Abstract
ALK1 is an endothelial-specific type I receptor of the TGFβ receptor family whose heterozygous mutations cause hereditary hemorrhagic telangiectasia type 2. Although TGFβ1 and TGFβ3 have been shown to bind ALK1 under specific experimental conditions, they may not represent the physiological ligands for this receptor. In the present study, we demonstrate that BMP9 induces the phosphorylation of Smad1/5/8 in microvascular endothelial cells, and this phosphorylation lasts over a period of 24 hours. BMP9 also activates the ID1 promoter–derived BMP response element (BRE) in a dose-dependent manner (EC50 = 45 ± 27 pg/mL), and this activation is abolished by silencing ALK1 expression or addition of ALK1 extracellular domain. Overexpression of endoglin increases the BMP9 response, whereas silencing of both BMPRII and ActRIIA expressions completely abolishes it. BMP10, which is structurally close to BMP9, is also a potent ALK1 ligand. Finally, we demonstrate that BMP9 and BMP10 potently inhibit endothelial cell migration and growth, and stimulate endothelial expression of a panel of genes that was previously reported to be activated by the constitutively active form of ALK1. Taken together, our results suggest that BMP9 and BMP10 are two specific ALK1 ligands that may physiologically trigger the effects of ALK1 on angiogenesis.
Introduction
Activin receptor-like kinase 1 (ALK1) is an endothelial-specific type I receptor of the TGFβ receptor family that is implicated in the pathogenesis of the Rendu-Osler disease also known as hereditary hemorrhagic telangiectasia (HHT).1 The disease is an autosomal dominant vascular dysplasia affecting 1 in 10 000 people. The clinical abnormalities in HHT are caused by direct arteriovenous connections without an intervening capillary bed. The resulting telangiectases occur in the oral cavity (lips and tongue), in the nose, and on the fingertips. Larger arteriovenous malformations (AVMs) can be encountered in the lung, brain, and liver.2 There is wide variation in the penetrance and severity of these symptoms in patients even within the same family, suggesting that environmental or other genetic factors influence the phenotype. The majority of cases are caused by mutations in either Endoglin (ENG) or ALK1 (ACVRL1) genes, thus defining HHT1 and HHT2, respectively. Recently, mutations in SMAD4 have also been described in a few cases with combined juvenile polyposis and HHT syndromes.3
Each of the 3 genes implicated in HHT (ENG, ACVRL1, and SMAD4) encode receptors or signaling molecules from the TGFβ family. Most TGFβ family ligands bind to heteromeric complexes of type I and type II serine/threonine kinase receptors (for review, see Shi and Massague4 ). In addition, the type III receptors (betaglycan and endoglin) act as coreceptors that can potentiate the signaling cascade. Upon ligand binding, the type II receptor phosphorylates and activates the type I receptor, also known as activin receptor-like kinase (ALK), which in turn phosphorylates a receptor-regulated Smad protein (Smad1, Smad2, Smad3, Smad5, or Smad8). This phosphorylated Smad dimerizes with a common partner, Smad4, and this complex translocates to the nucleus where it directly regulates the transcription of target genes. The inhibitory Smads, Smad6 and Smad7, can interrupt this signaling process (for review, see Derynck and Zhang5 ). ALK1 is one of the 7 type I receptors for TGFβ family members.6 It is predominantly expressed in endothelial cells. Activation of ALK1 induces phosphorylation of the receptor-regulated Smad1, Smad5, and Smad8.7
The specific ligand for ALK1 has yet to be identified, and the type II receptor(s) with which it interacts is not known. In experiments where type II TGFβ receptor (TβRII) or type II activin receptor (ActRIIA and ActRIIB) is overexpressed, ALK1 can bind TGFβ1 or activin, respectively.6,8–10 In nontransfected cells such as human umbilical vascular endothelial cells (HUVECs), results are still unclear. One study has demonstrated that antisera to ALK1 can immunoprecipitate endogenous ALK1/TβRII cross-linked complexes,11 while, in another study, these antibodies failed to immunoprecipitate TGFβ1 or TGFβ3 associated with receptor complexes.12 More recently, it was shown that TGFβ induced the phosphorylation of Smad2 and Smad1/5 in endothelial cells and that these effects could be blocked upon selective inhibition of ALK5 or ALK1, respectively.13 Research by the same group demonstrated that ALK5 was required for TGFβ signaling through ALK1.14 From these data, a mechanistic model has been proposed where TGFβ induces the formation of a heteromeric complex consisting of ALK5 and ALK1 and the phosphorylation of Smad1/5 in endothelial cells.14 However, this model is not supported by recent studies demonstrating that ALK1 and ALK5 present a nonoverlapping expression pattern in the vascular system, ALK1 being expressed in endothelial cells and ALK5 in vascular smooth muscle cells.15 In addition, a ligand for ALK1, distinct from TGFβ1 and TGFβ3 and present in the serum, has been described.16 Taken together, these data suggest that, although TGFβ1 and TGFβ3 may activate ALK1, they are not specific ligands, and that a more specific and still unidentified ALK1 ligand might exist. Very recently, a publication describing the crystal structure of BMP9 reported that BMP9 specifically binds biosensor-immobilized recombinant ALK1 and BMPRII extracellular domains and that ALK1 extracellular domain blocked BMP9-induced activation of alkaline phosphatase secretion by the mouse myoblast cell line C2C12.17 This prompted us to investigate whether BMP9 might be a ligand for ALK1 in endothelial cells.
In the present study, we demonstrate that BMP9 activates the Smad1/5/8 pathway in endothelial cells in a dose-dependent manner. This activation is specifically due to BMP9 binding to ALK1 as (1) this response was inhibited by siRNA-mediated down-regulation of ALK1, (2) only ALK1 overexpression increased the BMP9 response, and (3) ALK1 extracellular domain could specifically abolish the BMP9 response. Furthermore, we show that overexpression of the coreceptor endoglin potentiates the BMP9 response. We also determine, through the use of siRNAs, that BMP9 transduces its signal through either BMPRII or ActRIIA. We also observed that BMP10, which is the BMP family member with the highest homology to BMP9, is also a ligand for ALK1. Finally, we demonstrated that BMP9 and BMP10 are potent inhibitors of endothelial cell migration and growth.
Materials and methods
Approval was obtained from the Institut National de la Santé et de la Recherche Médicale (INSERM) institutional review board for these studies. Informed consent was provided in accordance with the Declaration of Helsinki.
Cell culture
Human dermal microvascular endothelial cells (HMVEC-d's; Cambrex, Verviers, Belgium) were maintained in endothelial growth medium (EGM-2-MV; Cambrex) supplemented with 5% fetal bovine serum (FBS; Cambrex). H5V endothelial cells (a generous gift from Dr Vecchi, Istituto Mario Negri, Milan, Italy) and NIH-3T3 fibroblasts were maintained in DMEM, 4.5 g/L glucose (Invitrogen, Cergy Pontoise, France) supplemented with 10% FBS (Biowest; Abcys, Paris, France).
Reagents
Recombinant human BMP9, recombinant human TGFβ1, recombinant human ALK1 extracellular domain/Fc (ALK1ecd), and human ALK3 extracellular domain/Fc (ALK3ecd) were purchased from R&D Systems (Abingdon, United Kingdom). Recombinant human BMP2 was purchased from Abcys.
Western blot analysis
HMVEC-d cells were stimulated with BMP9 at the doses indicated for 30 minutes up to 24 hours. Cells were then washed twice with PBS and lysed in radioimmunoprecipitation assay (RIPA) buffer. Proteins in the lysates were separated by sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS/PAGE, 10%) and analyzed by immunoblotting with polyclonal rabbit phospho-Smad1/5/8 (a generous gift from Dr C. H. Heldin, Ludwig Institute for Cancer Research, Uppsala, Sweden). The same membrane was stripped and reprobed with a monoclonal antibody against αtubulin given to us by Dr D. Job (INSERM U366, CEA-G, Grenoble, France) to confirm equal protein loading. ALK1 was visualized by Western blot using an anti-ALK1 antibody (a generous gift from Dr D. Marchuk, Duke University Medical Center, Durham, NC) after immunoprecipitation of 500 μg total proteins with 1 μg anti–human ALK1 antibody (AF370; R&D Systems).
Reporter gene constructs and expression plasmids
The reporter plasmid pGL3(BRE)-luc encoding firefly luciferase was kindly provided by Dr P. ten Dijke (Leiden University Medical Center, Leiden, the Netherlands). The pGL3(CAGA)12-luc encoding firefly luciferase and the pcDNA3 expression plasmids encoding human ALK1, ALK2, ALK3, ALK6, and Endoglin were kindly provided by Dr C. H. Heldin (Ludwig Institute for Cancer Research). The pRL-TK-luc plasmid encoding renilla luciferase was purchased from Promega (Madison, WI).
DNA transfection and dual luciferase activity assay
HMVEC-d's (60 000), H5V cells (50 000), and NIH-3T3 cells (70 000) were transfected in Opti-MEM (Invitrogen) using lipofectamine (Invitrogen) with 0.1 μg pGL3(BRE)-luc or 0.2 μg pGL3(CAGA)12-luc, 0.02 μg pRL-TK-luc, and 0.05 μg either pcDNA3 empty vector, pALK1, pALK2, pALK3, pALK6, or pEndoglin. Four hours after transfection, cells were treated with the appropriate ligand (BMP9, BMP2, BMP10, or TGFβ1) at different concentrations with or without ALK1ecd or ALK3ecd (6-fold molar excess over respective BMP9, BMP2, or BMP10 concentrations) for 15 hours. Firefly and renilla luciferase activities were measured sequentially with the Dual-Luciferase reporter assay (Promega). Results are expressed as ratios of firefly luciferase activity over renilla luciferase activity.
RNA interference
HMVEC-d's (50 000 cells) were transfected in Opti-MEM using lipofectamine (Invitrogen) with a mix of 4 siRNAs directed against human ALK1 obtained using the Silencer siRNA construction kit (Ambion, Austin, TX; final concentration: 200 nM; target sequences: 5′-AACTTGCACAGGGAGCTCTGC-3′, 5′-AACCACAACGTGTCCCTGGTG-3′, 5′-AACCACTACTGCTGCGACAGC-3′, and 5′-AAAGCATCTGAGCAGGGCGAC-3′) or Control 1 siRNA (Ambion, Huntingdon, United Kingdom) together with 0.1 μg pGL3(BRE)-luc and 0.02 μg pRL-TK-luc. H5V (50 000 cells) and NIH-3T3 (70 000 cells) were transfected in Opti-MEM using oligofectamine (Invitrogen) with a mix of 4 siRNAs (200 nM each) directed against either mouse ALK1 (M-043004), mouse BMPRII (M-040599), or mouse ActRIIA (M-040676), obtained from Dharmacon (Chicago, IL), or with control 1 siRNA (Ambion, Huntingdon, United Kingdom) for 24 hours alone or in combinations. Twenty-four hours later, the cells were transfected with pALK1 (0.05 μg in NIH-3T3 or 0.01 μg in H5V), 0.1 μg pGL3(BRE)-luc, and 0.02 μg pRL-TK-luc. Four hours after transfection, cells were treated for 15 hours with BMP9 or BMP2, at the indicated concentrations. Luciferase activity was determined as described in “DNA transfection and dual luciferase activity assay.”
RNA extraction and reverse transcription
Total RNA was extracted using RNeasy columns (Qiagen, Courtaboeuf, France). First-strand cDNAs were generated using 1 μg RNA, 1 μL Improm-II reverse transcriptase (Promega), and random hexamer primers (Amersham Pharmacia Biotech, Piscataway, NJ).
Semiquantitative reverse transcriptase–polymerase chain reaction (RT-PCR)
cDNAs were amplified with 1 U Taq DNA polymerase (Qiagen) in a final volume of 25 μL with 10 pmol of each primer (Table 1). After 5 minutes of initial denaturation at 94°C, reactions were cycled through 1 minute at 94°C, 1 minute annealing at primer-specific temperature (Table 1), and 1 minute primer extension at 72°C. Final extension was carried out for 5 minutes at 72°C. To ensure quantitative results, the number of PCR cycles for each set of primers was checked to be in the linear range of amplification. In addition, all cDNA samples were adjusted to yield equal amplification of hypoxanthine phosphoribosyltransferase (HPRT) as an internal standard.
Oligonucleotide primers used for RT-PCR analysis
| Genes . | Primer sequences, 5′-3′ . | Annealing temp, °C . | Cycle . | Size, bp* . | |
|---|---|---|---|---|---|
| Forward . | Reverse . | ||||
| Hprt | GCTGGTGAAAAGGACCTCT | CACAGGACTAGAACACCTGC | 59 | 28 or QPCR | 280 |
| Alk1 | TGCCTGTTCAGATGCCTTCAG | CTTGGGGAGCTTCAGAAGGGG | 59 | QPCR | 502 |
| Bmpr2 | TGGCAGTGAGGTCACTCAAG | TTGCGTTCATTCTGCATAGC | 53 | QPCR | 178 |
| Acvr2a | AGCAAGGGGAAGATTTGGTT | GGTGCCTCTTTTCTCTGCAC | 53 | QPCR | 168 |
| Acvr2b | CTGTGCGACTCCTTTAAGC | TCTTCACAGCCACAAAGTCG | 54 | QPCR | 174 |
| HPRT | TGCGACCTTGACCATCTTTGG | GCCATCACATTGTAGCCCTCT | 56 | QPCR | 358 |
| ID1 | ATCAGGGACCTTCAGTTGGAGC | AGACCCACAGAGCACGTAATTCC | 60 | QPCR | 236 |
| ID2 | TCCCAGGGTGTTCTCTTACTTGGACT | GGATCCTTCTGGTATTCACGCTCCA | 54 | QPCR | 200 |
| GDF15 | ACGGGAGGTGCAAGTGACCATGTG | TGAGCACCATGGGATTGTTAGC | 61 | QPCR | 158 |
| BMPRII | GCTAAAATTTGGCAGCAAGC | CTTGGGCCCTATGTGTCACT | 57 | QPCR | 224 |
| ENG | CACTAGCCAGGTCTCGAAGG | CTGAGGACCAGAAGCACCTC | 57 | QPCR | 165 |
| SMAD6 | AGGAGAAACTCGCTCCAAGTGCAT | AGCTCCCTTCTTACTCCCTGCAAA | 54 | QPCR | 152 |
| SMAD7 | TCGGACAGCTCAATTCGGACAACA | AACACCTTGTGTACCAACAGCGTC | 54 | QPCR | 190 |
| ALK1 | CCTTGCTGGCCCTGGTGGCCCT | GTGGGCAATGGCTGGTTTG | 63 | 30 | 607 |
| ALK2 | GAAGGGCTCATCACCACCAAT | GAACGGTGGCTTGTAATCCTC | 56 | 26 | 771 |
| ALK3 | GCACATTGCTTTGCCATCATA | CATTTGCCCATCCATACTTCT | 60 | 24 | 512 |
| ALK6 | ACACCACAGGGCTTTACTTAT | AATTGCTGGTTTGCCTTGAGT | 56 | 30 | 616 |
| Genes . | Primer sequences, 5′-3′ . | Annealing temp, °C . | Cycle . | Size, bp* . | |
|---|---|---|---|---|---|
| Forward . | Reverse . | ||||
| Hprt | GCTGGTGAAAAGGACCTCT | CACAGGACTAGAACACCTGC | 59 | 28 or QPCR | 280 |
| Alk1 | TGCCTGTTCAGATGCCTTCAG | CTTGGGGAGCTTCAGAAGGGG | 59 | QPCR | 502 |
| Bmpr2 | TGGCAGTGAGGTCACTCAAG | TTGCGTTCATTCTGCATAGC | 53 | QPCR | 178 |
| Acvr2a | AGCAAGGGGAAGATTTGGTT | GGTGCCTCTTTTCTCTGCAC | 53 | QPCR | 168 |
| Acvr2b | CTGTGCGACTCCTTTAAGC | TCTTCACAGCCACAAAGTCG | 54 | QPCR | 174 |
| HPRT | TGCGACCTTGACCATCTTTGG | GCCATCACATTGTAGCCCTCT | 56 | QPCR | 358 |
| ID1 | ATCAGGGACCTTCAGTTGGAGC | AGACCCACAGAGCACGTAATTCC | 60 | QPCR | 236 |
| ID2 | TCCCAGGGTGTTCTCTTACTTGGACT | GGATCCTTCTGGTATTCACGCTCCA | 54 | QPCR | 200 |
| GDF15 | ACGGGAGGTGCAAGTGACCATGTG | TGAGCACCATGGGATTGTTAGC | 61 | QPCR | 158 |
| BMPRII | GCTAAAATTTGGCAGCAAGC | CTTGGGCCCTATGTGTCACT | 57 | QPCR | 224 |
| ENG | CACTAGCCAGGTCTCGAAGG | CTGAGGACCAGAAGCACCTC | 57 | QPCR | 165 |
| SMAD6 | AGGAGAAACTCGCTCCAAGTGCAT | AGCTCCCTTCTTACTCCCTGCAAA | 54 | QPCR | 152 |
| SMAD7 | TCGGACAGCTCAATTCGGACAACA | AACACCTTGTGTACCAACAGCGTC | 54 | QPCR | 190 |
| ALK1 | CCTTGCTGGCCCTGGTGGCCCT | GTGGGCAATGGCTGGTTTG | 63 | 30 | 607 |
| ALK2 | GAAGGGCTCATCACCACCAAT | GAACGGTGGCTTGTAATCCTC | 56 | 26 | 771 |
| ALK3 | GCACATTGCTTTGCCATCATA | CATTTGCCCATCCATACTTCT | 60 | 24 | 512 |
| ALK6 | ACACCACAGGGCTTTACTTAT | AATTGCTGGTTTGCCTTGAGT | 56 | 30 | 616 |
Size of the amplified cDNA.
Quantitative real-time PCR
Quantitative PCR reactions were performed for ALK1, BMPRII, ActRIIA, ActRIIB, GDF15, SMAD6, SMAD7, ID1, ID2, ENG, HPRT, HPRT, and 18S (crosses with mouse and human; Table 1) on a Light Cycler apparatus using SYBR green PCR core reagents (Roche Diagnostics, Mannheim, Germany). Quantitative RT-PCR for human ALK1 was performed using TaqMan gene expression assay and ALK1 primers from Applied Biosystems (Foster City, CA), using Light Cycler FastStart DNA master hybridization probes (Roche Diagnostics). Results are expressed as the ratio of the studied mRNA levels over HPRT or 18S RNA levels.
Cell migration in the wound assay
Confluent monolayers of HMVEC-d's were wounded with a plastic pipette tip in the presence or the absence of BMP9 or BMP10, with or without ALK1ecd (15-fold molar excess over BMP9 concentration), placed back at 37°C in the incubator, and photographed at indicated times (0, 24, and 48 hours). Quantitation of monolayer closure was performed using the NIH Image J program (NIH, Bethesda, MD). Results are expressed as percent of wound closure.
Cell number
Cells were seeded in 96-well plates for 24 hours in the presence or the absence of BMP9 or BMP10 with or without ALK1ecd (15-fold molar excess over BMP9 concentration). The number of viable cells was determined using the WST-1 colorimetric assay (Roche Diagnostics) 24 hours or 48 hours later.
Statistics
Statistical analysis was performed using a Student t test from the software SigmaStat (Systat Software, Point Richmond, CA) (**P < .01; *P < .05). Fitting of the dose response curve and EC50 calculation were performed using the program MMfit (Michaelis-Menten fitting) of simfit suit (http://www.simfit.man.ac.uk/default.htm; University of Manchester).
Results
BMP9 activates the Smad1/5/8 pathway in HMVEC-d's
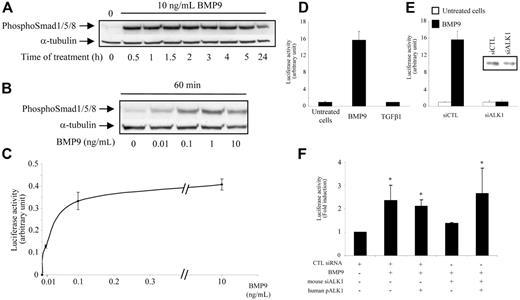
We first studied whether BMP9 could phosphorylate the Smad1/5/8 pathway in HMVEC-d's by immunoblotting analysis using specific antiphosphosite antibodies. As shown in Figure 1A, 10 ng/mL BMP9 rapidly induced the phosphorylation of Smad1/5/8 by 30 minutes, and this phosphorylation lasted for up to 24 hours. BMP9 was a very potent activator of Smad1/5/8 phosphorylation as this activation was observed with concentrations as low as 10 pg/mL and reached a plateau at 1 ng/mL (Figure 1B). The BRE promoter construct contains a repeated sequence of the Id1 promoter that is activated by several BMPs acting through the Smad1/5/8 pathway.18 We therefore used this promoter in order to confirm that BMP9 could induce the Smad1/5/8 pathway in endothelial cells. Our results demonstrated that BMP9 treatment of HMVEC-d's stimulated the luciferase reporter activity under the control of the BRE promoter (Figure 1C). This activation was concentration-dependent with an EC50 of around 45 ± 27 pg/mL (2 pM). We also examined whether BMP9 could activate the CAGA promoter, which is known to be specifically activated by the Smad2/3 pathway. We found that BMP9 was ineffective in activating the CAGA promoter, while TGFβ1, a known activator in this context, strongly activated CAGA (data not shown). Conversely, TGFβ1 did not activate the BRE promoter in HMVEC-d's under our experimental conditions (15 hours of treatment, Figure 1D), suggesting that it does not activate ALK1. We then checked whether the BMP9 response involves ALK1 by decreasing endogenous ALK1 gene expression using ALK1-specific siRNA. We observed that silencing of ALK1 (75% inhibition of mRNA levels as determined by quantitative RT-PCR and 50% inhibition at the protein level [insert in Figure 1E]) completely abolished the BMP9 response of the BRE promoter in HMVEC-d's (Figure 1E). We could also reproduce this effect in the mouse endothelial cell line H5V using mouse siRNA directed against ALK1 (60% inhibition of mRNA levels as determined by quantitative RT-PCR) as shown in Figure 1F. Further, this inhibitory effect could be rescued by ectopic expression of human ALK1, demonstrating the specificity of the inhibition (Figure 1F).
BMP9 activates the Smad1/5/8 pathway in HMVEC-d's. (A-B) HMVEC-d's were treated with BMP9 for different times and at different concentrations. Cell lysates (20 μg proteins) were resolved on a 10% SDS–polyacrylamide gel, and immunoblotted with antibodies against phospho-Smad1/5/8 or against α-tubulin. (C-D) HMVEC-d's were transiently transfected with pGL3(BRE)-luc and pRL-TK-luc. After 4 hours, cells were treated with different concentrations of BMP9 (0.01, 0.1, 1, or 10 ng/mL) in panel C and BMP9 (1 ng/mL) or TGFβ1 (1 ng/mL) in panel D for 15 hours. (E) HMVEC-d cells were transiently transfected with hALK1 siRNAs or control (CTL) siRNA together with pGL3(BRE)-luc and pRL-TK-luc. After 4 hours, cells were treated with BMP9 (1 ng/mL) for 15 hours. The luciferase activities were then measured as described in “Materials and methods.” The relative firefly luciferase activity was normalized to renilla luciferase activity. Data shown in panels C-E are representative of 1 experiment ± SD of 3. Inset in panel E represents ALK1 protein level revealed by Western blotting with an anti-ALK1 antibody after immunoprecipitation with another antibody directed against ALK1 of 500 μg total protein obtained from cells treated 48 hours with control siRNA or ALK1 siRNA. (F) H5V cells were transiently transfected with mALK1 siRNAs or CTL siRNA together with pGL3(BRE)-luc or pRL-TK-luc, with or without human pALK1. After 4 hours, cells were treated with BMP9 (0.1 ng/mL) for 15 hours. The firefly luciferase activity was normalized to renilla luciferase activity. Results are the mean ± SE of 3 experiments; *P < .05.
BMP9 activates the Smad1/5/8 pathway in HMVEC-d's. (A-B) HMVEC-d's were treated with BMP9 for different times and at different concentrations. Cell lysates (20 μg proteins) were resolved on a 10% SDS–polyacrylamide gel, and immunoblotted with antibodies against phospho-Smad1/5/8 or against α-tubulin. (C-D) HMVEC-d's were transiently transfected with pGL3(BRE)-luc and pRL-TK-luc. After 4 hours, cells were treated with different concentrations of BMP9 (0.01, 0.1, 1, or 10 ng/mL) in panel C and BMP9 (1 ng/mL) or TGFβ1 (1 ng/mL) in panel D for 15 hours. (E) HMVEC-d cells were transiently transfected with hALK1 siRNAs or control (CTL) siRNA together with pGL3(BRE)-luc and pRL-TK-luc. After 4 hours, cells were treated with BMP9 (1 ng/mL) for 15 hours. The luciferase activities were then measured as described in “Materials and methods.” The relative firefly luciferase activity was normalized to renilla luciferase activity. Data shown in panels C-E are representative of 1 experiment ± SD of 3. Inset in panel E represents ALK1 protein level revealed by Western blotting with an anti-ALK1 antibody after immunoprecipitation with another antibody directed against ALK1 of 500 μg total protein obtained from cells treated 48 hours with control siRNA or ALK1 siRNA. (F) H5V cells were transiently transfected with mALK1 siRNAs or CTL siRNA together with pGL3(BRE)-luc or pRL-TK-luc, with or without human pALK1. After 4 hours, cells were treated with BMP9 (0.1 ng/mL) for 15 hours. The firefly luciferase activity was normalized to renilla luciferase activity. Results are the mean ± SE of 3 experiments; *P < .05.
BMP9 activation of the BRE promoter is specific for ALK1
In order to further determine whether the observed activation was specific for ALK1, we next studied the effect of BMP9 in a cell line in which ALK1 is barely detectable by RT-PCR (mouse NIH-3T3 fibroblasts), and compared the level of activation between control cells and cells where human ALK1 was overexpressed (as assessed by the RT-PCR analysis shown in Figure 2B). BMP2, a known ligand for ALK3 or ALK6, was used as a control.4 Figure 2A shows that BMP9 treatment (0.5 ng/mL) increased BRE activity in NIH-3T3 cells and that this response was increased when ALK1 was overexpressed. In contrast, the BMP2 response (50 ng/mL) was not increased by ALK1 overexpression. We then studied whether other type I receptors could mediate the BMP9 response. To this end, we individually overexpressed the other type I receptors (Figure 2B) known to signal through the Smad1/5/8 pathway and measured the BMP9 response. As shown in Figure 2A, ALK2 expression did not modify the BMP9 response, while ALK3 or ALK6 expression increased it only slightly. On the other hand, the BMP2 response was increased by ALK3 or ALK6 overexpression but not by ALK2 expression. To confirm BMP9 specificity for ALK1, we tested the ability of the recombinant extracellular domain of ALK1 fused to the Fc region of human IgG1 (ALK1ecd) or ALK3ecd to interfere with the BMP9 and BMP2 responses. In this experiment, BMP9 (0.5 ng/mL) or BMP2 (50 ng/mL) was added to NIH-3T3 cells, alone or in the presence of a 6-fold molar excess of ALK1ecd or ALK3ecd. As shown in Figure 2C, addition of ALK1ecd completely inhibited the BMP9 response but had no effect on the BMP2 response. Conversely, addition of ALK3ecd inhibited the BMP2 response but had no effect on the BMP9 response (Figure 2C).
BMP9 activation of the BRE promoter is specific for ALK1. (A) NIH-3T3 cells were transiently transfected with pGL3(BRE)-luc and pRL-TK-luc and either pCDNA3 empty vector, pALK1, pALK2, pALK3, or pALK6 plasmids. After 4 hours, cells were treated with BMP9 (0.5 ng/mL) or BMP2 (50 ng/mL) for 15 hours. (B) Overexpression of the different human ALK receptor transcripts in transfected cells were controlled using semiquantitative RT-PCR. mHPRT was amplified to normalize for the amount of RNA used as starting material. (C) NIH-3T3 cells were transiently transfected with pGL3(BRE)2-luc and pRL-TK-luc. After 4 hours, cells were treated with BMP9 (0.5 ng/mL) or BMP2 (50 ng/mL) in the presence or absence of ALK1ecd or ALK3ecd (6-fold molar excess over respective BMP9 or BMP2 concentrations) for 15 hours. The luciferase activities were then measured as described in “Materials and methods.” The firefly luciferase activity was normalized to renilla luciferase activity. (A-C) Data show 1 representative experiment ± SD of 3.
BMP9 activation of the BRE promoter is specific for ALK1. (A) NIH-3T3 cells were transiently transfected with pGL3(BRE)-luc and pRL-TK-luc and either pCDNA3 empty vector, pALK1, pALK2, pALK3, or pALK6 plasmids. After 4 hours, cells were treated with BMP9 (0.5 ng/mL) or BMP2 (50 ng/mL) for 15 hours. (B) Overexpression of the different human ALK receptor transcripts in transfected cells were controlled using semiquantitative RT-PCR. mHPRT was amplified to normalize for the amount of RNA used as starting material. (C) NIH-3T3 cells were transiently transfected with pGL3(BRE)2-luc and pRL-TK-luc. After 4 hours, cells were treated with BMP9 (0.5 ng/mL) or BMP2 (50 ng/mL) in the presence or absence of ALK1ecd or ALK3ecd (6-fold molar excess over respective BMP9 or BMP2 concentrations) for 15 hours. The luciferase activities were then measured as described in “Materials and methods.” The firefly luciferase activity was normalized to renilla luciferase activity. (A-C) Data show 1 representative experiment ± SD of 3.
siRNA silencing of both BMPRII and ActRIIA abolishes the BMP9 response
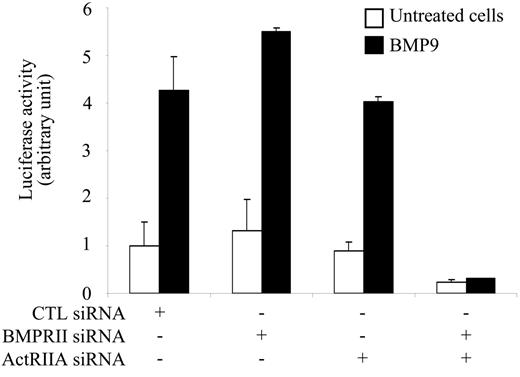
Having demonstrated that BMP9 is a specific ligand for ALK1, we then looked for the type II receptor implicated in this response. We first overexpressed the type II receptors known to be involved in the Smad1/5/8 pathway. We observed that BMPRII, ActRIIA, or ActRIIB overexpression together with ALK1 overexpression did not increase the BMP9 responsiveness of the BRE promoter (data not shown). We therefore tried an alternative strategy involving silencing of the type II receptors. Quantitative RT-PCR indicated that BMPRII (700 000 ± 140 000 copies/10 000 cells) and ActRIIA (500 000 ± 140 000 copies/10 000 cells) were expressed at similar levels in NIH-3T3 cells, while ActRIIB was expressed at a much lower level (20 000 ± 8000 copies/10 000 cells; ie, around 30 times less). Therefore, we choose to suppress only BMPRII and ActRIIA gene expression using specific siRNAs in NIH-3T3 cells transfected with pALK1. As shown in Figure 3, silencing of either BMPRII (70% inhibition of mRNA levels determined by quantitative RT-PCR) or ActRIIA (60% inhibition of mRNA levels) alone had no effect on the BMP9 response. However, silencing of both BMPRII and ActRIIA gene expression completely abolished the BMP9 response. These data demonstrate that both BMPRII and ActRIIA can cooperate with ALK1 to transduce the BMP9 response.
siRNA silencing of both BMPRII and ActRIIA abolishes the BMP9 response. NIH-3T3 cells were transiently transfected with control, BMPRII, or ActRIIA siRNAs. After 24 hours, NIH-3T3 cells were then transiently transfected with pGL3(BRE)-luc, pRL-TK-luc, and pALK1. After 4 hours, cells were treated with BMP9 (0.5 ng/mL) for 15 hours. The luciferase activities were then measured as described in “Materials and methods.” The firefly luciferase activity was normalized to renilla luciferase activity. Data show 1 representative experiment ± SD of 3.
siRNA silencing of both BMPRII and ActRIIA abolishes the BMP9 response. NIH-3T3 cells were transiently transfected with control, BMPRII, or ActRIIA siRNAs. After 24 hours, NIH-3T3 cells were then transiently transfected with pGL3(BRE)-luc, pRL-TK-luc, and pALK1. After 4 hours, cells were treated with BMP9 (0.5 ng/mL) for 15 hours. The luciferase activities were then measured as described in “Materials and methods.” The firefly luciferase activity was normalized to renilla luciferase activity. Data show 1 representative experiment ± SD of 3.
Endoglin overexpression increases the BMP9 response
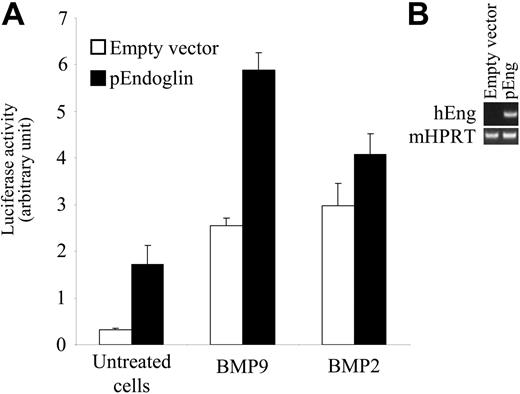
Endoglin is a coreceptor for ligands of the TGFβ family. Endoglin mutations have been shown to cause HHT1. Endoglin has also been shown to be present within the ALK1 signaling complex in endothelial cells and to increase the ALK1 response to TGFβ1.19,20 We therefore studied whether endoglin could also increase the BMP9 response. We overexpressed endoglin in NIH-3T3 cells (as assessed by the RT-PCR analysis shown in Figure 4B), where it is not normally expressed, together with ALK1 and then measured the BMP9 response. In order to detect an increase in BMP9 activation of ALK1, we used a low concentration of BMP9 (0.03 ng/mL) corresponding approximately to its EC50. As shown in Figure 4A, endoglin overexpression increased BMP9-induced BRE promoter activity in the presence of ALK1. On the other hand, endoglin overexpression had no significant effect on the BMP2 response in similarly transfected cells.
Endoglin overexpression increases the BMP9 response. (A) NIH-3T3 cells were transiently transfected with pGL3(BRE)-luc, pRL-TK-luc, pALK1, and either pCDNA3 empty vector or pEndoglin. After 4 hours, cells were treated with BMP9 (0.03 ng/mL) or BMP2 (10 ng/mL) for 15 hours. The firefly luciferase activity was normalized to renilla luciferase activity. Results are presented as mean values ± SD. (B) Overexpression of human endoglin transcripts in transfected cells was controlled using semiquantitative PCR. mHPRT was amplified to normalize for the amount of RNA used as starting material.
Endoglin overexpression increases the BMP9 response. (A) NIH-3T3 cells were transiently transfected with pGL3(BRE)-luc, pRL-TK-luc, pALK1, and either pCDNA3 empty vector or pEndoglin. After 4 hours, cells were treated with BMP9 (0.03 ng/mL) or BMP2 (10 ng/mL) for 15 hours. The firefly luciferase activity was normalized to renilla luciferase activity. Results are presented as mean values ± SD. (B) Overexpression of human endoglin transcripts in transfected cells was controlled using semiquantitative PCR. mHPRT was amplified to normalize for the amount of RNA used as starting material.
BMP9 inhibits HMVEC-d cell migration and growth
Since we have previously demonstrated that ALK1ca expression inhibits migration and proliferation of endothelial cells,21 we next tested the effects of BMP9 on these 2 processes. We found that BMP9 (10 ng/mL) inhibited HMVEC-d migration in the wound closure assay (Figure 5A). We further observed that the addition of ALK1ecd (15-fold molar excess) abolished BMP9-induced inhibition of wound closure, suggesting that BMP9-induced inhibition of endothelial cell migration is specifically mediated by ALK1. We also studied the effect of BMP9 treatment on endothelial cell growth. As seen in Figure 5B, BMP9 addition (10 ng/mL) inhibited HMVEC-d cell growth and ALK1ecd addition inhibited BMP9-induced inhibition of cell growth.
BMP9 inhibits HMVEC-d cell migration and cell growth, and induces GDF15 gene expression. (A) HMVEC-d monolayers were scratched to create a wound and incubated in low serum concentration (0.5% FBS) in the presence or absence of BMP9 (10 ng/mL) alone or together with ALK1ecd (15-fold molar excess over BMP9 concentration). At time 0, 24, and 48 hours after wounding, the cells were observed by phase-contrast microscopy and photographed. Results from 1 representative experiment of 5 are presented as the percent of wound closure at different times after wounding ± SE. (B) HMVEC-d's were treated for 24 hours and 48 hours with BMP9 (10 ng/mL) alone or together with ALK1ecd (15-fold molar excess over BMP9 concentration) in 5% FBS. The quantity of viable cells was determined using the WST-1 assay. Results from 1 representative experiment of 5 are expressed as absorbance (OD450) ± SE. (C) HMVEC-d's were treated for 24 hours in the presence or absence of BMP9 (10 ng/mL) alone or together with ALK1ecd (15-fold molar excess over BMP9 concentration). mRNAs were then extracted and quantitative RT-PCR was performed for GDF15 and HPRT. Results are expressed as level of GDF15/level of HPRT gene expression. Data show 1 representative experiment ± SD of 3. (A-B)**P < .01.
BMP9 inhibits HMVEC-d cell migration and cell growth, and induces GDF15 gene expression. (A) HMVEC-d monolayers were scratched to create a wound and incubated in low serum concentration (0.5% FBS) in the presence or absence of BMP9 (10 ng/mL) alone or together with ALK1ecd (15-fold molar excess over BMP9 concentration). At time 0, 24, and 48 hours after wounding, the cells were observed by phase-contrast microscopy and photographed. Results from 1 representative experiment of 5 are presented as the percent of wound closure at different times after wounding ± SE. (B) HMVEC-d's were treated for 24 hours and 48 hours with BMP9 (10 ng/mL) alone or together with ALK1ecd (15-fold molar excess over BMP9 concentration) in 5% FBS. The quantity of viable cells was determined using the WST-1 assay. Results from 1 representative experiment of 5 are expressed as absorbance (OD450) ± SE. (C) HMVEC-d's were treated for 24 hours in the presence or absence of BMP9 (10 ng/mL) alone or together with ALK1ecd (15-fold molar excess over BMP9 concentration). mRNAs were then extracted and quantitative RT-PCR was performed for GDF15 and HPRT. Results are expressed as level of GDF15/level of HPRT gene expression. Data show 1 representative experiment ± SD of 3. (A-B)**P < .01.
BMP10 is also an activator of ALK1
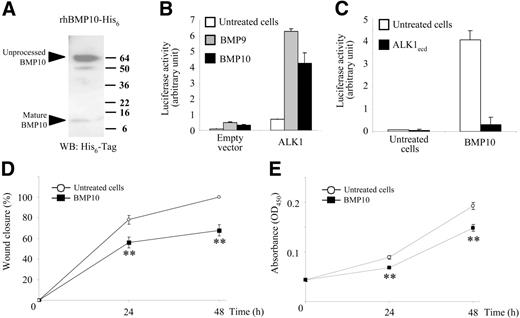
Based on protein sequence comparisons, BMP10 is the BMP that is the most closely related to BMP9 (65% amino acid identity). We therefore hypothesized that BMP10, like BMP9, might be a ligand for ALK1. BMP10 was purified from supernatants of human 293T cells transfected with an expression vector containing the BMP10 cDNA tagged with His6 as previously described.22 Two distinct bands were found under reducing conditions, BMP10 migrated as a 12-kDa band representing the monomeric active form and a 60-kDa band corresponding to the unprocessed monomer (Figure 6A). The active mature form of BMP10 represented about 10% of recombinant BMP10 and, therefore, we added higher doses of BMP10 than BMP9. Similarly to BMP9, BMP10 (10 ng/mL) increased BRE promoter luciferase activity in NIH-3T3 cells and this response was further increased when ALK1 was overexpressed (Figure 6B). Further, the BMP10 response could be completely abolished by addition of ALK1ecd (Figure 6C). We then studied the effect of BMP10 treatment on HMVEC-d cell migration and growth. We found that, like for BMP9, BMP10 treatment (100 ng/mL) inhibited HMVEC-d cell migration (Figure 6D) and growth (Figure 6E).
BMP10 is also an activator of ALK1. (A) BMP10 with an N-terminal His6 tag was purified from conditioned medium of 293T cells using metal chelate chromatography. BMP10 was electrophoresed on polyacrylamide gels under reducing conditions and detected by immunoblotting using antibodies against polyhistidine (His6). (B-C) NIH-3T3 cells were transiently transfected with pGL3(BRE)-luc, pRL-TK-luc, and either pCDNA3 empty vector or pALK1. After 4 hours, cells were treated with BMP9 (0.1 ng/mL) or BMP10 (10 ng/mL) in the presence or absence of ALK1ecd (15-fold molar excess over BMP10 concentrations) for 15 hours. The firefly luciferase activity was normalized to renilla luciferase activity. Results are presented as mean values ± SD. (D) HMVEC-d monolayers were scratched to create a wound and incubated under low serum concentration (0.5% FBS) in the presence or absence of BMP10 (100 ng/mL). At time 0, 24, and 48 hours after wounding, the cells were observed by phase-contrast microscopy and photographed. Results from 1 representative experiment of 3 are presented as the percent of wound closure at different times after wounding ± SE. (E) HMVEC-d's were treated for 24 and 48 hours with BMP10 (100 ng/mL) in 5% FBS. The quantity of viable cells was determined using the WST-1 assay. Results from 1 representative experiment of 3 are expressed as absorbance (OD450) ± SE. (D-E) **P < .01.
BMP10 is also an activator of ALK1. (A) BMP10 with an N-terminal His6 tag was purified from conditioned medium of 293T cells using metal chelate chromatography. BMP10 was electrophoresed on polyacrylamide gels under reducing conditions and detected by immunoblotting using antibodies against polyhistidine (His6). (B-C) NIH-3T3 cells were transiently transfected with pGL3(BRE)-luc, pRL-TK-luc, and either pCDNA3 empty vector or pALK1. After 4 hours, cells were treated with BMP9 (0.1 ng/mL) or BMP10 (10 ng/mL) in the presence or absence of ALK1ecd (15-fold molar excess over BMP10 concentrations) for 15 hours. The firefly luciferase activity was normalized to renilla luciferase activity. Results are presented as mean values ± SD. (D) HMVEC-d monolayers were scratched to create a wound and incubated under low serum concentration (0.5% FBS) in the presence or absence of BMP10 (100 ng/mL). At time 0, 24, and 48 hours after wounding, the cells were observed by phase-contrast microscopy and photographed. Results from 1 representative experiment of 3 are presented as the percent of wound closure at different times after wounding ± SE. (E) HMVEC-d's were treated for 24 and 48 hours with BMP10 (100 ng/mL) in 5% FBS. The quantity of viable cells was determined using the WST-1 assay. Results from 1 representative experiment of 3 are expressed as absorbance (OD450) ± SE. (D-E) **P < .01.
BMP9 and BMP10 increase the expression of genes known to be induced by ALK1ca expression
Several genes have been found to be induced by expression of ALK1ca in endothelial cells.21,23–25 We studied whether BMP9 and BMP10 could also induce some of these genes. We chose the ones that were shown to be regulated by ALK1 expression in all 3 microarrays analyses. We found that Smad6 expression was very strongly induced (8- and 13-fold by BMP10 and BMP9, respectively) (Table 2) Inhibitor of DNA binding 1 (Id1), from which is derived the BRE response element, Id2, Smad7, and endoglin were also up-regulated (from 2- up to 8-fold) by both BMP9 and BMP10 (Table 2). We also looked at the regulation of BMPRII, another gene of the TGFβ family that we previously found to be regulated by ALK1ca.21 BMPRII mRNA expression was also increased, about 3-fold, in response to either BMP9 or BMP10 treatment (Table 2).
BMP9 and BMP10 increase the expression of genes known to be induced by ALK1ca expression
| . | ID1 . | ID2 . | SMAD6 . | SMAD7 . | ENG . | BMPRII . |
|---|---|---|---|---|---|---|
| BMP9 | 4.50 | 1.93 | 13.09 | 3.52 | 4.89 | 2.67 |
| BMP10 | 8.00 | 1.73 | 7.77 | 3.57 | 5.98 | 3.04 |
| . | ID1 . | ID2 . | SMAD6 . | SMAD7 . | ENG . | BMPRII . |
|---|---|---|---|---|---|---|
| BMP9 | 4.50 | 1.93 | 13.09 | 3.52 | 4.89 | 2.67 |
| BMP10 | 8.00 | 1.73 | 7.77 | 3.57 | 5.98 | 3.04 |
HMVEC-d's were treated with BMP9 (10 ng/mL) or BMP10 (100 ng/mL) for 15 hours. Gene expression was assessed by qRT-PCR. The results are expressed in fold induction over untreated cells.
Discussion
ALK1 is an endothelial-specific type I receptor of the TGFβ family that has been implicated in angiogenesis. In absence of a specific ligand for ALK1, the study of its cellular function was possible only through the use of a constitutively active form of this receptor (ALK1ca).13,21,23 In the present study, we report that BMP9 and BMP10 are specific ALK1 ligands and activators of the Smad1/5/8 signaling pathway in endothelial cells and that they are potent inhibitors of endothelial cell migration and growth.
ALK1 has long been described as an orphan receptor. Subsequently, TGFβ1 and TGFβ3, primarily known as ligands for ALK5, were also shown to bind ALK1 but only in the presence of ALK5.13,14 It was also shown that, while TGFβ-induced Smad2 phosphorylation remains stable over time (for at least 6 hours), TGFβ-induced Smad5 phosphorylation is very transient (peaking after 90 minutes of treatment),13 and that this might be due to recruitment of protein phosphatase 1α by Smad7.26 Unlike TGFβ, we show that BMP9 induced phosphorylation of Smad1/5/8 over a wide range of BMP9 concentrations (0.01-10 ng/mL), and, in contrast to TGFβ, BMP9-induced phosphorylation of Smad1/5/8 is sustained over at least 24 hours. Furthermore, BMP9 could activate the BMP response element (BRE) present within the Id1 promoter, while TGFβ did not activate this BRE under our experimental conditions (overnight stimulation with 1 ng/mL TGFβ1). However, as previously described,26 we could demonstrate a transient activation of the BRE promoter by TGFβ when HMVEC-d's were stimulated for only 6 hours (data not shown). Finally, we show that BMP9 treatment inhibited human microvascular endothelial cell migration (in the cellular wound closure assay) and cell growth, while TGFβ had no significant effect on HMVEC-d migration (data not shown). Taken together, these data demonstrate that BMP9 is a new ligand for ALK1, which is more specific and more potent than TGFβ at activating the Smad1/5/8 pathway in endothelial cells.
Our data demonstrate that BMP9 activates the BRE promoter element at very low concentrations in HMVEC-d's (EC50 = 45 ± 27 pg/mL). This response could be completely abolished by ALK1 silencing in HMVEC-d's and rescued by addition of ectopic ALK1. We also show that ALK1 overexpression in NIH-3T3 cells strongly increases the BMP9 response, while ALK2 overexpression has no effect, and ALK3 and ALK6 overexpressions only slightly increase the BMP9 response. Moreover, addition of ALK1ecd completely abolishes the BMP9 response, while ALK3ecd has no effect. These data therefore suggest that BMP9 preferentially binds and activates ALK1, although we cannot totally exclude BMP9 binding to ALK3 or ALK6.
It was reported last year that BMP9 binds to biosensor-immobilized recombinant ALK1ecd and BMPRIIecd and also has a very small binding capacity for ActRIIAecd.17 This is slightly different to what we observe here in a cellular context; we show here that silencing of BMPRII or ActRIIA alone had no effect on the BMP9 response, whereas when both type II receptors are down-regulated, the BMP9 response is completely abolished (Figure 3). This suggests that each receptor can compensate for the other and that BMPRII and ActRIIA are equally able to transduce the BMP9 response through ALK1.
We also show here that endoglin overexpression strongly increases the BMP9 response on the BRE promoter, while the BMP2 response is only slightly increased (Figure 4). This is interesting as it suggests that endoglin preferentially increases signaling of ligands that bind to ALK1. Endoglin is a coreceptor that has been shown to interact with TGFβ1, TGFβ3, activin-A, BMP7, and BMP2 ligands solely in the presence of the type I or type II receptor.27 Endoglin function in endothelial cells is not completely clear. However, in a large number of reports, endoglin has been shown to potentiate TGFβ/ALK1 signaling and to interfere with TGFβ/ALK5 signaling.19,20 Of interest, it was very recently described that ALK1, but not ALK5, could specifically phosphorylate endoglin and that this phosphorylation could mediate ALK1-induced regulation of endothelial migration and proliferation.28
In the present work, we present data showing that BMP10 is also an activator of ALK1. Indeed, as for the BMP9 response, the BMP10 response is increased by ALK1 expression, and this response is abolished by addition of ALK1ecd (Figure 6). Protein sequence identity analysis of the entire repertoire of BMP/GDF ligands demonstrates that BMP10 is most closely related to BMP9. Based on sequence comparison of their mature forms, a subgroup comprising BMP9 and BMP10 can be identified. These proteins share 65% amino acid homology and are therefore likely to share the same receptors. BMP10 has recently been shown to bind to ALK3 and ALK6 in MC3T3 cells.22 However, although not discussed in this paper, the BMP10 response was also increased when ALK1 was expressed in Cos-7 cells (Figure 522 ). Therefore, these data together with ours suggest that BMP10, like BMP9, binds to ALK1, ALK3, and ALK6 and that ligand specificity for these receptors might depend on the cellular context.
Taken together with the current literature on TGFβ and BMP signaling,4 our data allow us to propose a model where BMP9 or BMP10 binds to ALK1. After binding, either BMPRII or ActRIIA can be recruited to these complexes, resulting in the phosphorylation of Smad1/5/8. Endoglin is also present within this complex and has the capacity to increase BMP9 signaling. So far, we have documented only BMP9 and BMP10 activation of the Smad-dependent pathway. However it is clear now that BMPs also transduce signals through Smad-independent pathways such as Erk, JNK, and p38MAPK kinase.5 Therefore it will be very interesting to study other signaling pathways such as the LIM kinase 1, which was described to bind to BMPRII cytoplasmic tail and to regulate actin dynamics.29
The cellular function of ALK1 has long been debated.13,21,23 In the present work, we show that BMP9 and BMP10 inhibit endothelial cell migration and growth. This is in strong accordance with our previous observations showing that ALK1ca expression inhibited endothelial cell migration and proliferation.21 Moreover, we show here that BMP9-induced inhibition of migration is abolished by addition of the extracellular domain of ALK1. In addition, we demonstrate that 7 genes, previously shown to be induced by ALK1ca expression,21,23–25 are also induced in response to BMP9 or BMP10 treatment (Table 2). Taken together, these data demonstrate that BMP9 and BMP10 trigger ALK1 biologic functions. These data also favor the hypothesis that ALK1 is implicated in the inhibition of endothelial cell proliferation and migration, and therefore in the maturation phase of angiogenesis. This hypothesis has been corroborated by recent data obtained in vivo in the zebrafish showing that down-regulation of ALK1 gene expression increased endothelial cell proliferation.30 These data are also in accordance with the phenotypes of mouse ALK1−/− embryos that exhibit hyperfusion and hyperdilation of blood vessels and loss of the morphologic, molecular, and functional distinctions between arteries and veins.11,31 With this in mind, we propose the following model for the role of ALK1 in the development of HHT. ALK1 activation by BMP9 inhibits endothelial cell proliferation and migration and therefore maintains existing vessels in a quiescent state. In HHT, expression levels of functional ALK1 are decreased and endothelial cells proliferate and migrate, and this results in aberrant fusion of sprouting arterial and venous endothelial tubes.
BMP9 has been described as a hematopoietic,32 hepatogenic,33 osteogenic,34 and chondrogenic factor.35 It was also reported as a potent inducer of the cholinergic phenotype in the central nervous system36 and more recently as a regulator of glucose metabolism capable of reducing glycemia in diabetic mice.37 Our data demonstrate for the first time that BMP9 is also an important regulator of endothelial cell functions. In contrast to the disperse expression pattern of individual TGFβ family members, BMP9 expression is mainly restricted to the liver.33 It has been determined that nonparenchymal cells of the liver (endothelial, Kupffer, and stellate cells), rather than hepatocytes, are the major source of BMP9.38 Of interest, a receptor with a high binding affinity for BMP9 was characterized in primary cultures of hepatic endothelial cells and Kupffer cells but remained unidentified.38 The binding of BMP9 to this receptor was not competed by BMP2, BMP4, BMP6, BMP7, BMP12, TGFβ1, or TGFβ3.38 This receptor could be ALK1 as none of these ligands can bind to ALK1. The specific expression of BMP9 in the liver is particularly interesting in regard to the genotype-phenotype relationship in HHT. Mutations in the ENG and ACVRL1 genes lead to HHT1 and HHT2, respectively. It is now clearly described that pulmonary and cerebral arterious malformations occur more often in HHT1, whereas hepatic arterious malformations (HAVMs) are more frequent in HHT2.39 In HHT2, the frequency of HAVM is between 38% and 41%, while in HHT1, it is between 2.5% and 8%.3 The specific expression of the ALK1 ligand, BMP9, in the liver might explain this tissue-specific manifestation in HHT2.
BMP10 expression is restricted to the developing and postnatal heart.40 BMP10-deficient mice die in utero between E9.5 and E10.5 due to a defect in cardiogenesis.41 These mutant embryos display cardiac dysgenesis with profound hypoplastic ventricular walls and absence of ventricular trabeculae, while they have a normal allantoic/umbilical connection and undergo normal vascular development in yolk sac and embryo. It has been previously demonstrated that BMP10 can also bind to ALK322 and, of interest, targeted deletion of ALK3 in neural crest cells leads to acute embryonic heart failure.42 These data suggest that BMP9, rather than BMP10, might be the relevant physiological ALK1 ligand mediating its effects on endothelial cells.
ACVRL1, ENG, and SMAD4 mutations are responsible of HHT. However, after intensive screening there remain about 12% of patients with a confirmed HHT clinical diagnosis with no known mutations,43 suggesting the existence of other genes that might be responsible for this disease. Based on our results, BMP9 and BMP10 will certainly be 2 important candidates to examine in this context.
Authorship
Contribution: L.D., C.M., and S.B. performed research; S.M. contributed vital new reagents; S.B. and J.-J.F. designed research and wrote the paper.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: Sabine Bailly, INSERM EMI 01-05, DRDC/ANGIO, CEA-Grenoble, 17 rue des Martyrs, 38054 Grenoble, France; e-mail: sbailly@cea.fr.
An Inside Blood analysis of this article appears at the front of this issue.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 USC section 1734.
Acknowledgments
This work was supported by INSERM, CEA, GEFLUC (Groupement des Entreprises Françaises pour la Lutte contre le Cancer), and PHRC (Projet Hospitalier de Recherche Clinique, Hospices Civils de Lyon, grant no. 27).
We thank the students Julia Stefanski and Camille Buissière for their helpful participation in this work. We thank Dr Aaron J. W. Hsueh's laboratory (Stanford University, CA) for providing us with recombinant BMP10. We are indebted to Dr S. Souchelnytskyi (Ludwig Institute for Cancer Research, Uppsala, Sweden) for providing us several molecular tools used in this study and for helpful discussions. We also thank Dr J. LaMarre (University of Guelph, ON, Canada) for his review of the paper and his constant enthusiastic support.
L.D. was supported by a Coordination de la Formation par la Recherche (CFR) grant from the Commissariat à l'Energie Atomique.



This feature is available to Subscribers Only
Sign In or Create an Account Close Modal