Abstract
We have recently described a CD19– B220+CD117low bone marrow subpopulation with B, T, and myeloid developmental potential, which we have called “early progenitors with lymphoid and myeloid potential” or EPLM. These cells also expressed Fms-like tyrosine kinase 3, Flt3, or CD135. Treatment of mice with the corresponding ligand, Flt3L, showed a 50-fold increase in EPLM. In addition to the expected increase in dendritic cell numbers, Flt3L treatment had a reversible inhibitory effect on B lymphopoiesis. Limiting dilution analysis of sorted EPLM from Flt3L-treated mice showed that B-lymphocyte progenitor activity was reduced 20-fold, but that myeloid and T-cell progenitor activity was largely preserved. EPLM from treated mice transiently reconstituted the thymus and bone marrow of recipient mice, generating cohorts of functional T and B cells in peripheral lymphoid organs. Thus, Flt3L treatment results in a dramatic increase in a novel bone marrow cell with lymphoid and myeloid progenitor activity.
Introduction
In the bone marrow (BM) of normal adult mice, we have recently characterized a subpopulation of B220+CD117low cells with T, B, and myeloid developmental potential.1 As a population, B220+CD117low BM cells are heterogeneous, containing about 95% CD93+CD19+ committed B-cell progenitors, about 3% CD93–NK1.1+ natural killer (NK) progenitors, and about 2% CD19–NK1.1– cells. Further analysis indicated that this latter CD19–NK1.1– subpopulation expressed CD127+ (IL-7Rα+) and CD135, the so-called fetal liver kinase 2 (flk-2), also known as Fms-like tyrosine kinase 3 (Flt3). When cultured on different stromal cells and in the presence of appropriate cytokines, including Flt3 ligand (Flt3L), sorted CD19–NK1.1–B220+CD117low cells showed, in addition to potent B-cell potential, T and myeloid potential. When injected intravenously into sublethally irradiated immunodeficient Rag2.γc recipients, freshly isolated cells transiently reconstituted both the T- and B-cell progenitor compartments, giving rise to cohorts of both mature T and B cells. Myeloid reconstitution in vivo was difficult to demonstrate. Based on these phenotypic and functional results, we have called these cells early progenitors with lymphoid and myeloid potential, or EPLM.
In the adult BM, the Flt3 kinase (CD135) is expressed by cells at many different stages of lymphoid and myeloid development,2-4 starting with CD117high, Lin– multipotent high proliferative potential progenitors and including both CD117low, Lin– so-called common lymphoid progenitors (CLPs)5 and EPLM.1 Expression levels of CD135 tend to decrease with increasing differentiation. In the B-cell lineage CD135 expression is lost on CD19 acquisition but in the myeloid lineage CD135 expression persists particularly during dendritic cell (DC) development.4 Thus, treatment of mice with the ligand for Flt3 (Flt3 ligand [Flt3L]) is a frequently used protocol for increasing the number of BM DC progenitors.6
Flt3L is a type 1 membrane protein expressed on multiple cell types including BM stroma and lymphoid cells.7,8 Flt3L can also exist in soluble form and both membrane-bound and soluble forms activate the corresponding CD135 receptor.9 Generally, Flt3L acts in combination with other cytokines,10 for example, stem cell factor,4 GM-CSF, IL-4, IL-7,11 and IL-15.12 It has been reported that B lymphopoiesis increases following Flt3L treatment11 and decreases in Flt3 knock-out (KO) mice.13 In addition, Flt3L treatment is known to result in the accumulation of B220+CD4+ and B220+CD8+, so-called “plasmacytoid” DCs, in both BM and spleen6,14 ; there are also reports of an increase in NK cells.15 However, to our knowledge, a detailed phenotypic and functional analysis of the effect of Flt3L treatment on nondendritic BM progenitors has not been carried out.
Because the in vitro culture systems for the analysis of the commitment status of EPLM contained Flt3L1 and because freshly isolated cells expressed the corresponding receptor, CD135, we wanted to see what effect in vivo treatment with Flt3L would have on the B220+CD117low BM compartment. Results obtained indicated that after 10 days of Flt3L injection, the proportion and absolute number of EPLM among B220+CD117low cells increased dramatically. However, the frequency of B-cell progenitors among these EPLM was reduced 20-fold. Associated with the reduced in vitro B-cell potential of EPLM and reduced proportion of CD19+ cells among B220+CD117low cells, BM B lymphopoiesis was drastically reduced. Freshly isolated EPLM from Flt3L-treated mice were fully capable of functionally reconstituting the T- and B-cell compartments of immunodeficient recipient mice. Thus, treatment with Flt3L provides a valuable way of expanding a novel BM cell that has both B- and T-cell progenitor activity.
Materials and methods
Mice
Female C57BL/6, C57BL/6-Rag2–deficient (Rag2–/–),16 and C57BL/6.Ly5.1 congenic mice of 5 to 8 weeks of age were used. All mice were bred and maintained in our animal facility under specific pathogen–free conditions. All animal experiments were carried out within institutional guidelines.
Flt3L treatment
Recombinant human Flt3L (rFlt3L) was a kind gift of Amgen (Thousand Oaks, CA). A stock solution containing 50 μg rFlt3L/mL was prepared in PBS and aliquots stored at –20°C until use. For Flt3L treatment, mice generally received 10 μg rFlt3L (0.2 mL) by intraperitoneal injection daily for 10 days, a treatment schedule previously used to increase DC number.6 In titration experiments (“Results”), mice were treated with graded doses of 0.4, 2, or 10 μg/0.2-mL injection. Control mice received 0.2 mL PBS. Mice were humanely killed by CO2 inhalation and organs removed by standard procedures.
Stromal cell lines
OP9 or OP9 stromal cells expressing the Notch ligand δ-like-1 (OP9-DL1) were kindly provided by Professor Juan-Carlos Zúñiga-Pflücker (University of Toronto, ON, Canada) and maintained in IMDM (Life Technologies, Basel, Switzerland) supplemented with 5 × 10–5 M β-mercaptoethanol, 1 mM glutamine, 0.03% (wt/vol) Primatone (Quest, Naarden, The Netherlands), 100 U/mL penicillin, 100 μg/mL streptomycin, and 2% heat-inactivated fetal bovine serum (FBS). ST-2 stroma was also maintained in the medium.
Flow cytometry and cell sorting
FITC-, PE-, APC-, or biotin-conjugated monoclonal antibody (mAb) specific for B220, NK1.1 (PK 136), CD11b (M1/70), CD11c (HL3), CD3ϵ (2C-11), CD4 (GK1.5), CD8α (53-6.7), CD19 (1D3), CD21 (CR2/CR1), CD23 (B3B4), CD25 (7D4), CD44 (IM7), CD45.1 (A20), TCRβ (H57), and TCRγ/δ (GL3) were purchased from BD Biosciences (Milan, Italy); APC-conjugated CD117 (2B8), PE-conjugated CD127 (A7R34), and PE-conjugated CD135 (A2F10) were purchased from eBioscience (San Diego, CA); anti-C1qRp (PB493, CD93) antibody was purified from the hybridoma supernatant and labeled with biotin by standard methods. Staining of cells was performed as previously described. Flow cytometry was performed using a fluorescence-activated cell sorting FACSCalibur (BD Biosciences) and data were analyzed using the CellQuest Pro Software (BD Biosciences). For cell sorting the FACS Aria (BD Biosciences) was used. Erythrocyte-depleted BM cells were stained in IMDM 2% FBS with saturating concentrations of anti-B220FITC, anti-CD19PE plus anti-NK1.1PE, anti-CD117APC, and biotinylated anti-CD93 (PB493/AA4.1). Following a 30-minute incubation at 4°C, cells were washed in PBS 2% FBS and resuspended in PBS containing SAPE/Cy7. After a further 30 minutes at 4°C, cells were washed in IMDM, filtered through a 20-μm diameter nylon mesh, and resuspended at about 20 × 106/mL in filtered PBS 2% FBS prior to sorting. Reanalysis of sorted cells indicated that in all instances they were more than 98% pure.
Stromal cell cocultures
Two days prior to coculture, 2 × 103 stromal cells were seeded per well of a 96-well flat bottom plate or 104 per well of a 24-well plate. At day 0, stromal cells had grown to semiconfluency and were then γ-irradiated with 3000 rad. Then, culture medium was removed and replaced by IMDM supplemented with 5 × 10–5 M β-mercaptoethanol, 1 mM glutamine, 0.03% (wt/vol) primatone, 100 U/mL penicillin, 100 μg/mL streptomycin, 2% FBS. To these cultures were added various numbers of sorted BM cells. For limiting dilution analysis (LDA), 48 replicate cultures containing graded numbers of sorted EPLM were plated in 96-well microtiter plates containing irradiated ST2 stroma plus Flt3L for myeloid, OP9 plus IL-7 and Flt3L for B, or OP9-DL1 stroma plus IL-7 and Flt-3L for T cells. The presence of Flt3L in stromal cell cultures did not alter results obtained. At day 14 of culture, all wells were inspected using an inverted microscope. Wells containing colonies of more than 50 cells were scored positive.
Transplantation
Groups of C57Bl/6.Rag2–/– mice were irradiated with 450 rad and 1 to 10 × 105 cells injected intravenously. After the indicated time, mice were humanely killed and organ cell suspensions prepared by mechanical disruption, stained, and subsequently analyzed by flow cytometry. In some experiments (“Results”), recipients also received daily injections of 4 μg Flt3L.
Immunizations
To induce a T-dependent antibody response, reconstituted mice were injected intraperitoneally with 50 μg alum-precipitated NIP-ovalbumin. Sera were obtained from tail bleeding prior to and 14 days after immunization and stored at –20°C. Total and hapten-specific IgG levels were determined by enzyme-linked immunosorbent assay (ELISA).
Results
Cells obtained from the BM of PBS control or Flt3L-treated mice were stained with combinations of mAb used to define early lymphoid progenitors. As shown in Figure 1A, upper left panels, staining with B220FITC versus CD117APC revealed the expected 1.5% B220+CD117low subpopulation (boxed insert). Further analysis (right panel) of gated B220+CD117low cells shows that they contained about 93% CD93+CD19+, 3% CD93–NK1.1+, and about 4% cells with a broad CD93 staining pattern but negative for both CD19 and NK1.1. As previously shown,1 the latter cells comprise EPLM.
The lower panels of Figure 1A show the strikingly different staining pattern obtained with BM cells from mice treated with 10 μg/injection of Flt3L, a dose taken from previously published reports.6 First, the proportion of B220+CD117low cells increased to 4.9% of gated cells and, second, there is a drastic alteration in CD93 versus CD19/NK1.1 distribution. Whereas the proportion of NK1.1+ cells is about the same (2.2%), the proportion of CD19+ cells is reduced to only 6% and cells negative for both CD19 and NK1.1 increased from about 4% to over 90%. Again, the CD93 distribution on CD19– cells is unchanged compared with controls (upper panel). Tables 1 and 2 summarize the numbers of cells in each subpopulation from a series of 5 independent groups of treated mice.
BM cell numbers in Flt3L-treated mice
Group . | Total, 2 femora . | CD19+ . | B220+ CD19- . | B220+ CD117+ . | B220+CD19- CD117+NK1.1- . | IgM+ . | RecB* . | CD11c+ . |
|---|---|---|---|---|---|---|---|---|
| PBS | 25.6 | 9.8 | 1 | 0.38 | 0.02 | 3.2 | 1.6 | 0.5 |
| Flt3L | 25.7 | 1.4 | 7.2 | 1.26 | 1.15 | 0.35 | 0.05 | 8.5 |
| Ratio† | 1.0 | 0.14 | 7.2 | 3.3 | 57 | 0.1 | 0.03 | 17 |
Group . | Total, 2 femora . | CD19+ . | B220+ CD19- . | B220+ CD117+ . | B220+CD19- CD117+NK1.1- . | IgM+ . | RecB* . | CD11c+ . |
|---|---|---|---|---|---|---|---|---|
| PBS | 25.6 | 9.8 | 1 | 0.38 | 0.02 | 3.2 | 1.6 | 0.5 |
| Flt3L | 25.7 | 1.4 | 7.2 | 1.26 | 1.15 | 0.35 | 0.05 | 8.5 |
| Ratio† | 1.0 | 0.14 | 7.2 | 3.3 | 57 | 0.1 | 0.03 | 17 |
BM cell numbers are shown as ×10-6.
RecB refers to B220bright IgMlow recirculating B cells.
Ratio is of cell number in Flt3L-treated versus PBS control.
Spleen cell numbers in Flt3L-treated mice
Group . | Total . | CD19+ . | B220+ CD19- . | FolB . | MZB . | CD4+ . | CD8+ . | CD11c . | NK . |
|---|---|---|---|---|---|---|---|---|---|
| PBS | 76 | 41.2 | 0.6 | 32.5 | 2 | 12.6 | 7.5 | 1.5 | 2.6 |
| Flt3L | 138 | 56.9 | 11.9 | 43.6 | 3 | 12.1 | 10.1 | 32.3 | 7.3 |
| Ratio* | 1.8 | 1.4 | 19.8 | 1.3 | 1.5 | 0.96 | 1.35 | 21.5 | 2.8 |
Group . | Total . | CD19+ . | B220+ CD19- . | FolB . | MZB . | CD4+ . | CD8+ . | CD11c . | NK . |
|---|---|---|---|---|---|---|---|---|---|
| PBS | 76 | 41.2 | 0.6 | 32.5 | 2 | 12.6 | 7.5 | 1.5 | 2.6 |
| Flt3L | 138 | 56.9 | 11.9 | 43.6 | 3 | 12.1 | 10.1 | 32.3 | 7.3 |
| Ratio* | 1.8 | 1.4 | 19.8 | 1.3 | 1.5 | 0.96 | 1.35 | 21.5 | 2.8 |
Spleen cell numbers are shown as × 10-6.
Ratio is of cell number in Flt3L-treated versus PBS control.
Reduction in in vivo B lymphopoiesis
Staining of BM cell suspensions with a panel of markers defining the different stages of B lymphopoiesis unexpectedly revealed a drastic reduction in B lymphopoiesis in treated mice (Figure 1B). Shown are cytogram displays of cells stained with B220 versus CD19 (left panels) or B220 versus IgM (right panels). Classically, in PBS-treated controls (upper panels), 45% BM cells were B220+CD19+ B cells and 3.5% mostly B220bright, CD19– cells. As previously shown,17 B220bright, CD19– cells comprise a heterogeneous mixture of both mature CD4– NK and plasmacytoid DCs. Staining with B220 and IgM shows the expected pattern of B220+IgM– (mostly pre-BII), B220+IgMlow (immature B), B220+IgMbright (mature B), and B220brightIgMlow (recirculating) B cells. In the BM of treated mice (lower panels), the proportion of CD19+ B cells was reduced from 45% to only 2.8% gated cells. Additional staining with IgM showed that only a small population of immature B cells remained, with an almost total absence (0.9%) of IgMbright mature and B220bright IgMlow recirculating B cells.
Effects of Flt3L treatment on EPLM, B lymphopoiesis, and DCs. (A) Effect of Flt3L treatment on EPLM. The left cytograms show the B220 versus CD117 distribution on forward/side scatter-gated bone marrow cells from PBS-injected (top panels) or Flt3L-injected mice (bottom panels). The boxed areas indicate B220+CD117low cells and the figures the percent cells in these areas. The right cytograms show the CD93 versus CD19/NK1.1 distribution on gated B220+CD117low cells and the percent cells in each quadrant. (B) Effect of Flt3L treatment on B lymphopoiesis. Shown are cytogram displays of BM cells from PBS-treated (top panels) or Flt3L-treated (bottom panels) stained with the indicated markers. In each panel, the figures represent the percent cells in each quadrant. (C) Effect of Flt3L treatment on DCs. Shown are cytogram displays of B220 versus CD11c on BM (left) or spleen cells (right) from PBS-(top) or Flt3L-treated mice. The figures in each panel indicate the percent cells in each quadrant. (D) Flt3L dose response. Shown is a summary of the BM (left) or spleen (right) profile of mice treated with 10 daily injections of the indicated dose of Flt3L. Each histogram shows, on a logarithmic scale, the mean ± SD number of cells in the indicated subpopulations from groups of 3 mice. Note the different vertical scale for BM and spleen.
Effects of Flt3L treatment on EPLM, B lymphopoiesis, and DCs. (A) Effect of Flt3L treatment on EPLM. The left cytograms show the B220 versus CD117 distribution on forward/side scatter-gated bone marrow cells from PBS-injected (top panels) or Flt3L-injected mice (bottom panels). The boxed areas indicate B220+CD117low cells and the figures the percent cells in these areas. The right cytograms show the CD93 versus CD19/NK1.1 distribution on gated B220+CD117low cells and the percent cells in each quadrant. (B) Effect of Flt3L treatment on B lymphopoiesis. Shown are cytogram displays of BM cells from PBS-treated (top panels) or Flt3L-treated (bottom panels) stained with the indicated markers. In each panel, the figures represent the percent cells in each quadrant. (C) Effect of Flt3L treatment on DCs. Shown are cytogram displays of B220 versus CD11c on BM (left) or spleen cells (right) from PBS-(top) or Flt3L-treated mice. The figures in each panel indicate the percent cells in each quadrant. (D) Flt3L dose response. Shown is a summary of the BM (left) or spleen (right) profile of mice treated with 10 daily injections of the indicated dose of Flt3L. Each histogram shows, on a logarithmic scale, the mean ± SD number of cells in the indicated subpopulations from groups of 3 mice. Note the different vertical scale for BM and spleen.
Increase in in vivo DC development
As expected, treatment with Flt3L resulted in a dramatic increase in both BM and spleen DC subsets.14,18 Confirming previous reports,6,19 in the BM of control mice (Figure 1C upper left) 2.2% cells were B220brightCD11clow DCs, whereas the spleen (right panel) contained 2.3% B220–CD11cbright and 0.8% B220+CD11cdull DC subpopulations. After Flt3L treatment, the BM (lower left) contained not only 23.1% B220brightCD11clow, but also 16.2% B220–CD11c+ cells; the CD11c expression of the latter extended from CD11cdull to CD11cbright. Likewise the spleen contained 23% B220+ and 9.4% B220– subpopulations of CD11c+ cells.
Tables 1 and 2 summarize the mean cell numbers in the different BM and spleen subpopulations. For B cells, results indicate the severe (approximately 7-fold) reduction in BM B-cell numbers, which is mostly manifest as a reduction in B-cell development beyond the pre–B cell stage. In contrast, the dramatic increase in DC development is seen by the 17-fold increase in CD11c+ cells. For the spleen, B-cell development, as measured by total numbers of CD19+, CD23+CD21+ follicular B (FB), or CD23lowCD21+ marginal zone B (MZB) cells is not drastically altered. The number of mature spleen T cells was unchanged and there was a slight increase in NK1.1+ cell numbers. However, the most dramatic changes were seen in DC numbers, with an approximately 20-fold increase in both plasmacytoid B220+ and conventional B220–CD11c+ cells. There was no discernible effect on the T-cell progenitor compartment of the thymus (data not shown).
To monitor how the dose of injected Flt3L affected BM and spleen cell recoveries, groups of mice were treated with graded doses of Flt3L and the cellularity and phenotype of BM and spleen directly compared. As shown in Figure 1D, left panel, there was a dose-dependent increase of BM EPLM, B220+, and B220– DC numbers and concomitant decrease in IgM+ B cells. For DCs, there was a plateau in cell number between the 2- and 10-μg dose. In the spleen (right panel), there was a slight (< 3-fold) increase in total cellularity with a relatively constant number of IgM+ B cells. As expected, most of the additional cellularity was accounted for by plasmacytoid and conventional DCs, whose number progressively increased with increasing Flt3L dose.
To show that Flt3L treatment had only transient effects on BM B lymphopoiesis and DC generation, mice initially treated for 10 days with Flt3L were allowed to recover for 2 weeks and BM and spleen cellularity and phenotype were monitored. It is known that the turnover of B cells in the BM is relatively rapid, whereas most peripheral B cells are long-lived.20,21 Short-term treatment of mice with Flt3L had no discernible effect on mature B cells and 2 weeks after stopping treatment, B lymphopoiesis in the BM had returned to normal (data not shown). For DCs, it is known that their turnover in the periphery is relatively rapid.22,23 Therefore, not surprisingly, following the cessation of Flt3L treatment, the BM and spleen DC compartments were indistinguishable from controls (data not shown).
In vitro functional analysis of CD19–NK1.1–B220+CD117low cells from Flt3L-treated mice
We have used appropriate high plating efficiency stromal cell culture systems and LDA to assess the developmental potential of various sorted BM and thymus-derived lymphoid progenitors.1,24 In this way, we could show that CD19–NK1.1–B220+CD117low BM cells, or EPLM, possessed potent B, T, and myeloid developmental potential. Therefore, we wished to compare the developmental potential of EPLM from PBS-treated, control, with those from Flt3L-treated mice. To this end, we sorted CD19–NK1.1–B220+CD117low BM cells from Flt3L-treated mice and placed them at limiting dilution in cultures of OP9 stroma plus IL-7 and Flt3L to measure B-cell potential, ST-2 stroma and Flt3L for myeloid and OP9-DL1 plus IL-7 and Flt3L to measure T-cell potential. In every experiment, the same cells were sorted from PBS-treated mice and cultured in parallel. In addition, CD19+NK1.1–B220+CD117low BM cells, which have a high plating efficiency for B-cell progenitors, were included.
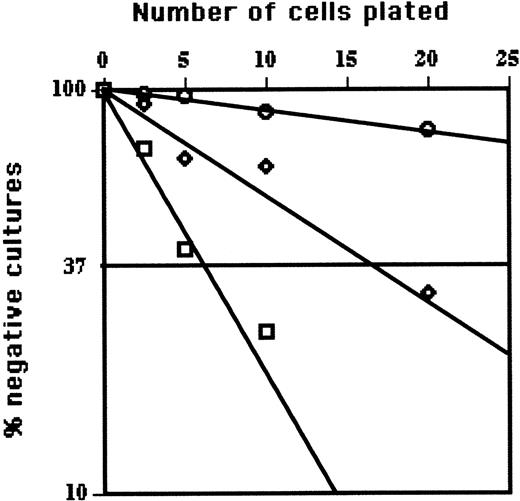
LDA of EPLM from Flt3L-treated mice. The figure summarizes the LDA for myeloid (□), T cell (⋄), and B cell (○) among EPLM from Flt3L-treated mice. Shown are the semilogarithmic plots of cell dose (horizontal linear scale) versus the percent negative cultures (vertical logarithmic scale) of 48 replicate cultures containing on average 2.5, 5, 10, or 20 sorted EPLM plated in 96-well microtiter plates containing irradiated ST2 stroma plus Flt3L for myeloid, OP9 plus IL-7 and Flt3L for B, or OP9-DL1 stroma plus IL-7 and Flt-3L for T cells. After 10 to 14 days, wells were scored for growth by microscopic examination. When 37% of replicate cultures are negative (horizontal line), according to Poisson statistics, each well would on average contain one precursor.
LDA of EPLM from Flt3L-treated mice. The figure summarizes the LDA for myeloid (□), T cell (⋄), and B cell (○) among EPLM from Flt3L-treated mice. Shown are the semilogarithmic plots of cell dose (horizontal linear scale) versus the percent negative cultures (vertical logarithmic scale) of 48 replicate cultures containing on average 2.5, 5, 10, or 20 sorted EPLM plated in 96-well microtiter plates containing irradiated ST2 stroma plus Flt3L for myeloid, OP9 plus IL-7 and Flt3L for B, or OP9-DL1 stroma plus IL-7 and Flt-3L for T cells. After 10 to 14 days, wells were scored for growth by microscopic examination. When 37% of replicate cultures are negative (horizontal line), according to Poisson statistics, each well would on average contain one precursor.
Whereas EPLM from normal mice generated myeloid, T, and B cells with a frequency of approximately 1 in 5 to 10, CD19–NK1.1–B220+CD117low BM cells from FLT3L-treated mice showed high myeloid- (1 in 5), intermediate T- (1 in about 18), and low B-lineage (1 in 180) potential (Figure 2). Thus, the most significant difference in developmental potential of CD19–NK1.1–B220+CD117low BM cells from Flt3L-treated mice was the reduction in B-cell progenitor activity. It is important to note that the presence of Flt3L in limiting dilution cultures influences neither the plating efficiency nor clone size of committed (CD19+B220+CD117low) BM cells used as controls (not shown).
Does Flt3L induce myeloid differentiation of EPLM in vivo?
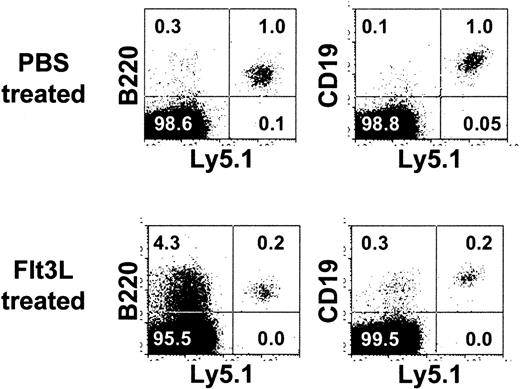
The in vitro finding that EPLM derived from Flt3L-treated mice had a bias in myeloid differentiation potential might suggest that Flt3L is myeloid instructive for EPLM. To test this possibility, Ly5.2.C57Bl/6.RAG-2–/– mice were treated daily for 3 days with either PBS or 4 μg Flt3L, irradiated and then injected intravenously with 104 EPLM derived from the BM of wild-type C57Bl/6.Ly5.1 mice. Injections of PBS or Flt3L were continued for a further 5 days prior to analysis. Results obtained (Figure 3) showed that in the 3 control recipients, the BM contained 0.7% ±0.5%, or 3 ± 2 × 104 donor-derived CD19+ cells, whereas in the Flt3L-treated mice, values were 0.4% ± 0.2%, or 7 ± 3 × 104 cells. Note that following irradiation, Flt3L treatment increased BM cellularity 4-fold. In addition, as seen by the increase in B220 expression among host (Ly5.1–) cells (lower left cytogram), the myeloid compartment was particularly affected. However, despite these changes, we failed to detect donor-derived myeloid cells in either BM (Figure 3) or spleen (data not shown). Therefore, increasing Flt3L availability in vivo does not seem to influence EPLM differentiation.
No effect of Flt3L treatment on EPLM injected in vivo. Shown are cytogram displays of BM from one of 3 PBS (top panels) or Flt3L-treated (bottom panels) Ly5.2.C57Bl/6.Rag2–/– mice injected 5 days previously with 104 EPLM from C57Bl/6.Ly5.1 mice stained for Ly5.2 (horizontal scale) and either B220 (left panels) or CD19 (right panels). Figures in each panel represent the percent positive cells.
No effect of Flt3L treatment on EPLM injected in vivo. Shown are cytogram displays of BM from one of 3 PBS (top panels) or Flt3L-treated (bottom panels) Ly5.2.C57Bl/6.Rag2–/– mice injected 5 days previously with 104 EPLM from C57Bl/6.Ly5.1 mice stained for Ly5.2 (horizontal scale) and either B220 (left panels) or CD19 (right panels). Figures in each panel represent the percent positive cells.
Thymus and T- and B-cell reconstitution. (A) Thymus reconstitution by EPLM from Flt3L-treated mice. A total of 2 × 104 EPLM from C57Bl/6CD45.1 mice were injected intravenously into C57Bl/6.Rag2–/–.Ly5.2 recipients. Three weeks after injection, the thymus contained 95% CD45.1+ cells (left histogram). The cytogram displays show the CD4 versus CD8 (middle panel) and CD4 versus TCR-β distribution on gated CD45.1+ cells. The figures in each panel indicate the percent cells in each quadrant. (B) EPLM reconstitute both T and B spleen cells. Shown are results of staining spleen cells from C57Bl/6.Rag2KO.Ly5.2 mice 7 weeks after reconstitution with 2 × 104 sorted EPLM from C57Bl/6.Ly5.1 Flt3L-treated mice. The spleen contained 11.1% donor derived CD45.1+ cells (top histogram) and cytogram displays show the CD4 versus CD8 (top left), CD3 versus TCR-β (top right), B220 versus IgM (lower left), and CD23 versus CD21 (bottom right) distribution on gated CD45.1+ cells. Figures in each panel represent the percent positive cells in each quadrant. In the lower right cytogram, the regions show the 19.1% CD23+CD21+ FB and 24.5% CD23–CD21+ MZB cells.
Thymus and T- and B-cell reconstitution. (A) Thymus reconstitution by EPLM from Flt3L-treated mice. A total of 2 × 104 EPLM from C57Bl/6CD45.1 mice were injected intravenously into C57Bl/6.Rag2–/–.Ly5.2 recipients. Three weeks after injection, the thymus contained 95% CD45.1+ cells (left histogram). The cytogram displays show the CD4 versus CD8 (middle panel) and CD4 versus TCR-β distribution on gated CD45.1+ cells. The figures in each panel indicate the percent cells in each quadrant. (B) EPLM reconstitute both T and B spleen cells. Shown are results of staining spleen cells from C57Bl/6.Rag2KO.Ly5.2 mice 7 weeks after reconstitution with 2 × 104 sorted EPLM from C57Bl/6.Ly5.1 Flt3L-treated mice. The spleen contained 11.1% donor derived CD45.1+ cells (top histogram) and cytogram displays show the CD4 versus CD8 (top left), CD3 versus TCR-β (top right), B220 versus IgM (lower left), and CD23 versus CD21 (bottom right) distribution on gated CD45.1+ cells. Figures in each panel represent the percent positive cells in each quadrant. In the lower right cytogram, the regions show the 19.1% CD23+CD21+ FB and 24.5% CD23–CD21+ MZB cells.
In vivo functional analysis of CD19–NK1.1–B220+CD117low cells from FLT3L-treated mice
Previously we showed that intravenous injection of 2 × 104 ex vivo sorted B220+CD117+CD19–NK1.1– BM cells from wild-type mice transiently reconstituted the T- and B-cell progenitor compartments and resulted in the formation of cohorts of long-lived peripheral B and T cells.1 To test the ability of EPLM from Flt3L-treated mice to reconstitute mice, we injected 2 × 104 ex vivo sorted B220+CD117+CD19–NK1.1– BM cells from FLT3L-treated C57Bl/6.Ly5.1 mice into C57Bl/6.Rag2KO.Ly5.2 recipients. We detected T- and B-cell reconstitution in all recipient mice. As previously reported,1 we did not detect myeloid reconstitution in vivo. The thymuses of recipients were analyzed between 3 and 4 weeks after reconstitution (Figure 4A left histogram) showing that the thymus was predominantly (95%) composed of donor (CD45.1+) cells. Gated CD45.1+ cells (right panels) contained all 4 CD4- and CD8-defined thymocyte subsets (middle panel), each expressing the appropriate levels of surface TCR-β (right panel). Likewise, the BM at this time contained CD45.1+ B cell progenitors (not shown).
At 7 weeks following reconstitution, whereas the thymus only contained either CD4+ or CD8+ single-positive cells (not shown), the spleen contained 11.1% CD45.1+ cells (Figure 4B upper histogram) and of these, 30% were CD4 and 21% CD8 (left middle cytogram) and TCR-β+ (right middle cytogram). The spleen also contained donor-derived mature B cells that were for the most part IgMhigh (left lower cytogram). As expected in situations where the B-cell compartment was not fully reconstituted,25 there was a relative enrichment in MZB, comprising 24.5% of gated B220+ cells (lower right cytogram). Thus, after 7 weeks, the spleens of reconstituted RAG-2–/– mice contained populations of mature T and B cells.
Immune responsiveness of reconstituted mice
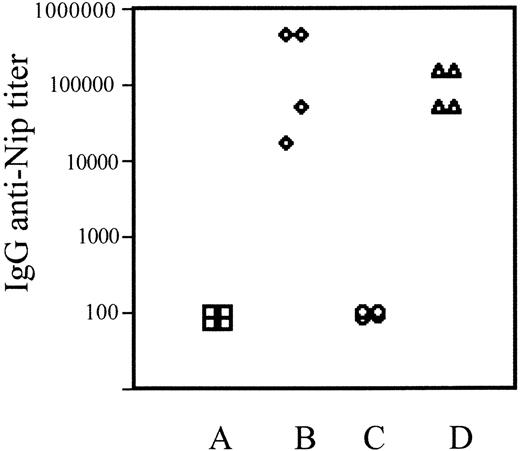
To assess the combined function of T- and B-cell compartments, groups of 4 reconstituted or control C57Bl/6 mice were immunized with the T cell–dependent antigen NIP-ovalbumin. The magnitude of the antigen-specific serum IgG antibody responses of individual mice measured by ELISA showed that reconstituted mice were as capable as controls in mounting a T cell–dependent B-cell response (Figure 5). This shows that for responsiveness to this prototype antigen, both T- and B-cell compartments of reconstituted mice were fully functional.
Discussion
Recently, we have described in the BM of normal adult mice a CD19–NK1.1–B220+CD117low cell, designated EPLM, which has B, T, and myeloid developmental potential.1 Furthermore, phenotypic analysis of EPLM indicated that they expressed CD135, the so-called Flt3 receptor. Therefore, we wished to see if increasing the in vivo availability of Flt3L, the corresponding ligand for Flt3, would alter the number or functional activity of EPLM.
Increasing Flt3L availability in vivo was achieved by administration of graded quantities of recombinant human Flt3L and resulted in a dose-dependent increase of BM lymphopoiesis, characterized by an approximately 50-fold increase in EPLM number (Tables 1 and 2) and an expected dramatic effect on DC generation. Unexpectedly, however, Flt3L treatment resulted in a concomitant dramatic reduction in B lymphopoiesis. The effects on both B and DC compartments were transient as shown by the rapid recovery to normal of BM and spleen cellularity and phenotype following cessation of Flt3L treatment. Functionally, when tested in LDA and compared with cells from controls, EPLM from mice treated with 10 μg Flt3L showed a dramatic (∼20-fold) reduction in frequency of B progenitors, a slight reduction (∼1.5-fold) in T progenitors, and slightly increased myeloid developmental potential. Combining the changes in EPLM cell recovery and precursor frequency, this amounts to an approximately 2.5-fold increase in total B progenitors, a 30-fold increase in T and a 50-fold increase in myeloid progenitors. The apparently high number of B-cell progenitors still present among EPLM from Flt3L-treated mice despite a massive decrease in in vivo B lymphopoiesis might seem paradoxical. However, it must be remembered that in vivo and in vitro microenvironments are completely different and, in addition, that the concentration (20 ng/mL) and bioavailability of Flt3L used for in vitro cultures probably differ significantly from those in vivo. The presence of Flt3L in the in vitro cultures did not alter the cloning efficiency or burst size of colonies. Taken together, it is clear that in vitro cultures can reveal B- and T-cell developmental potentials unavailable or not used in vivo.
T cell–dependent antibody responses in EPLM-reconstituted mice. Shown are the IgG anti-Nip–specific antibody titers from 4 individual control C57Bl/6 (A-B) or EPLM-reconstituted C57Bl/6.RAG-2KO mice (C-D) before (A,C) or 14 days after (B,D) immunization with NIP-ovalbumin.
T cell–dependent antibody responses in EPLM-reconstituted mice. Shown are the IgG anti-Nip–specific antibody titers from 4 individual control C57Bl/6 (A-B) or EPLM-reconstituted C57Bl/6.RAG-2KO mice (C-D) before (A,C) or 14 days after (B,D) immunization with NIP-ovalbumin.
The failure to reveal myeloid potential of EPLM following intravenous injection is rather surprising. By treating irradiated recipients with Flt3L injections and by analyzing mice soon after reconstitution, we tried to provide optimal conditions for revealing the myeloid potential of injected cells. However, despite showing an effect of Flt3L injections on the recovering hemopoietic system of the host (Figure 3), we were unable to detect EPLM-derived myeloid cells. One simple explanation for this is that despite being able to find donor-derived B cells in the BM, injected EPLM were unable to enter the correct microenvironment supporting myelopoiesis. Thus the apparent lineage potential of a cell is very much influenced by the assay used; this is particularly acute for myeloid cells.
These results raise the issue of how cytokine availability in vivo might alter the developmental potential of hemopoietic progenitors. The “instructive” role played by cytokines in guiding hemopoiesis was invoked many years ago.26,27 Progenitor cells frequently express extremely low numbers of cytokine receptor molecules,28 often below the reliable detection limits of flow cytometry. In this regard, bioresposiveness of cells is a far more sensitive assay than staining by FACS. One such example is the evident IL-7 responsiveness of the earliest population of CD117+, CD44+, CD25–, so-called DN1, progenitor thymocytes, which by flow cytometry appear IL-7Rα (CD127) negative.24 Interestingly, in the BM of IL-7 overexpressing transgenic mice,29 where B lymphopoiesis is massively increased, the proportion of CD19–NK1.1– EPLM among B220+CD117low cells is reduced and treatment of IL-7 transgenics with Flt3L did not alter this phenotype (not shown). When added to in vitro cultures or injected into recipient mice, increasing Flt3L availability did not discernibly alter the differentiation of EPLM. Taken together, these results suggest that Flt3L has little direct effect on EPLM, but rather acts on an upstream precursor. This would appear logical because Flt3L receptor (CD135) expression progressively decreases along the B-cell developmental pathway.2-4 By promoting development along a particular lineage, for example, IL-7 toward B or Flt3L toward myeloid, the cytokine milieu surrounding particular BM progenitors might significantly alter their cellular composition and developmental potential, thereby favoring the notion that cytokines can play an instructive role in lymphoid lineage commitment.27
Using in vitro experiments, we have shown that potent T-cell potential can be revealed among EPLM when they were cultured on OP9 stroma expressing the Notch ligand δ-like 1 (DL1) and that following prolonged Notch signaling, there was a dramatic up-regulation of c-kit expression and loss of B-lineage potential.1 In addition, using EPLM and Pax5–/– pre–B cell lines, we have recently shown that up-regulation of c-kit gene transcription and protein expression is a direct consequence of DL1 signalling.30 Thus, in addition to cytokines, Notch signaling is known to influence the T versus B, or lineage commitment status, of lymphoid progenitors from both mice31 and humans.32 However, in Flt3L-treated mice, we did not observe changes to the thymus T-cell progenitor compartment. In addition, we have not been able to find a thymocyte with the EPLM phenotype. Therefore, under physiologic conditions, we do not think that EPLM contribute to thymopoiesis. Rather, EPLM probably represent the last stage along a lymphoid developmental pathway at which a Notch signal can rescue T-cell commitment.
The most significant observation from these studies, however, is the increased number of BM EPLM induced by Flt3L. Freshly isolated EPLM from both control and Flt3L-treated mice could reconstitute both the T- and B-cell compartments of recipients, generating cohorts of functional B and T cells. Immune responses in EPLM-reconstituted mice showed the characteristic features of T/B cell collaboration. Taken together, these results may have considerable practical, perhaps clinical, applications in the context of BM transplantation.
Prepublished online as Blood First Edition Paper, May 4, 2006; DOI 10.1182/blood-2005-10-006643.
Supported by grants from the Swiss National Science Foundation (A.G.R.) and Contrat d'Interface INSERM, Etablissement Français du Sang (R.C.). A.G.R. is holder of the Chair in Immunology endowed by F. Hoffman-La Roche, Basel, Switzerland.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 U.S.C. section 1734.
We thank Amgen for a generous gift of human recombinant Flt3L. R.C. thanks INSERM for support.



This feature is available to Subscribers Only
Sign In or Create an Account Close Modal