be useful in treating immune deficiency conditions. (Blood. 2006;107:2453-2460)
Introduction
Keratinocyte growth factor (KGF) is a 28-kDa fibroblast growth factor family member (FGF-7) that mediates epithelial cell proliferation and differentiation in a variety of tissues, including the gut (gut epithelial cells), skin (keratinocytes), and thymus (thymic epithelial cells).1-3 KGF is produced by mesenchymal cells and has a paracrine effect on epithelial cells4,5 ; it binds FGFR2IIIb, a splice variant of FGF receptor 2, expressed predominantly on these cell types. FGFR2IIIb is activated by 4 known ligands: FGF-1, FGF3, FGF-7, and FGF-10.6,7
The heterogeneous stromal cell compartment of the thymus includes both cortical and medullary epithelial cells, as well as mesenchymal cells (including fibroblasts). Mesenchymal cells produce fibroblast growth factors and support thymocyte development, especially in cortical areas (reviewed in Anderson and Jenkinson8 ). Jenkinson et al9 reported that mesenchymal cells regulate the proliferation of thymic epithelial cells via the production of KGF (FGF-7) and fibroblast growth factor-10 (FGF-10) during fetal development, but the role of mesenchymal cells in regulating the composition of thymic stroma in the neonatal and postnatal period is unclear.
Erikson et al10 have demonstrated that KGF and FGFR2IIIb signaling can affect the development and function of thymic epithelium (TE). In the adult thymus, mature αβ+ thymocytes are capable of producing KGF, which leads to the expansion of thymic medullary epithelial cells.10 However, KGF expression is not detectable in the triple negative (CD3-CD4-CD8-) thymocyte precursors.10
In contrast, peripheral αβ- T cells do not secrete KGF, even in epithelial tissues that comprise the skin, intestine, and vagina. However, γδ- T cells in epithelial tissues do produce KGF and may also regulate epithelial cell growth.11
KGF can function as a growth factor for epithelial protection and repair, is found in a variety of tissues (extensively reviewed by Finch and Rubin12 ), and is up-regulated after various forms of injury and tissue damage, including cutaneous injury,13 surgical bladder injury,14 chemically induced kidney injury,15 and a bleomycin-induced acute lung injury model.16 These protective effects on epithelial cells in mucosal tissues could have therapeutic potential when developing clinical strategies aimed at decreasing mucosal damage after chemotherapy or radiation therapy. KGF administration has recently been approved by the FDA for the prevention of oral mucositis, after intensive therapy in patients with hematologic cancers.17 Murine models using pretransplantation administration of KGF to recipients of allogeneic bone marrow transplants demonstrated a decrease in mortality from graft-versus-host disease (GVHD) and less GVHD-associated pathology in various tissues.18-21 In one murine model, KGF administration also improved leukemia-free survival by decreasing the mortality from GVHD while preserving donor T-cell graft-versus-leukemia (GVL) activity.21
In this study, we used KGF-/- mice and KGF administration to analyze the role of KGF in postnatal thymic development and recovery.
Materials and methods
Mice and BMT
Female C57BL/6J (B6, H-2b), C3FeB6F1 ([B6xC3H] F1, H-2b/k), B6D2F1/J (H-2b/d), B10.BR (H-2k), CBA/J (H-2k), BALB/c (H-2d), B6.129 (H-2b), and C57BL/6J (Ly5.1+) mice were obtained from The Jackson Laboratory (Bar Harbor, ME). We obtained KGF-/- mice with a B6.129 background from The Jackson Laboratory, and KGF+/- mice were generated in our animal care facility by crossing KGF-/- mice with B6.129 WT mice. Mice used in BMT experiments were 8 to 10 weeks old or 10 to 18 months old (CBA/J). BMT protocols were approved by the Memorial Sloan-Kettering Cancer Center Institutional Animal Care and Use Committee. Bone marrow (BM) cells were aseptically removed from femurs and tibias. Donor BM was T-cell-depleted (TCD) by incubation with anti-Thy-1.2 for 30 minutes at 4°C followed by incubation with Low-TOX-M rabbit complement (Cedarlane Laboratories, Hornby, ON, Canada) for 40 minutes at 37°C. Cells (5-10 × 106 BM cells) were resuspended in Dulbecco Modified Eagle Medium (DMEM; Life Technologies, Grand Island, NY) and transplanted by tail vein injection (0.25 mL total volume) into lethally irradiated recipients on day 0. On day 0 prior to transplantation, recipients received (900-1300 cGy) total body irradiation (137Cs source) as a split dose with a 3-hour interval between doses (to reduce gastrointestinal toxicity). Mice were housed in sterilized microisolator cages and received normal chow and autoclaved hyperchlorinated drinking water (pH 3.0).
Reagents and antibodies
Anti-murine CD16/CD32 FcR block (2.4G2) and all of the following fluorochrome-labeled antibodies against murine antigens were obtained from BD PharMingen (San Diego, CA): Ly-9.1 (30C7), H2D (34-2-12), CD3 (145-2C11), CD4 (RM4-5), CD8 (53-6.7), CD62L (MEL-14), CD122 (TM-B1), CD44 (IM7), CD45R/B220 (RA3-6B2), NK1.1 (PK136), CD11b (M1/70), CD11c (HL3), TER 119 (TER119), CD25 (PC61), CD69 (H1.2F3), TCRαβ (H57-597), GR-1 (RB6-8C5), c-kit (2B8), SCA-1 (D7), EpCAM (G8.8), PECAM (MEC13.3). Isotype controls were rat IgG2a-κ (R35-95), rat IgG2a-λ (B39-4), rat IgG2b-κ (A95-1), rat IgG1-κ (R3-34), hamster IgG-group1-κ (A19-3), hamster IgG-group 1-λ (Ha4/8), streptavidin-FITC, -PE, and -TR. Recombinant human KGF was kindly provided by Amgen (Thousand Oaks, CA). MTS-15 was provided by R.L.B.
Tissue culture medium consisted of RPMI-1640 or DMEM supplemented with 10% heat-inactivated fetal calf serum, 100 U/mL penicillin, 100 μg/mL streptomycin, and 2 mM l-glutamine.
Drug and cytokine administration
Recombinant human KGF was administered for 3 days at 5 mg/kg/d subcutaneously. Dexamethasone was administered at a dose of 5 mg/kg/mouse via a single intraperitoneal injection. In some experiments, mice were also treated with cyclophosphamide (300 mg/kg) 3 days after KGF administration.
Cells
Single-cell suspensions were prepared from spleen, thymus, BM, and lymph nodes (LNs) according to standard protocols and analyzed by fluorescence-activated cell sorting (FACS).
Flow cytometric analysis
Cells were washed in FACS buffer (PBS/0.5% BSA/0.1% sodium Azide), and 106 cells/mL were incubated for 20 minutes at 4°C with CD16/CD32 FcR block. Subsequently, cells were incubated for 30 minutes at 4°C with antibodies and washed twice with FACS buffer. The stained cells were resuspended in FACS buffer and analyzed on a FACSCalibur and LSR-I flow cytometer (Becton Dickinson, San Jose, CA) with CellQuest software or with Flowjo software (Treestar, San Carlos, CA).
Thymic stromal cell preparation
We used a previously published method in preparing the thymic stromal cells.22 Briefly, thymi were cut into 1- to 2-mm pieces. Then they were incubated with a solution containing DNAse-I (0.1% wt/vol) and collagenase D (0.125% wt/vol)(both from Roche, Indianapolis, IN). CD45- stromal cells were obtained after incubating and washing 4 to 5 times. The different stromal cells were defined according to their surface expression of anti-EpCAM, anti-MTS-15, anti-PECAM, and anti-MHC class II, respectively. Thymic epithelial cells are CD45- MHC Class II+ EpCAM+; fibroblasts are CD45-, MTS-15+, PECAM-, epCAM-; and, endothelial cells are CD45-, MTS15-, PECAM+, epCAM-.
Proliferation assay
Splenocytes (4 × 105 cells/well from KGF-treated recipients) were incubated for 3 days with Concanavalin A (2.5 μg/mL; Sigma-Aldrich, St Louis, MO) or for 5 days with irradiated (2000 cGy) host splenocytes as stimulators (2 × 105 cells/well) in 96-well plates. Cultures were pulsed during the final 18 hours with 1 μCi (0.037 MBq) of 3H-thymidine/well, and cells were harvested with a Topcount Harvester (Packard, Meriden, CT). The proliferation of the cells was determined as counts per minute (cpm).
Statistics
All values are expressed as mean ± SEM. Statistical comparison of experimental data were performed with the nonparametric unpaired Mann-Whitney U test. A P value less than .05 was considered statistically significant.
Results
KGF-/- mice have intact thymopoiesis
KGF-/- mice were generated by Guo et al23 to determine the effects of KGF deficiency on wound healing. Interestingly, the absence of KGF did not result in any abnormalities in epidermal growth or wound healing, and all tissues with important mesenchymal-epithelial interactions were normal. However, thymopoiesis and T-cell development were not studied in detail, because they were beyond the scope of this report.
We observed normal thymopoiesis and normal numbers of thymic subsets, including double positives (DPs), single positives (SPs), and double negatives (DNs) in KGF-/- mice (Figure 1A-D). The distribution and numbers of thymic stromal cells were similar in KGF-/- mice compared with WT and KGF+/- controls (Figure 1E). We also evaluated the thymic stromal cell composition in KGF-/-, KGF+/-, and WT mice by immunohistochemistry. The expression of the keratin subunits (K5 and K8), ER-TR-7, pan-keratin, Aire, and MTS-15 were found to be similar among these mice (data not shown). In determining whether KGF deficiency affects thymic involution during the aging process, we found that 16-month-old KGF-/- and KGF+/- mice showed similar thymic cellularity and no difference in thymocyte distribution. These data suggest that thymic involution is not accelerated in KGF-/- mice (Figure S1 on the Blood website; see the Supplemental Figure link at the top of the online article).
However, splenic cellularity was decreased in KGF-/- mice in comparison with WT and heterozygote controls (Figure 1A). Further analysis of the spleen revealed that KGF-/- mice have a similar distribution of T cells (CD3+), B cells (B220+), monocytes (CD11B+Gr-1-), granulocytes and their precursors (CD11b+GR-++), and dendritic cells (CD11c+), but absolute numbers were reduced in KGF-/- mice (Figure 1F; data not shown). We also analyzed peripheral LNs (axillary and inguinal) and observed no difference in cellularity or T-cell distribution (Figure 1G; data not shown). To determine whether KGF deficiency affected the numbers of hematopoietic precursors in the BM, we harvested BM from KGF-/- and KGF+/- mice to determine hematopoietic precursor (lineage- c-kit+ Sca-1+) distribution and number and found no differences between KGF-/- and KGF+/- mice (Figure 1H-I).
These data suggest that KGF deficiency does not affect thymopoiesis and T-cell development but does decrease splenic size and cellularity.
KGF-/-mice have intact T-cell development in the thymus. (A) Nine- to 12-week-old KGF-/-, KGF+/-, and WT controls (B6.129) were killed, and thymic, splenic, and BM cellularities were determined. (B-D) Ten- to 12-week-old KGF-/- and KGF+/- mice were killed and stained with anti-CD4, -CD8, -CD3, -CD44, and -CD25 antibodies, and thymic subpopulations were determined (DP, CD4+CD8+; DN, CD4-CD8-; DN1, CD44+CD25-CD4-CD8-CD3-; DN2, CD44+CD25+CD4-CD8-CD3-; DN3, CD44-CD25+CD4-CD8-CD3-; DN4, CD44-CD25-CD4-CD8-CD3-). (E) Thymic stromal cell populations were determined in KGF-/- and KGF+/- mice (according to the protocol described in “Materials and methods”). (F) Splenocytes from KGF-/- and KGF+/- mice were stained with CD4- and CD8- antibodies, and total CD4+ and CD8+ T-cell numbers were determined by flow cytometric analyses and splenic counts. (G) Peripheral lymph node (axillary and inguinal) cellularities of KGF-/-, KGF+/-, and WT mice were determined. (H-I) Bone marrow cells from KGF-/- and KGF+/- mice were stained with lineage-specific antibodies (anti-CD3, -CD11b, -Gr-1, -B220, and -NK1.1), anti-C-kit, and anti-SCA-1 antibodies. Stem cell populations were determined by multicolor flow cytometric analysis of lineage-negative cells, and the BM cell count from one mouse leg. Values represent mean (± SEM) and n = 6 to 12 mice per group. *P < .05.
KGF-/-mice have intact T-cell development in the thymus. (A) Nine- to 12-week-old KGF-/-, KGF+/-, and WT controls (B6.129) were killed, and thymic, splenic, and BM cellularities were determined. (B-D) Ten- to 12-week-old KGF-/- and KGF+/- mice were killed and stained with anti-CD4, -CD8, -CD3, -CD44, and -CD25 antibodies, and thymic subpopulations were determined (DP, CD4+CD8+; DN, CD4-CD8-; DN1, CD44+CD25-CD4-CD8-CD3-; DN2, CD44+CD25+CD4-CD8-CD3-; DN3, CD44-CD25+CD4-CD8-CD3-; DN4, CD44-CD25-CD4-CD8-CD3-). (E) Thymic stromal cell populations were determined in KGF-/- and KGF+/- mice (according to the protocol described in “Materials and methods”). (F) Splenocytes from KGF-/- and KGF+/- mice were stained with CD4- and CD8- antibodies, and total CD4+ and CD8+ T-cell numbers were determined by flow cytometric analyses and splenic counts. (G) Peripheral lymph node (axillary and inguinal) cellularities of KGF-/-, KGF+/-, and WT mice were determined. (H-I) Bone marrow cells from KGF-/- and KGF+/- mice were stained with lineage-specific antibodies (anti-CD3, -CD11b, -Gr-1, -B220, and -NK1.1), anti-C-kit, and anti-SCA-1 antibodies. Stem cell populations were determined by multicolor flow cytometric analysis of lineage-negative cells, and the BM cell count from one mouse leg. Values represent mean (± SEM) and n = 6 to 12 mice per group. *P < .05.
KGF is required for thymic reconstitution after sublethal irradiation. KGF-/-, KGF+/-, and WT mice were sublethally irradiated (450 cGy), and mice were harvested at day 21 and at day 28 after irradiation. (A-B) Thymic cellularity was determined, and DP, DN, CD4+, and CD8+ thymocyte numbers were calculated by flow cytometric analysis and thymic counts. (C) Splenic cellularity was determined, and splenic CD3+CD4+, and CD3+CD8+ T-cell numbers were calculated by flow cytometric analysis and splenic counts. (D-F) Peripheral blood was obtained from these mice at different time points, and white blood cell (WBC) counts, platelet counts, and hemoglobin levels were determined by an automated cell counter. Values represent mean (± SEM) and n = 3-10 per group. *P < .05.
KGF is required for thymic reconstitution after sublethal irradiation. KGF-/-, KGF+/-, and WT mice were sublethally irradiated (450 cGy), and mice were harvested at day 21 and at day 28 after irradiation. (A-B) Thymic cellularity was determined, and DP, DN, CD4+, and CD8+ thymocyte numbers were calculated by flow cytometric analysis and thymic counts. (C) Splenic cellularity was determined, and splenic CD3+CD4+, and CD3+CD8+ T-cell numbers were calculated by flow cytometric analysis and splenic counts. (D-F) Peripheral blood was obtained from these mice at different time points, and white blood cell (WBC) counts, platelet counts, and hemoglobin levels were determined by an automated cell counter. Values represent mean (± SEM) and n = 3-10 per group. *P < .05.
Thymic recovery is defective in KGF-/- mice after sublethal irradiation
We evaluated the contribution of KGF to thymic and peripheral T-cell recovery after insults such as irradiation. KGF-/-, KGF+/-, and WT mice were sublethally irradiated (450 cGy) and followed for 4 weeks. We observed that thymic cellularity and thymic subpopulations in KGF-/- mice were significantly lower than in the control groups (KGF+/- mice and WT mice) when analyzed at days 21 and 28 after irradiation (Figure 2A-B). However, the relative distribution of thymocytes was not different in the recovering thymus of KGF-/- mice versus KGF+/- mice after sublethal irradiation. Splenic cellularity and splenic T, B, and natural killer (NK) cell numbers were found to be similar in KGF-/- and KGF+/-) mice (Figure 2C; data not shown). In addition, splenic T-cell numbers were higher in WT mice than in KGF-/- mice when analyzed at day 28 after sublethal irradiation. We also evaluated peripheral LN recovery on day 28 after sublethal irradiation and found that WT and KGF-/- mice had similar lymph node cellularity. Interestingly, LN cellularity was slightly higher in KGF+/- controls than in WT and KGF-/- mice (data not shown). Additionally, peripheral hematopoietic recovery (white blood cell [WBC] count, hemoglobin [HGB] level, and platelet count) was comparable in KGF-/-, KGF+/- mice, and WT mice when analyzed at day 28 (Figure 2D-F). We did not find any differences in the relative recovery of neutrophils and lymphocytes between KGF-/- and KGF+/- mice (data not shown). This suggests that KGF is required for thymic regeneration after irradiation but has no role in the recovery of other hematopoietic lineages.
Stromal cell-derived KGF production plays a critical role in thymic recovery after irradiation
We used KGF-/- and KGF+/- mice as donors and hosts in murine syngeneic and allogeneic BMT models to test which cells were responsible for the production of KGF required for thymic recovery after irradiation. We first analyzed the role of host KGF production by using KGF-/- mice as recipients of syngeneic BM (B6.129 [TCD-BM]→B6.129 KGF-/- or KGF+/-) or allogeneic BM (BALB/c(TCD-BM)→B6.129 KGF-/- or KGF+/-) and noted significant defects in the posttransplantation recovery of thymocytes and peripheral T cells in both cases (Figure 3A-F). In the allogeneic model (BALB/c→B6.129 KGF-/-), we were able to separate donor from host-derived T cells in the spleen by using the H-2d marker, which is expressed only on donor-derived cells (BALB/c). Interestingly, we observed that counts of both donor- and host-derived T cells in KGF-/- recipients were significantly lower than in the control group (KGF+/-). Next, we examined the role of donor-derived KGF by using KGF-/- and KGF+/- mice as donors. We found a slight, statistically insignificant (P = .08) decrease in thymic cellularity in recipients of KGF-/- BM in the allogeneic model. Thymocyte subsets (except for CD4+SP) and peripheral T cells were found to be similar in the recipients of both donor strains (Figure 3G-L). We conclude that stromal cell-derived KGF is required for optimal thymic and peripheral T-cell recovery after irradiation in syngeneic and allogeneic BMT models.
Host-derived KGF is required for thymic reconstitution after syngeneic and allogeneic BMT. Lethally irradiated (1100 cGy) KGF-/- or KGF+/- mice were transplanted with B6.129-WT bone marrow (A-C) or BALB/c BM (D-F). Lethally irradiated (1100 cGy) B6.129 (G-H) or (900 cGy) BALB/c mice (J-L) were transplanted with KGF-/- or KGF+/- BM and were killed at day 28. Thymic and splenic cellularity was determined, and splenic CD3+, CD4+, and CD8+ T-cell numbers were calculated by flow cytometric analysis. Anti-H2d antibody was used to determine the chimerism in allogeneic experiments. Values represent mean (± SEM) and n = 5-11 per group. *P < .05.
Host-derived KGF is required for thymic reconstitution after syngeneic and allogeneic BMT. Lethally irradiated (1100 cGy) KGF-/- or KGF+/- mice were transplanted with B6.129-WT bone marrow (A-C) or BALB/c BM (D-F). Lethally irradiated (1100 cGy) B6.129 (G-H) or (900 cGy) BALB/c mice (J-L) were transplanted with KGF-/- or KGF+/- BM and were killed at day 28. Thymic and splenic cellularity was determined, and splenic CD3+, CD4+, and CD8+ T-cell numbers were calculated by flow cytometric analysis. Anti-H2d antibody was used to determine the chimerism in allogeneic experiments. Values represent mean (± SEM) and n = 5-11 per group. *P < .05.
KGF administration enhances thymopoiesis in young and middleaged normal mice. Normal 3-month-old CBA mice received 5 mg/kg KGF or PBS subcutaneously on 3 consecutive days. Mice were harvested at different time points after KGF administration, and thymic and splenic cellularity were determined. (A) Thymic cellularity at days 1, 4, 7, 14, and 28 after KGF administration in young mice. Dotted gray line represents normal number of thymocytes (3-month-old CBA). (B) Splenic CD3+ T-cell numbers at days 4, 7, 14, and 28 after KGF administration in young mice. (C-H) Thymic cellularity and the number of thymocyte subsets were determined at days 4 and 7 after the final administration of KGF. Thymocytes were stained, and subpopulation numbers were calculated as described in Figure 1. Values represent mean (± SEM) and n = 4-8 per group. *P < .05.
KGF administration enhances thymopoiesis in young and middleaged normal mice. Normal 3-month-old CBA mice received 5 mg/kg KGF or PBS subcutaneously on 3 consecutive days. Mice were harvested at different time points after KGF administration, and thymic and splenic cellularity were determined. (A) Thymic cellularity at days 1, 4, 7, 14, and 28 after KGF administration in young mice. Dotted gray line represents normal number of thymocytes (3-month-old CBA). (B) Splenic CD3+ T-cell numbers at days 4, 7, 14, and 28 after KGF administration in young mice. (C-H) Thymic cellularity and the number of thymocyte subsets were determined at days 4 and 7 after the final administration of KGF. Thymocytes were stained, and subpopulation numbers were calculated as described in Figure 1. Values represent mean (± SEM) and n = 4-8 per group. *P < .05.
KGF administration results in increased thymopoiesis in young mice
Having investigated the importance of endogenous KGF in T-cell development, we next explored the effects exerted by exogenous KGF on thymopoiesis and peripheral T-cell development. In these experiments we used the same KGF dose previously used in murine BMT models.19-21 KGF was administered subcutaneously for 3 consecutive days to 3-month-old CBA mice, which were then killed and analyzed for T-cell development in the thymus at different time points up to day 28. We found that KGF administration initially resulted in decreased thymic cellularity on days 1 and 4 (Figure 4A). However, by day 7 thymic cellularity was significantly increased and remained so for at least 4 weeks. When we analyzed thymic subpopulations and DN precursors, we observed an increase in the percentages of DN, SP-CD4, and SP-CD8 populations and a decrease of the DP population in the KGF-treated group at day 4 (Figure 4C-E). Moreover, the percentage and the absolute number of DN3 cells were higher in the KGF-treated group than in the KGF+/- controls (Figure 4E; data not shown). This suggests that a block initially occurred in the transition of DN3 to DN4 but resolved in only a few days, resulting in a net increase in thymic cellularity in every thymic subpopulation by day 7 (Figure 4F-H).
We found no differences in splenic T cells after KGF treatment (Figure 4B). We also analyzed B cells (B220+), granulocytes and their precursors (Gr-1+CD11b-), macrophages (Gr-1+CD11b+), erythroid cells (Ter119+), NK cells (NK1.1+), and dendritic cells (CD 11c+) and found a slight increase in the erythroid population only (data not shown). We also analyzed Foxp3+ CD4+ regulatory T cells after KGF administration in old and young mice and did not observe any differences in the number of regulatory T cells (data not shown). In conclusion, despite causing an early transient developmental block in the thymus, administering KGF to young mice enhances thymopoiesis.
KGF administration enhances thymopoiesis in irradiated, dexamethasone-, and cyclophosphamide-treated mice
To analyze whether KGF administration enhances thymic recovery after irradiation-induced thymic damage, we treated mice with KGF for 3 consecutive days before sublethally irradiating them (450 cGy). We harvested 28 days later to assess T-cell reconstitution. KGF administration resulted in enhanced thymic cellularity in every thymic subset we examined, including DP, SP, and DN populations (Table 1). KGF did not confer protective or beneficial effects on splenocytes or splenic T-cell populations after irradiation.
KGF administration enhances thymic recovery after irradiation, dexamethasone, or cyclophosphamide
. | Irradiation, day 28 . | . | Dexamethasone, day 7 . | . | Cyclophosphamide, day 10 . | . | |||
|---|---|---|---|---|---|---|---|---|---|
. | PBS, × 106 . | KGF, × 106 . | PBS, × 106 . | KGF, × 106 . | PBS, × 106 . | KGF, × 106 . | |||
| Thymus | 26.5 ± 4.6 | 68.2 ± 10.4* | 17.7 ± 2.9 | 47.5 ± 2.0* | 2.7 ± 0.3 | 24.8 ± 4.8* | |||
| DP | 21.3 ± 4.3 | 54.4 ± 8.9* | 15.2 ± 2.5 | 39.5 ± 1.8* | 2.0 ± 0.3 | 20.4 ± 4.1* | |||
| SP-CD4 | 2.6 ± 0.4 | 5.9 ± 0.9* | 0.94 ± 0.2 | 2.45 ± 0.15* | 0.33 ± 0.03 | 1.98 ± 0.41* | |||
| SP-CD8 | 0.8 ± 0.1 | 2.5 ± 0.4* | 0.57 ± 0.12 | 1.92 ± 0.2* | 0.13 ± 0.01 | 0.64 ± 0.11* | |||
| DN1 | 0.1 ± 0.04 | 0.26 ± 0.1 | 0.09 ± 0.02 | 0.11 ± 0.01 | 0.01 ± 0.0004 | 0.03 ± 0.003 | |||
| DN2 | 0.07 ± 0.03 | 0.38 ± 0.07* | 0.04 ± 0.01 | 0.12 ± 0.02* | 0.01 ± 0.001 | 0.04 ± 0.004 | |||
| DN3 | 0.58 ± 0.12 | 2.15 ± 0.41* | 0.26 ± 0.05 | 1.07 ± 0.06* | 0.08 ± 0.02 | 0.49 ± 0.1* | |||
| DN4 | 0.69 ± 0.14 | 1.18 ± 0.3* | 0.4 ± 0.09 | 2.01 ± 0.09* | 0.09 ± 0.02 | 0.91 ± 0.22* | |||
| Spleen | 41.2 ± 6.6 | 49.0 ± 7.4 | 69.4 ± 8.9 | 65.6 ± 5.8 | 92 ± 6.3 | 116 ± 14.7 | |||
| CD4 | 5.5 ± 0.5 | 6.5 ± 0.4 | 11.7 ± 1.5 | 10.4 ± 1.4 | 7.2 ± 0.6 | 8.8 ± 1.4 | |||
| CD8 | 2.3 ± 0.2 | 2.9 ± 0.2 | 7.4 ± 1.0 | 5.7 ± 0.7 | 4.4 ± 0.6 | 6.0 ± 0.9 | |||
. | Irradiation, day 28 . | . | Dexamethasone, day 7 . | . | Cyclophosphamide, day 10 . | . | |||
|---|---|---|---|---|---|---|---|---|---|
. | PBS, × 106 . | KGF, × 106 . | PBS, × 106 . | KGF, × 106 . | PBS, × 106 . | KGF, × 106 . | |||
| Thymus | 26.5 ± 4.6 | 68.2 ± 10.4* | 17.7 ± 2.9 | 47.5 ± 2.0* | 2.7 ± 0.3 | 24.8 ± 4.8* | |||
| DP | 21.3 ± 4.3 | 54.4 ± 8.9* | 15.2 ± 2.5 | 39.5 ± 1.8* | 2.0 ± 0.3 | 20.4 ± 4.1* | |||
| SP-CD4 | 2.6 ± 0.4 | 5.9 ± 0.9* | 0.94 ± 0.2 | 2.45 ± 0.15* | 0.33 ± 0.03 | 1.98 ± 0.41* | |||
| SP-CD8 | 0.8 ± 0.1 | 2.5 ± 0.4* | 0.57 ± 0.12 | 1.92 ± 0.2* | 0.13 ± 0.01 | 0.64 ± 0.11* | |||
| DN1 | 0.1 ± 0.04 | 0.26 ± 0.1 | 0.09 ± 0.02 | 0.11 ± 0.01 | 0.01 ± 0.0004 | 0.03 ± 0.003 | |||
| DN2 | 0.07 ± 0.03 | 0.38 ± 0.07* | 0.04 ± 0.01 | 0.12 ± 0.02* | 0.01 ± 0.001 | 0.04 ± 0.004 | |||
| DN3 | 0.58 ± 0.12 | 2.15 ± 0.41* | 0.26 ± 0.05 | 1.07 ± 0.06* | 0.08 ± 0.02 | 0.49 ± 0.1* | |||
| DN4 | 0.69 ± 0.14 | 1.18 ± 0.3* | 0.4 ± 0.09 | 2.01 ± 0.09* | 0.09 ± 0.02 | 0.91 ± 0.22* | |||
| Spleen | 41.2 ± 6.6 | 49.0 ± 7.4 | 69.4 ± 8.9 | 65.6 ± 5.8 | 92 ± 6.3 | 116 ± 14.7 | |||
| CD4 | 5.5 ± 0.5 | 6.5 ± 0.4 | 11.7 ± 1.5 | 10.4 ± 1.4 | 7.2 ± 0.6 | 8.8 ± 1.4 | |||
| CD8 | 2.3 ± 0.2 | 2.9 ± 0.2 | 7.4 ± 1.0 | 5.7 ± 0.7 | 4.4 ± 0.6 | 6.0 ± 0.9 | |||
Three-month-old CBA mice received 5 mg/kg KGF or PBS subcutaneously on 3 consecutive days. Mice were sublethally irradiated (450 cGy) and harvested 28 days after KGF administration (irradiation columns). Mice were then treated with either dexamethasone (5 mg/kg 24 hours after the last KGF administration) or cyclophosphamide (300 mg/kg 72 hours after the last KGF administration) and harvested at day 7 and day 10, respectively. Thymic and splenic cellularities were analyzed and thymocytes were stained with anti-CD4, -CD8, -CD3, -CD44, and -CD25 antibodies. Thymic subpopulations were calculated from thymic cellularity and the percentage of particular subsets analyzed by multicolor flow cytometry (DP, CD4+ CD8+; DN, CD4− CD8−; DN1, CD44+ CD25− CD4− CD8− CD3−; DN2, CD44+ CD25+ CD4− CD8− CD3−; DN3, CD44− CD25+ CD4− CD8− CD3−; and, DN4, CD44− CD25− CD4− CD8− CD3−). Splenocytes were stained with anti-CD3, -CD4, and -CD8 antibodies, and the number of T cells was calculated by splenic cellularity and multicolor flow cytometry. Values represent mean (± SEM) and n = 4-8 mice per group.
Significantly different from control group.
We also administered dexamethasone (DEX), a synthetic adrenocortical steroid known to suppress thymopoiesis,24 and examined the potential of KGF to enhance thymic recovery. As DEX treatment is known to induce early apoptosis in the thymus after a single injection, we killed the mice at an earlier time point (day 7 versus day 28) after sublethal irradiation. KGF administration resulted in faster recovery kinetics from DEX-induced damage and increased thymic cellularity, when analyzed at day 7 after treatment (Table 1). Again, all thymic subsets were increased in the KGF-treated group, whereas no effect on splenic reconstitution was observed. Finally, we assessed the effects of KGF prophylaxis after cyclophosphamide treatment and again found a significant improvement in thymic recovery but no effect on splenic T-cell reconstitution (Table 1). KGF administration can enhance thymic recovery from damage caused by irradiation, dexamethasone, and cyclophosphamide.
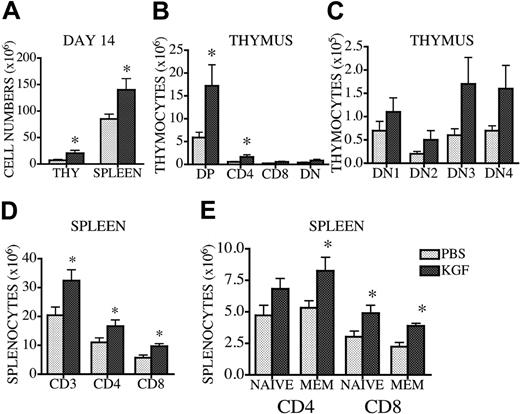
KGF administration enhances thymopoiesis and increases the number of peripheral T cells in 18-month-old mice
Aging is associated with decreased thymic cellularity because of thymic involution.25 To determine whether KGF administration would enhance thymopoiesis in aged mice, we treated 18-month-old CBA mice with KGF for 3 days. This resulted in a 3-fold increase in overall thymic cellularity and the number of DP thymocytes at day 14 after treatment (Figure 5A-B). DN subsets (especially DN3) also increased after KGF treatment, although no statistical significance was reached (Figure 5C). Interestingly, KGF also increased the number of total splenocytes and splenic T cells (both naive and memory T cells), which we did not observe in previous experiments with young mice (Figure 5D-E). T-cell function was also tested using mixed lymphocyte reactions (MLRs) and Concanavalin A stimulation after KGF administration, and no differences in T-cell proliferation were observed in either young or old mice (data not shown).
Thus, KGF administration can enhance thymopoiesis and peripheral T-cell numbers in 18-month-old mice.
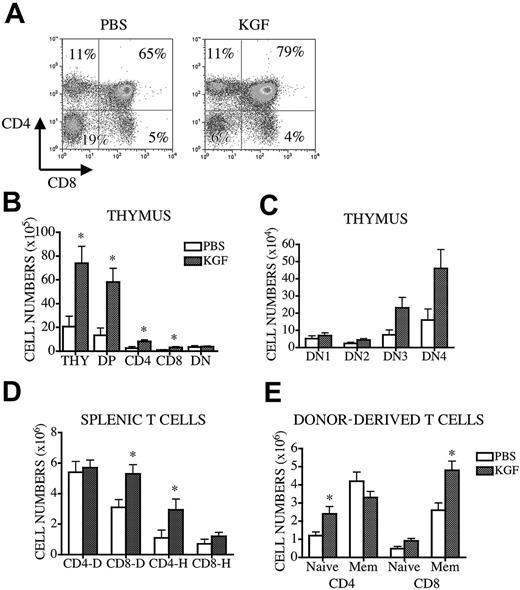
KGF administration enhances thymopoiesis in middle-aged recipients of allogeneic BMT
Finally, we investigated the effects of KGF on T-cell development in middle-aged (12-month-old) recipients of allogeneic BMT. We chose a well-described, clinically relevant murine MHC-matched allogeneic BMT model with minor antigen disparities: B10.BR→CBA/J.26 We observed a decreased percentage of DP cells (63.1% ± 4.6%; normal, 76%-80%) and an increased percentage of DN cells (18.1% ± 4.2; normal, 3%-5%), which suggested a partial block in the DN to DP transition in the middle-aged recipients of allo BMT (Figure 6A, left). KGF administration not only increased thymic cellularity and the number of DP and SP thymocytes but also improved the thymocyte subset distribution when analyzed at day 56 after BMT (Figure 6A, right, and 6B). KGF did not affect the relative distribution of DN populations (Figure 6C). KGF treatment also enhanced posttransplantation peripheral T-cell development. KGF-treated recipients had increased numbers of donor-derived CD8+ and naive CD4+ T cells, as well as host-derived CD4+ T cells (Figure 6D-E). Pre-BMT KGF administration to middle-aged recipients of an allogeneic BMT resulted in increased thymopoiesis and peripheral T-cell numbers.
KGF administration enhances thymopoiesis and peripheral T-cell development in 18-month-old mice. Eighteen-month-old CBA mice received 5 mg/kg KGF or PBS subcutaneously on 3 consecutive days. Mice were harvested 14 days after KGF administration, and thymic and splenic cellularity were determined. (A) Thymic and splenic cellularity are depicted. (B-C) Thymic subpopulations were calculated as described in Figure 1. (D) Splenic T-cell numbers were determined as described in Table 1. (E) Splenocytes were stained with anti-CD4, -CD8, and -CD44 antibodies, and the percentages of T cells (naive CD4, CD4+CD44-; memory CD4, CD4+CD44+; naive CD8, CD8+CD44-; and memory CD8, CD8+CD44+) were determined by multicolor flow cytometry. Values represent mean (± SEM) and n = 4-8 per group. *P < .05.
KGF administration enhances thymopoiesis and peripheral T-cell development in 18-month-old mice. Eighteen-month-old CBA mice received 5 mg/kg KGF or PBS subcutaneously on 3 consecutive days. Mice were harvested 14 days after KGF administration, and thymic and splenic cellularity were determined. (A) Thymic and splenic cellularity are depicted. (B-C) Thymic subpopulations were calculated as described in Figure 1. (D) Splenic T-cell numbers were determined as described in Table 1. (E) Splenocytes were stained with anti-CD4, -CD8, and -CD44 antibodies, and the percentages of T cells (naive CD4, CD4+CD44-; memory CD4, CD4+CD44+; naive CD8, CD8+CD44-; and memory CD8, CD8+CD44+) were determined by multicolor flow cytometry. Values represent mean (± SEM) and n = 4-8 per group. *P < .05.
Pretransplantation administration of KGF in middle-aged recipients of allogeneic BM transplant increases thymopoiesis. Lethally irradiated (1300 cGy) 12-month-old CBA mice were transplanted with T-cell-depleted B10.BR BM (10 × 106). Recipients received either KGF (5 mg/kg/d) or PBS subcutaneously from days -6 to -4. Mice were harvested at day 56, and thymic and splenic cellularities were determined. (A-C) Thymic subpopulations were calculated as described in Figure 1. (D) The number of donor- or host-derived splenic CD4+ and CD8+ T cells from recipients of allogeneic BM transplant are shown. (E) Splenocytes stained with anti-CD4, -CD8, -CD44, and -Ly9.1 antibodies and donor-derived naive and memory T-cell numbers were calculated as described in Figure 5. Values represent mean (± SEM) and n = 6 mice per group. *P < .05.
Pretransplantation administration of KGF in middle-aged recipients of allogeneic BM transplant increases thymopoiesis. Lethally irradiated (1300 cGy) 12-month-old CBA mice were transplanted with T-cell-depleted B10.BR BM (10 × 106). Recipients received either KGF (5 mg/kg/d) or PBS subcutaneously from days -6 to -4. Mice were harvested at day 56, and thymic and splenic cellularities were determined. (A-C) Thymic subpopulations were calculated as described in Figure 1. (D) The number of donor- or host-derived splenic CD4+ and CD8+ T cells from recipients of allogeneic BM transplant are shown. (E) Splenocytes stained with anti-CD4, -CD8, -CD44, and -Ly9.1 antibodies and donor-derived naive and memory T-cell numbers were calculated as described in Figure 5. Values represent mean (± SEM) and n = 6 mice per group. *P < .05.
Discussion
The effects of KGF on mesenchymal-epithelial interactions in the immune system, particularly in the thymus, suggest that it might be profitable to explore the administration of KGF as a therapeutic strategy to enhance T-cell development. We have shown that endogenous stromal cell-derived KGF is required for postnatal thymic regeneration after irradiation-induced thymic damage and that administration of exogenous KGF can (1) enhance thymopoiesis in young and middle-aged mice; (2) enhance thymic recovery after irradiation, dexamethasone, or cyclophosphamide; and (3) improve posttransplantation T-cell reconstitution.
Our data suggest that KGF is not required for prenatal or postnatal development under normal circumstances. Deficiency in KGF may be compensated for by the actions of other fibroblast growth factors, especially FGF-10, which plays a role in thymic epithelial cell (TEC) development and, like KGF, is a FGFR2IIIb ligand.9,27 Indeed, FGF-10-deficient mice (similar to FGFR2IIIb-/- mice) have a significant reduction in thymic cellularity.27 However, we also showed that KGF-/- mice were more vulnerable to sublethal irradiation and that their thymic recovery was delayed, although they did not have a defect in hematopoietic recovery. This suggests that stromal cell-derived KGF is required for postnatal thymic regeneration after irradiation.
Our BMT experiments using KGF-/- mice as donors or hosts indicated that nonhematopoietic stromal cells produced KGF required for thymic recovery after irradiation. However, this does not completely rule out the contribution of thymocytes to KGF production (reported by Erickson et al10 ) because (1) there is a lack of donor-derived mature thymocytes in the early posttransplantation period and (2) a small number of residual mature host thymocytes can be present in the thymus after irradiation and BMT.
KGF has been studied as a cytoprotective agent, because of its unique effect on epithelial cells in various tissues (intestine, lung, skin, etc).12 We did not observe any defects in the thymic architecture of KGF-/- mice; however, KGF has been shown to protect the thymic architecture and most of the cortical and medullary TECs during GVHD in vivo18 and increases BrdU+ TECs in reaggregate thymic organ cultures in vitro.9 Thymic epithelial cells play a critical role in the differentiation and survival of immature thymocytes via different mechanisms, including the production of IL-7 and chemotactic molecules, such as thymic epithelial chemokine (TECK, or CCL25)28 and macrophage inhibitory protein-1β (MIP-1β),29 and by providing WNT signals for thymocytes.30 WNT signals activate β-catenin-mediated transcription of T-cell factor-1 (TCF-1) and lymphoid enhancer factor (LEF), which enhance the survival of immature thymocytes.30,31 Min et al32 reported that KGF-treated BM transplant recipients had significantly more IL-7 mRNA+ cells in their thymus than the controls did, and that KGF treatment had no effect on thymic reconstitution in BMT recipients deficient for IL-7, suggesting that the effects of KGF on thymopoiesis are dependent on the presence of IL-7. Interestingly, our preliminary studies on thymocyte signaling showed that KGF administration resulted in significantly increased levels of serine-727-phosphorylated STAT-3 in thymocytes, instead of the expected IL-7-induced increase in phosphorylated STAT-5A,33 suggesting that KGF perhaps supports thymocyte development through alternative pathways.
KGF may also enhance stem cell entry into the thymus. Studies by Foss et al34 and Prockop and Petrie35 have suggested that the entry of lymphoid precursors from the BM into the thymus is a gated phenomenon and depends on the opening and closing of individual thymic niches, which accommodate the development of lymphoid precursors into mature T cells. Once a lymphoid precursor enters an empty thymic niche, the niche closes; KGF may increase the number of gates in the thymus, thus helping to improve thymopoiesis after insults such as chemo/radiotherapy and BMT. In preliminary studies, we observed that administering KGF before transplantation with an infusion of T-cell precursors increased thymic cellularity after BMT, when compared with infusing T-cell precursors alone (J. L. Zakrzewski and O. A., unpublished observation, June 2005).
Involution of the thymus and diminished output of T lymphocytes are some of the major changes that take place in an aging immune system (reviewed in Linton and Dorshkind25 ). Thymic involution leads to thymic atrophy, causing decreased thymic cellularity and significant alteration of the thymic microenvironment.36-38 Our studies showed that KGF deficiency does not accelerate thymic involution (Figure S1). It was recently reported that the frequency of intrathymic T-cell precursors declines with age and that their potential to reconstitute the thymus is diminished.39 Our data indicate that KGF treatment can enhance thymopoiesis and T-cell development in mice as old as 18 months. This suggests that stimulating TECs augments thymopoiesis, despite any intrinsic stem cell defects in an aged population.40,41
Immune reconstitution is impaired after bone marrow transplantation, and all BM transplant recipients are subject to varying periods of immune deficiency after transplantation. Thymopoiesis is significantly enhanced in experimental BMT models when KGF treatment is added.32 KGF administration also protects the thymic microenvironment from alterations that are normally seen during acute GVHD.18 However, both studies were performed in a parent-F1 model with young recipients and did not address clinically relevant conditions, such as unrelated BMT and BMT in adult populations. A longitudinal analysis of lymphoid reconstitution after BMT and life-threatening opportunistic infections in transplant recipients demonstrated that adult recipients of TCD unrelated BM transplants had prolonged and severe deficiencies in CD3+, CD4+, and CD8+ T-cell numbers and significantly extended recovery times, when compared with pediatric recipients of TCD unrelated BM transplants.42 We therefore chose a MHC-matched with minorAg disparity model and used 12-month-old (middle-aged) mice in our allogeneic BMT experiments to demonstrate the feasibility of pre-KGF administration in adult recipients of BM transplants.
We did find increased numbers of donor-derived T cells in the spleen 2 months after transplantation in old recipients of allogeneic BM transplants and KGF (Figure 6), but not in nonirradiated normal young recipients or recipients of sublethal irradiation, which were analyzed at earlier time points during their T-cell recovery. Min et al32 also found that pretransplantation treatment with KGF enhanced posttransplantation peripheral T-cell reconstitution in a parent-into-F1 allogeneic BMT model. Interestingly, the maximum increase in peripheral T-cell reconstitution from KGF was not reached until at least 2 to 3 months after transplantation. We used RAG2-eGFP transgenic (Tg) mice to determine the effects of increased thymopoiesis on peripheral T-cell reconstitution 2 to 4 weeks after KGF administration (RAG2-eGFP Tg mice were kindly provided by Dr Michel Nussenzweig, Rockefeller University, New York, NY). We found similar numbers of eGFPhigh peripheral T cells, which represent recent thymic emigrants, in KGF-treated mice and in control-treated mice. There was no difference in the fraction of Annexin-V+ apoptotic cells in the thymus and in the spleen between the KGF-treated group and the control group. RAG2-eGFP Tg mice then were used as donors in allogeneic BMT experiments, and we again found similar numbers of eGFPhigh and eGFPlow T cells in the periphery, when analyzed at day 28 after BMT. Taken together, we hypothesize that an effect of KGF-induced increased thymopoiesis will not be immediately apparent in the periphery, and, for example, recipients of an allogeneic BM transplant treated with KGF will not have significantly increased numbers of peripheral T cells until at least 2 months after transplantation. In addition, the effects of KGF on T-cell recovery might be increased in older recipients because of the positive effects of KGF on age-associated thymic involution and (possibly) peripheral T-cell deficiency.
We noted a significant increase in residual host CD4+ T cells in KGF-treated recipients of an allo BM transplant. Previous studies showed a similar effect on residual T cells in the early posttransplantation period,32 which suggests that KGF administration could have extrathymic effects on residual T cells. This effect could be mediated through indirect action on stromal cells in secondary lymphoid organs. In this context, it is interesting to note that we observed a consistent, statistically significant decrease in splenic cellularity in our KGF-/- mice (Figure 1A).
We have previously shown that homeostatic proliferation of de novo-generated donor T cells plays an important role in posttransplantation T-cell reconstitution, especially CD8+ T-cell reconstitution.43 T cells acquire a memory phenotype while undergoing homeostatic expansion.43-45 It is therefore not surprising that the overall increase in newly generated donor CD8+ T cells consists primarily of the memory phenotype (Figure 6E).
We believe that KGF has a broader therapeutic potential than as an antimucositis agent alone. The interaction between TECs and thymocytes suggests that KGF may be used effectively as a thymopoietic growth factor to enhance and protect immune reconstitution in various conditions of immune deficiency, which is an increasingly important clinical condition, because of the increasing number of patients with HIV, a proportionally increased aged population, and patients undergoing chemotherapy and radiation therapy.
Prepublished online as Blood First Edition Paper, November 22, 2005; DOI 10.1182/blood-2005-07-2831.
Supported by the National Institutes of Health (grants HL69929, HL72412, CA33049, and CA107096 [M.R.M.v.d.B.]) and (grant P20-CA103694 [O.A.]) and by awards from the Emerald Foundation and The Experimental Therapeutics Center of Memorial Sloan-Kettering Cancer Center funded by Mr William H. Goodwin and Mrs Alice Goodwin and the Commonwealth Foundation for Cancer Research (M.R.M.v.d.B.). Ö.A. is the recipient of an Amy Strelzer Manasevit Scholar Award from The National Marrow Donor Program (NMDP) and The Marrow Foundation.
The online version of this article contains a data supplement.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 U.S.C. section 1734.
We thank the staff of the Research Animal Resource Center for excellent animal care.





This feature is available to Subscribers Only
Sign In or Create an Account Close Modal