Abstract
Dasatinib is an oral small molecule inhibitor of Abl and Src family tyrosine kinases (SFK), including p56Lck (Lck). Given the central importance of Lck in transmitting signals from the T-cell receptor (TCR) signaling complex and the potent ability of dasatinib to inhibit Lck activity, we hypothesized this agent could provide a novel route of immunomodulation via targeted inhibition of antigen-induced signaling. Herein, we show that dasatinib inhibits TCR-mediated signal transduction, cellular proliferation, cytokine production, and in vivo T-cell responses. However, dasatinib-mediated inhibition does not induce apoptosis because the effect is reversible or may be overcome by signals bypassing the TCR, such as phorbol ester. Signal transduction and proliferative responses via IL-2 remain essentially unperturbed, suggesting that dasatinib displays specificity for TCR signaling. In addition, dasatinib combined with cyclosporine A or rapamycin led to a much more potent inhibition of T-cell activation, suggesting that targeted inhibition of Lck could be a useful adjunct for enhanced immunomodulation. In combination with currently available immunomodulatory agents, SFK inhibition could potentially increase immunomodulatory efficacy while minimizing toxicity of individual agents.
Introduction
T lymphocytes are intimately involved in the pathophysiology of autoimmune diseases, graft-versus-host disease (GVHD), and transplant rejection. The T-cell receptor signaling complex (TCR) recognition of self and foreign peptides presented by major histocompatibility complex (MHC) proteins is the crucial event determining whether T cells will become activated. Qualitative and quantitative aspects of this TCR-MHC interaction along with additional costimulatory or coinhibitory signals delivered by accessory receptors are integrated among multiple signal transduction networks to determine the fate of a given response.1 Despite the myriad biochemical pathways that ultimately account for T-cell activation, the TCR proximal signaling pathway is essential given its primacy in the overall activation process. There are 4 main families of protein tyrosine kinases associated with TCR signaling: Src family kinases (SFK), ZAP-70/Syk, Tec, and Csk.2 Two SFK members, Lck and Fyn, are involved in the very earliest steps of TCR activation, and Lck is suggested to be more important for TCR signaling.3 Thus, it is conceivable that agents targeting SFK activity could become useful adjuvants in therapeutic immunomodulatory regimens targeting aberrant T-cell activation
Dasatinib (BMS-354825, Sprycel, Bristol-Meyers Squibb, New York, NY), a novel protein tyrosine kinase inhibitor targeting SFK and Abl family kinases, was recently approved for use in the treatment of adults with chronic myeloid leukemia (CML) and resistance or intolerance to prior therapy, including imatinib.4-9 In addition to potently inhibiting the constitutively active Bcr-Abl kinase, dasatinib inhibits Lck at low picomolar concentrations.10 Imatinib (STI571, Gleevec; Novartis Pharma, Basel, Switzerland), was the first approved protein tyrosine kinase inhibitor for treating CML and has also been shown to inhibit Lck, but at much higher concentrations. In addition, imatinib was recently shown to inhibit T-cell activation in vitro and exhibited immunosuppressive activity in an animal model of rheumatoid arthritis.11-14
Most current immunomodulatory therapies are not entirely specific for T cells; instead, they result in lymphodepletion or act further downstream of the TCR signaling complex, predominantly restricted to subsets of second messenger pathways. In addition, significant toxicities and narrow therapeutic windows can limit the usefulness of these drugs in certain patients.15 Thus, we set out to examine the effects of dasatinib on TCR signaling, proliferation, and cytokine production in primary human T cells. The ability of dasatinib to specifically target the earliest events in TCR signaling and enhance the inhibitory effects of cyclosporine and rapamycin, as we show here, opens new therapeutic opportunities to address autoimmune diseases, GVHD, and transplant allograft rejection with SFK inhibitors.
Methods
Cell isolation and culture
Peripheral blood was obtained from healthy volunteers after informed consent was obtained according to protocols approved by the Institutional Review Board of the Cleveland Clinic Foundation (Cleveland, OH). Peripheral blood mononuclear cells (PBMCs) were isolated and cultured as previously described.16 Peripheral blood T lymphoblasts (PBTs) were expanded from PBMC after 48 hours stimulation with phytohemagglutinin (PHA; 0.5%) followed by expansion with interleukin (IL)-2 (5 ng/mL; R&D Systems, Minneapolis, MN) for 7 days. This protocol consistently yields a population of more than 98% CD3+ T cells. Photomicrographs of cells in culture were obtained on an Olympus CK40 microscope (Olympus, Center Valley, PA) with a 20× objective (0.4 NA) and an Olympus DP10 digital camera. Images were analyzed using Preview, version 3.0.9 (Apple, Cupertino, CA).
T-cell stimulation, immunoblotting, and electrophoretic mobility shift assay
PBTs were stimulated as previously described with OKT3 (Ortho, Raritan, NJ) and IL-2,16,17 in the presence or absence of the SFK/Abl inhibitor dasatinib (kind gift from Bristol-Myers Squibb). Proteins were separated on 10% Tris-glycine gels under reducing conditions and transferred to nitrocellulose membranes (Invitrogen, Carlsbad, CA). Immunoblotting was performed as previously described,16,17 using the following antibodies: pan antiphosphotyrosine (PY20-HRP, sc-508, Santa Cruz Biotechnology, Santa Cruz, CA) PathScan Multiplex Western Cocktail I (phospho-p90RSK, phospho-AKT, phospho-ERK, phospho-S6, and loading control eIF4E, 7100, Cell Signaling Technology, Beverly, MA). Chemiluminescence was detected with a FluorChem SP CCD camera (Alpha Innotech, San Leandro, CA). Nuclear isolation and electrophoretic mobility shift assay (EMSA) were performed as previously described.18 The following consensus oligonucleotides were used: STAT5 (5′-AGA TTT CTA GGA ATT CAA TCC-3′) and NF-κB (5′-AGT TGA GGG GAC TTT CCC AGG C-3′).
Flow cytometry
PBMCs were stimulated with OKT3, PHA (Sigma, St Louis, MO), or anti-CD3/anti-CD28 beads (Dynabeads CD3/CD28 T Cell Expander, Invitrogen) in the presence or absence of dasatinib, as indicated in the text. Cells were stained with fluorochrome–conjugated antibodies to CD3, CD4, CD8, CD38, or CD69 (Beckman-Coulter, Fullerton, CA). Data were acquired on a Coulter Epics XL-MCL flow cytometer and analyzed with WinMDI 2.8. T-cell apoptosis was analyzed by flow cytometry using a Vybrant Apoptosis Assay Kit (Molecular Probes, Eugene, OR) containing annexin V-phycoerythrin and SYTOX Green dye, per manufacturer's instructions.
Cell-cycle analysis
DNA analysis of cell cycle status was performed using flow cytometry as previously described.19 Briefly, cells were harvested from culture and washed with phosphate-buffered saline (PBS) plus 2% fetal bovine serum. Cells were then fixed with ice-cold 70% ethanol, washed, and resuspended in a solution containing 10 μg/mL propidium iodide and 10 μg/mL bovine RNAase (Boehringer Mannheim, Indianapolis, IN). After 30 minutes, DNA content was analyzed by flow cytometry.
Intracellular phospho-specific flow cytometry
PBTs were stimulated with OKT3 or IL-2 in 100 μL of RPMI 1640 + 2.5% HEPES (Invitrogen), in the presence or absence of dasatinib. The stimulation was terminated by addition of 50 μL prewarmed 4% formaldehyde (Cytofix Buffer, 554655; BD Biosciences, San Jose, CA) at 37°C for 10 minutes, resulting in a final concentration of 1.3% formaldehyde. After fixation, cells were placed on ice for 5 minutes. The volume was adjusted to 2 mL with cold PBS and the cells were pelleted and then resuspended in 1 mL of ice-cold 90% methanol (Perm Buffer III, 558050, BD Biosciences). Cells were incubated on ice for 30 minutes to allow permeabilization, then washed once in 3 mL of cold PBS and resuspended at 106 cells in 100 μL PBS plus 4% BSA for intracellular staining with either antiphosphorylated ERK 1/2-Alexa Fluor 488 (T202/Y204, 612592, BD Biosciences) or antiphosphorylated STAT5-Alexa Fluor 488 (Y694, 612598, BD Biosciences) for 20 minutes at room temperature. Cells were washed once in 3 mL of PBS and then resuspended in PBS for flow cytometric analysis on a Coulter Epics XL-MCL flow cytometer (Beckman Coulter). Data were analyzed using WinMDI 2.8 (Scripps Institute, La Jolla, CA).
Cytokine analysis
PBMCs were stimulated with OKT3, PHA, or anti-CD3/anti-CD28 beads in the presence or absence of dasatinib, as indicated in the text. Supernatants were analyzed for cytokines using the Proteome Profiler Array (ARY005; R&D Systems) per manufacturer's instructions, and chemiluminescence was detected with a FluorChem SP CCD camera.
T-cell proliferation
T-cell proliferation was analyzed by staining with carboxy-fluorescein diacetate succinimidyl ester (CFSE) at a final concentration 5μM (Molecular Probes, Invitrogen) according to the manufacturer's instructions. Briefly, cells were resuspended in warm PBS containing 5 μM CFSE and incubated at 37°C for 10 minutes. Subsequently, cells were washed twice and resuspended in RPMI 1640 containing 10% FBS before the experimental treatments were initiated. Cells were analyzed by flow cytometry after 5 days in culture, unless otherwise noted. Alternatively, a 96-well plate was coated with a monoclonal antibody to CD3 (G19-4), the antibody was allowed to bind, and then the plate was washed. Normal human peripheral blood T cells were added to the wells, along with test compound and anti-CD28 antibody (9.3) to provide costimulation. After 3 days, [3H]thymidine was added to the cells and incubated for 6 hours. The cells were harvested and counted in a liquid scintillation counter (TopCount, Perkin-Elmer) to measure proliferation. Compounds were tested in triplicate and IC50 values were calculated by the 4-parameter logistic equation (Hill equation) using the GraphPad Prism 4 program.
Mice
Adult male 6- to 8-week-old C57BL/6, C3H/HeJ, and BALB/c mice were purchased from Harlan (Indianapolis, Indiana) and used in all in vivo studies.
In vivo mixed lymphocyte reaction
Responder lymphocytes were isolated from the spleen and lymph nodes of C57BL/6 mice and labeled with CFSE (Molecular Probes). CFSE-labeled responder lymphocytes were adoptively transferred to lethally irradiated C3H/HeJ recipient mice (40 × 106 cells per recipient). Lethally irradiated C57BL/6 recipient mice were used as syngeneic controls for homeostatic proliferation. Each experimental group consisted of 3 animals. Lymphocytes were harvested from the spleens of recipient mice on day 3 after transfer and analyzed by flow cytometry for the CFSE content of donor CD4+ lymphocytes. Compounds were administered by oral gavage at the doses indicated in a vehicle consisting of 50% propylene glycol and 50% water. Dosing was performed once per day for the duration of the study (3 days) for a total of 3 doses.
Results
Dasatinib inhibits signal transduction through the TCR complex
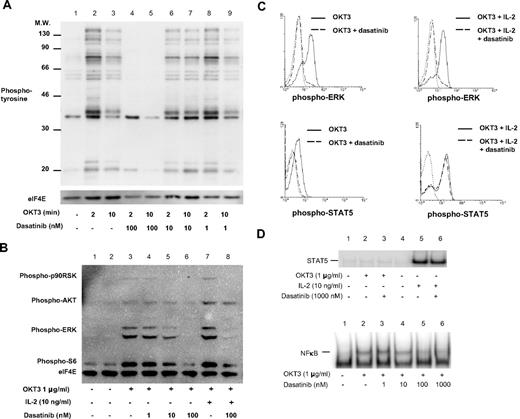
Considering the critical role for SFK in the earliest steps of TCR signaling, we examined the ability of dasatinib to inhibit the tyrosine phosphorylation cascade initiated on TCR/CD3 engagement. PBTs were stimulated with OKT3 (anti-CD3ϵ),16,20 and the ability of dasatinib to inhibit global protein tyrosine phosphorylation at doses ranging from 1 to 100 nM was investigated (Figure 1A). Based on these results, we showed that inhibition of TCR signaling is evident at dasatinib levels as low as 10 nM (Figure 1A, lane 2 vs 6), and complete inhibition of TCR induced tyrosine phosphorylation with 100 nM dasatinib (Figure 1A, lanes 4 and 5).
TCR complex signal transduction, but not IL-2, is inhibited by dasatinib. T cells were stimulated in the presence or absence of dasatinib for 2 or 10 minutes at 37°C, then immediately lysed. Proteins were separated by SDS-PAGE, transferred to nitrocellulose, and probed with (A) pan antiphosphotyrosine or (B) PathScan Multiplex Western Cocktail I (phospho-p90RSK, phospho-AKT, phospho-ERK, phospho-S6, and loading control eIF4E). Chemiluminescent detection was performed with a FluorChem SP CCD camera. (C) T cells were stimulated for 15 minutes with IL-2 and/or 5 minutes with OKT3, then fixed with formaldehyde at 37°C for 10 minutes. Cells were then permeabilized with methanol and stained for phospho-ERK or phospho-STAT5. Solid lines, stimulated T cells (10 μg/mL OKT3 alone or with 20 ng/mL IL-2 as indicated); dashed lines, stimulated T cells in the presence of 10 nM dasatinib; dotted lines, control. (D) T cells were stimulated with OKT3 or IL-2 for 30 minutes at 37°C, nuclear extracts were isolated, and DNA binding of STAT5 and NFκB was determined by EMSA.
TCR complex signal transduction, but not IL-2, is inhibited by dasatinib. T cells were stimulated in the presence or absence of dasatinib for 2 or 10 minutes at 37°C, then immediately lysed. Proteins were separated by SDS-PAGE, transferred to nitrocellulose, and probed with (A) pan antiphosphotyrosine or (B) PathScan Multiplex Western Cocktail I (phospho-p90RSK, phospho-AKT, phospho-ERK, phospho-S6, and loading control eIF4E). Chemiluminescent detection was performed with a FluorChem SP CCD camera. (C) T cells were stimulated for 15 minutes with IL-2 and/or 5 minutes with OKT3, then fixed with formaldehyde at 37°C for 10 minutes. Cells were then permeabilized with methanol and stained for phospho-ERK or phospho-STAT5. Solid lines, stimulated T cells (10 μg/mL OKT3 alone or with 20 ng/mL IL-2 as indicated); dashed lines, stimulated T cells in the presence of 10 nM dasatinib; dotted lines, control. (D) T cells were stimulated with OKT3 or IL-2 for 30 minutes at 37°C, nuclear extracts were isolated, and DNA binding of STAT5 and NFκB was determined by EMSA.
In addition to overall protein tyrosine phosphorylation, we specifically analyzed the induction of 2 key second messenger pathways downstream from the TCR signaling apparatus, the extracellular signal regulated kinase (ERK) and phosphatidylinositol-3 kinase (PI3K)–AKT pathways. In the presence of 10 nM dasatinib, there was a prominent inhibition of TCR-induced ERK phosphorylation, whereas AKT phosphorylation was minimally affected (Figure 1B, lane 5, compared with no inhibitor in lane 3). Similar to the results of pan-tyrosine phosphorylation, with 100 nM dasatinib a complete loss of phospho-ERK and further reduction in phospho-AKT were observed (Figure 1B, lane 6). Diminished ERK and AKT activity were closely paralleled by decreased phosphorylation of their downstream targets p90RSK and S6 ribosomal protein (Figure 1B). Interestingly, the difference of TCR versus IL-2 receptor signaling with regard to ERK and AKT activation was highlighted by the fact that AKT phosphorylation was predominantly mediated via IL-2 (resistant to dasatinib, Figure 1C) and ERK was mostly activated by the TCR (sensitive to dasatinib, Figure 1B).
In PBT, both TCR and IL-2R engagement can activate the PI3K-AKT pathway; however, IL-2 appears to be a more potent stimulus in this regard.20 The observation that AKT phosphorylation, a downstream mediator of PI3K signaling, was essentially unperturbed by dasatinib in the presence of IL-2 suggested that this agent was highly specific for TCR signaling. To further investigate the mechanism of dasatinib action, we analyzed TCR and IL-2R signaling at the single-cell level by intracellular phospho-specific flow cytometry (Figure 1C). Phospho-ERK was strongly induced by OKT3 and OKT3 plus IL-2 after 5 minutes in 71% and 75% of PBT, respectively (Figure 1C top panels, solid line). When stimulated in the presence of 10 nM dasatinib, only 5% of OKT3 stimulated or 13% of OKT3 + IL-2 stimulated PBT stained positive for phospho-ERK (Figure 1C top panels, dashed line), and at 100 nM dasatinib there was a complete inhibition (data not shown). In addition, phospho-STAT5, which is predominantly induced by JAK3 associated with the IL-2 receptor, was weakly elevated in 11% of PBT stimulated by OKT3 alone and strongly induced in 65% of PBT stimulated with IL-2 in addition to OKT3 (Figure 1C, bottom panels, solid line). The presence of 10 nM dasatinib completely abrogated any phospho-STAT5 resulting from OKT3 alone, yet showed no effect on phospho-STAT5 induced by OKT3 plus IL-2 (Figure 1C bottom panels, dashed line).
In addition to STAT5 phosphorylation analysis at the single-cell level, we investigated the ability of dasatinib to inhibit STAT5 DNA binding activity (Figure 1D top panel). We did not observe any increase in STAT5 DNA binding when PBTs were stimulated with OKT3 alone (Figure 1D top panel, lane 2). However, there was a prominent increase in STAT5 DNA binding when PBTs were treated with IL-2, and this was largely resistant to the presence of dasatinib even at concentrations up to 1000 nM (Figure 1D top panel, lanes 5 and 6).
To further characterize T-cell signaling pathways that may be affected by dasatinib, we examined NFκB activation by EMSA (Figure 1D, bottom panel). PBTs were stimulated with OKT3 for 30 minutes in the presence or absence of increasing concentrations of dasatinib. A minimal inhibition of NFκB DNA binding at 10 nM and nearly complete inhibition at 100 nM dasatinib was observed (Figure 1D bottom panel, lanes 4 and 5).
Dasatinib inhibits induction of T-cell early activation markers
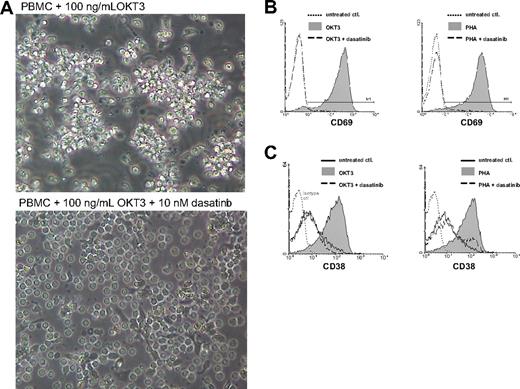
When PBMCs are stimulated with OKT3 alone, monocytes bind the Fc portion of the antibody, leading to activation of both T cells and monocytes which in turn results in increased cell-cell contact, polarization, and clustering (Figure 2A top). However, in the presence of 10 nM dasatinib a marked inhibition of T-cell clustering around monocytes was observed (Figure 2A bottom). Importantly, T cells persisted in culture in the presence of dasatinib and retained similar viability as the unstimulated control cells without dasatinib (data not shown). After 20 hours, T cells were analyzed for CD69 expression (an early marker of T-cell activation) by flow cytometric analysis. OKT3 stimulation potently induces CD69 expression, as shown in this representative donor (Figure 2B, left panel, gray shaded area). However, in the presence of 10 nM dasatinib, complete inhibition of CD69 induction on T cells was achieved (Figure 2B left panel, dashed line). Whereas OKT3 stimulates T cells via direct engagement of the TCR complex, the lectin PHA simultaneously cross-links multiple receptors on T cells and antigen presenting cells, acting as a potent mitogen and more complex stimulus.21 As expected, PHA induced high levels of CD69 (Figure 2B right panel, gray shaded area). However, inhibition of SFK activity with dasatinib markedly reduced the surface expression of these activation markers even though multiple costimulatory cell surface receptors were being triggered (Figure 2B right panel, dashed line).
Dasatinib inhibits TCR-mediated activation of primary human T cells. (A) PBMCs were cultured with OKT3 in the absence (top) or presence (bottom) of dasatinib for 4 hours (original magnification, ×400). (B,C) PBMCs were stimulated with OKT3 (100 ng/mL) or PHA (0.5%) in the presence or absence of dasatinib (10 nM). T cells were analyzed for CD69 expression after 20 hours (B) or CD38 after 72 hours (C) by flow cytometry. Solid gray, OKT3 or PHA stimulation; dashed lines, stimulated T cells in the presence of 10 nM dasatinib; dotted lines, control.
Dasatinib inhibits TCR-mediated activation of primary human T cells. (A) PBMCs were cultured with OKT3 in the absence (top) or presence (bottom) of dasatinib for 4 hours (original magnification, ×400). (B,C) PBMCs were stimulated with OKT3 (100 ng/mL) or PHA (0.5%) in the presence or absence of dasatinib (10 nM). T cells were analyzed for CD69 expression after 20 hours (B) or CD38 after 72 hours (C) by flow cytometry. Solid gray, OKT3 or PHA stimulation; dashed lines, stimulated T cells in the presence of 10 nM dasatinib; dotted lines, control.
CD38 is an ecto-nicotinamide adenine dinucleotide glycohydrolase that is expressed on lymphocytes after activation. We assessed CD38 expression on T cells 72 hours after stimulation with OKT3 or PHA (Figure 2C left and right panels, respectively). Similar to the results observed with CD69 expression, CD38 was strongly up-regulated by both OKT3 and PHA and equally inhibited in the presence of 10 nM dasatinib. Further analysis of CD4 and CD8 T-cell subsets did not reveal any significant differences with respect to dasatinib-mediated inhibition of activation markers (data not shown). Interestingly, although certain patients appear to be susceptible to dasatinib inhibition of TCR signaling at much lower concentrations, we have found that 10 nM dasatinib consistently inhibits T-cell activation in all patients tested.
Dasatinib-mediated T-cell inhibition can be overcome by stimulation bypassing the TCR complex
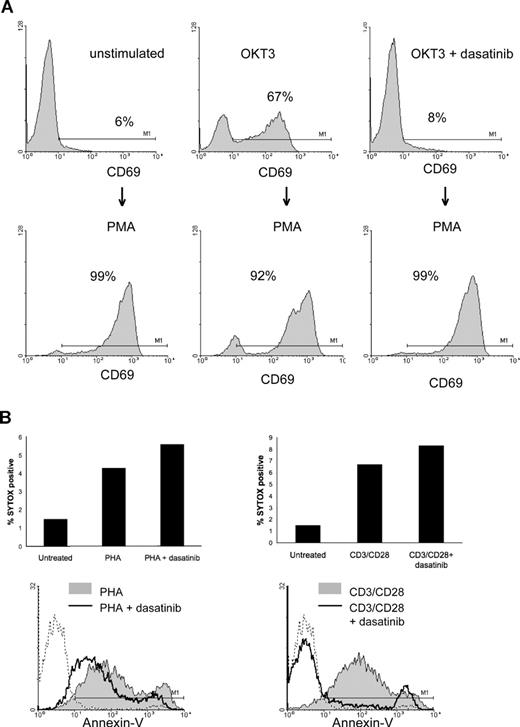
Recently, dasatinib was shown to induce apoptosis in cells with an EGFR mutation that is dependent on EGFR for survival.22 However, dasatinib did not induce apoptosis in PBMC, IL-2–dependent PBT, or Jurkat leukemic T cells (data not shown). Because the dasatinib-treated T cells were unresponsive to TCR signaling but responsive to IL-2 and remained viable in culture, we examined the ability of phorbol 12-myristate 13-acetate (PMA) to induce CD69 expression in the presence of dasatinib (Figure 3A). PMA is capable of inducing CD69 expression via protein kinase C, effectively bypassing the TCR and thus Lck dependency.23 PBMCs were placed into overnight culture with OKT3 in the presence or absence of dasatinib, and CD69 expression was analyzed by flow cytometry. As shown, CD69 is readily induced on OKT3-stimulated T cells but completely inhibited when dasatinib was added (Figure 3A top panels). When PMA is added to the culture for an additional 16 hours, CD69 expression is prominently induced in all treatment groups (Figure 3A bottom panels). Thus, T cells not only survive dasatinib treatment, but they remained capable of being activated in its presence by stimuli bypassing the TCR, highlighting the exquisite specificity of dasatinib against the TCR signaling pathway. To further support the above data that dasatinib does not induce T-cell apoptosis, we assessed the ability to stain cellular nucleic acids with the membrane impermeant dye SYTOX green (Figure 3B). This dye will only gain access to nucleic acids when the integrity of the plasma membrane is impaired, such as during cell death. PBMCs were stimulated with PHA or anti-CD3/anti-CD28 beads (CD3/CD28 beads) for 72 hours and then stained with SYTOX green. There was only slight increase (4%-6%) in SYTOX green staining on T-cell stimulation, and there was no significant further increase in the presence of dasatinib (Figure 3B top panels). These data also suggest that dasatinib does not paradoxically preserve T cells from cell death. In addition to SYTOX green, we performed the standard assessment of exposed phosphatidylserine (PS) on the outer leaflet of the plasma membrane by annexin V staining (Figure 3B bottom panels).24,25 However, this assay is not a reliable tool for measuring apoptosis induction in T cells stimulated through the TCR because TCR signaling inhibits the inward-directed PS transport without affecting the outward transport, leading to increased PS exposure on the cell surface that correlates with activation.26 In agreement with the previously published findings, T cells stimulated with both PHA and CD3/CD28 beads display markedly increased annexin V staining (Figure 3B bottom panels, gray shaded area). However, it is interesting to note that, in the presence of dasatinib, there is decreased annexin V staining after stimulation with PHA or CD3/CD28 beads (Figure 3B top panels, black line), which parallels the decrease in T-cell activation.
Stimulation bypassing the TCR complex can overcome the effects of SFK inhibition. (A) PBMCs were stimulated with OKT3 in the presence or absence of 10 nM dasatinib for 20 hours, and a portion were analyzed by flow cytometry for CD69 (top row). The same groups were then stimulated with PMA (50 ng/mL) for an additional 20 hours and T cells analyzed again for CD69 expression (bottom row). (B) PBMCs were stimulated with PHA or CD3/CD28 beads for 72 hours and then stained with SYTOX green and annexin V and analyzed by flow cytometry.
Stimulation bypassing the TCR complex can overcome the effects of SFK inhibition. (A) PBMCs were stimulated with OKT3 in the presence or absence of 10 nM dasatinib for 20 hours, and a portion were analyzed by flow cytometry for CD69 (top row). The same groups were then stimulated with PMA (50 ng/mL) for an additional 20 hours and T cells analyzed again for CD69 expression (bottom row). (B) PBMCs were stimulated with PHA or CD3/CD28 beads for 72 hours and then stained with SYTOX green and annexin V and analyzed by flow cytometry.
Proinflammatory cytokine production is inhibited by dasatinib
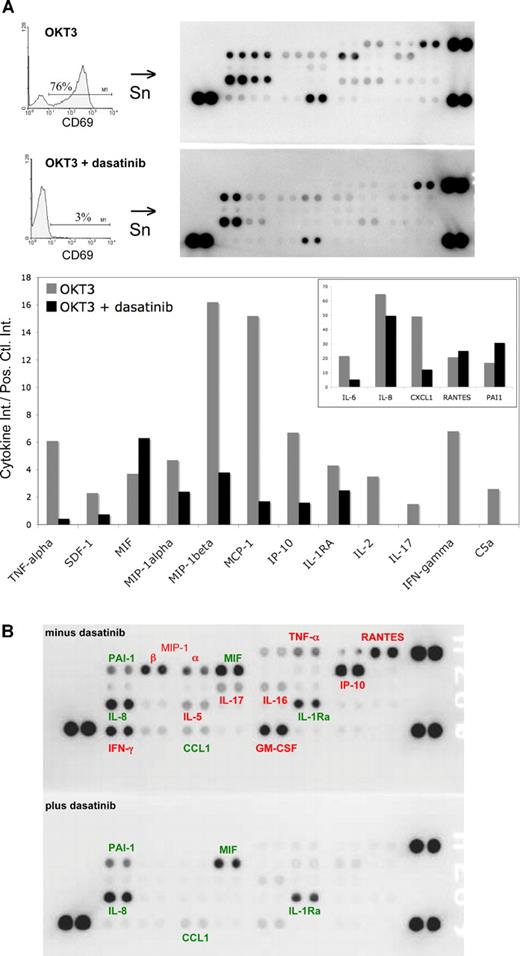
Cytokine production is a key process by which activated T cells participate in immune responses; and in situations of aberrant immune activity (autoimmunity, GVHD, transplant rejection), cytokines are involved in the pathophysiologic sequelae.27-29 As in previous experiments, we cultured PBMCs with OKT3 in the presence or absence of dasatinib. After 24 hours, T cells were analyzed for CD69 expression and supernatants were collected for cytokine analysis (Figure 4A). As shown earlier, a marked inhibition of CD69 induction with 10 nM dasatinib was detected. This effect was associated with inhibition of multiple cytokines usually induced by OKT3, including proinflammatory cytokines (TNF-α, IFN-γ, IL-2, IL-6, and IL-17) and chemotactic factors (SDF-1, MIP-1α, MIP-1β, MCP-1, and CXCL-1; see accompanying graph in Figure 4A). Interestingly, production of several cytokines by PBMCs was either not affected or slightly increased in the presence of dasatinib after 24 hours.
Proinflammatory cytokine production is inhibited by dasatinib. (A) PBMCs were stimulated with OKT3 (100 ng/mL) in the presence or absence of dasatinib (10 nM). After 24 hours, supernatants (Sn) were collected and T cells were analyzed for CD69 expression, as in Figure 3. Cytokines in the supernatants were detected by capture antibodies spotted in duplicate on nitrocellulose membranes (R&D Systems). Chemiluminescence signal was detected with a FluorChem SP CCD camera and intensity was quantitated using Quantity One 1-D Analysis Software (Bio-Rad, Hercules, CA). The high-intensity spots in the 3 corners are positive controls; the top left corners contain the negative controls. A graph of the relative intensity (compared with average of positive controls) is shown for selected cytokines. The inset graph is the same data, but on a larger scale for cytokines with higher expression levels. (B) PBTs were stimulated with 10 ng/mL OKT3 in the presence or absence of 10 nM dasatinib for 48 hours. Supernatants were collected for cytokine analysis as in Figure 4A, selected cytokines are labeled: red, cytokines that decreased by greater than 2-fold in the presence of dasatinib; green, cytokines that decreased by less than 2-fold or did not change in the presence of dasatinib.
Proinflammatory cytokine production is inhibited by dasatinib. (A) PBMCs were stimulated with OKT3 (100 ng/mL) in the presence or absence of dasatinib (10 nM). After 24 hours, supernatants (Sn) were collected and T cells were analyzed for CD69 expression, as in Figure 3. Cytokines in the supernatants were detected by capture antibodies spotted in duplicate on nitrocellulose membranes (R&D Systems). Chemiluminescence signal was detected with a FluorChem SP CCD camera and intensity was quantitated using Quantity One 1-D Analysis Software (Bio-Rad, Hercules, CA). The high-intensity spots in the 3 corners are positive controls; the top left corners contain the negative controls. A graph of the relative intensity (compared with average of positive controls) is shown for selected cytokines. The inset graph is the same data, but on a larger scale for cytokines with higher expression levels. (B) PBTs were stimulated with 10 ng/mL OKT3 in the presence or absence of 10 nM dasatinib for 48 hours. Supernatants were collected for cytokine analysis as in Figure 4A, selected cytokines are labeled: red, cytokines that decreased by greater than 2-fold in the presence of dasatinib; green, cytokines that decreased by less than 2-fold or did not change in the presence of dasatinib.
PBMCs constitute a mixed cell population comprising T cells, monocytes, B cells, natural killer cells, as well as some contaminating neutrophils. Therefore, to more specificallyaddress the ability of dasatinib to inhibit SFK-dependent T-cell cytokine production, we analyzed supernatants after PBT (> 98% CD3-positive) were stimulated in the presence or absence of 10 nM dasatinib (Figure 4B). PBT stimulated through the TCR-induced multiple proinflammatory cytokines and chemokines that were mostly inhibited when dasatinib was present, including RANTES, MIP-1α, MIP-1β, IP-10, TNF-α, IFN-γ, IL-17, and GM-CSF. Cytokines in red decreased by more than or equal to 2-fold in the presence of dasatinib; cytokines in green decreased by less than 2 fold or were slightly increased in the presence of dasatinib (Figure 4B).
T-cell proliferation induced by TCR engagement, but not IL-2, is markedly inhibited by dasatinib
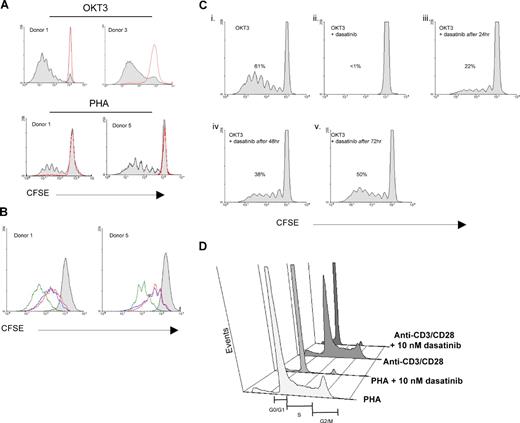
Another consequence of T-cell activation is entry into the cell cycle and clonal expansion. We therefore examined the proliferative capacity of T cells stimulated with OKT3 or PHA treated with dasatinib (Figure 5). When PBMCs are stimulated with OKT3 (Figure 5A top panel) or PHA (Figure 5A bottom panel), a significant portion of T cells proliferate and can be tracked via flow cytometry with the intracellular dye CFSE, as it is diluted in half with each cell division.30 However, in the presence of 10 nM dasatinib, a complete inhibition of OKT3 and PHA-induced T-cell proliferation (Figure 5A red lines) was observed, correlating with the effects of dasatinib on TCR signaling and surface marker expression.
T-cell proliferation induced by TCR engagement, but not IL-2, is markedly inhibited by dasatinib. (A) PBMCs were loaded with CFSE and then stimulated with OKT3 (100 ng/mL; top panels) or PHA (0.5%; bottom panels) in the presence or absence of dasatinib (10 nM). After 5 days, T lymphocytes were analyzed by flow cytometry for proliferation based on CFSE dilution using WinMDI 2.8 and ModFit LT. OKT3 or PHA stimulation alone is shown by the gray-shaded area, whereas OKT3 or PHA in the presence of dasatinib is indicated by the red line, which overlapped the untreated control. (B) PBT expanded in IL-2 for 7 days were rested overnight, loaded with CFSE, and then stimulated with IL-2 or IL-2 + OKT3 in the presence or absence of dasatinib (10 nM). After 5 days, T lymphocytes were analyzed by flow cytometry for proliferation. Gray, unstimulated; blue, IL-2 alone; green, IL-2 + OKT3; red, IL-2 + OKT3 + dasatinib. (C) PBMCs were loaded with CFSE and then stimulated with OKT3 (100 ng/mL). Dasatinib (10 nM) was added at time zero (ii), after 24 hours (iii), after 48 hours (iv), or after 72 hours (v) before analysis at 120 hours. The percentage of proliferating T cells is indicated in each panel. (D) PBMCs were stimulated with PHA or CD3/CD28 beads for 72 hours and then stained with propidium iodide. DNA content was then measured by flow cytometry.
T-cell proliferation induced by TCR engagement, but not IL-2, is markedly inhibited by dasatinib. (A) PBMCs were loaded with CFSE and then stimulated with OKT3 (100 ng/mL; top panels) or PHA (0.5%; bottom panels) in the presence or absence of dasatinib (10 nM). After 5 days, T lymphocytes were analyzed by flow cytometry for proliferation based on CFSE dilution using WinMDI 2.8 and ModFit LT. OKT3 or PHA stimulation alone is shown by the gray-shaded area, whereas OKT3 or PHA in the presence of dasatinib is indicated by the red line, which overlapped the untreated control. (B) PBT expanded in IL-2 for 7 days were rested overnight, loaded with CFSE, and then stimulated with IL-2 or IL-2 + OKT3 in the presence or absence of dasatinib (10 nM). After 5 days, T lymphocytes were analyzed by flow cytometry for proliferation. Gray, unstimulated; blue, IL-2 alone; green, IL-2 + OKT3; red, IL-2 + OKT3 + dasatinib. (C) PBMCs were loaded with CFSE and then stimulated with OKT3 (100 ng/mL). Dasatinib (10 nM) was added at time zero (ii), after 24 hours (iii), after 48 hours (iv), or after 72 hours (v) before analysis at 120 hours. The percentage of proliferating T cells is indicated in each panel. (D) PBMCs were stimulated with PHA or CD3/CD28 beads for 72 hours and then stained with propidium iodide. DNA content was then measured by flow cytometry.
PBTs will proliferate with exogenous IL-2 added to the culture (Figure 5B blue lines), but not with OKT3 alone, because of the absence of costimulation provided by APC (data not shown). However, signaling through the TCR can significantly enhance IL-2–induced proliferation (Figure 5B green lines). When dasatinib was added to those cultures containing IL-2 only, there was no effect on proliferation (data not shown); however, when added to cultures containing IL-2 plus OKT3, there was reduced proliferation to the level of IL-2 only, as if the OKT3 was not present (Figure 5B red lines). These results again highlight the specificity of dasatinib in targeting TCR signaling, whereas IL-2 signaling and activation are essentially unaffected.
We have previously shown that signal transduction through the TCR is dynamically regulated by opposing kinase and phosphatase activities and that ongoing Lck activity is required for optimal activation of second messenger pathways.31 Therefore, we examined the ability of the SFK inhibitor dasatinib to decrease T-cell proliferation at various time points after PBMCs were stimulated through the TCR/CD3 complex (Figure 5C). In the presence of 10 nM dasatinib at the initiation of T-cell activation, there was insufficient signaling to enter the cell cycle (Figure 5Cii). Importantly, even after 24- to 48-hour stimulation of PBMCs, inhibition of SFK activity can still decrease TCR-mediated T-cell proliferation for the duration of the 5-day culture (36% and 62% of control, respectively).
Having shown that dasatinib effectively inhibits T-cell proliferation, we sought to determine the stage of the cell cycle in which dasatinib arrests T cells by analyzing DNA content with propidium iodide (Figure 5D). As expected, there is a marked increase in the number of T cells in the S phase and G2/M on PHA or CD3/CD28 bead stimulation. However, in the presence of dasatinib, the cells fail to enter the cell cycle. This is in agreement with our data showing that the very earliest events of TCR signaling are inhibited, thus inhibiting the second messenger systems that trigger entry into the cell cycle.
T-cell activation induced by TCR/CD3 engagement plus CD28 costimulation is inhibited by dasatinib
T-cell stimulation with OKT3 and PHA requires the presence of monocytes for complete activation by providing costimulatory ligands. In an effort to more efficiently engage the costimulatory pathway through CD28, we stimulated PBMCs with beads bearing both anti-CD3 and anti-CD28 (CD3/CD28 beads) that have been specifically designed to provide optimal activation signals for proliferation.32 T cells engaged by CD3/CD28 beads undergo lymphoblastic growth (Figure 6A left panel), proliferation (Figure 6B left panel), and increased cytokine production (Figure 6C left panel). When PBMCs are stimulated in the presence of 10 nM dasatinib, T cells are bound by the CD3/CD28 beads yet fail to undergo growth (Figure 6A, right panel), proliferation (Figure 6B right panel), or cytokine production (Figure 6C right panel). Furthermore, dasatinib was found to inhibit the proliferation of normal human peripheral blood T cells stimulated by TCR/CD3 engagement by solid phase anti-CD3 antibody plus CD28 costimulation by soluble anti-CD28 antibody. Dasatinib inhibited proliferation with an IC50 value of 2.8 nM, and achieved essentially complete inhibition of proliferation at a concentration of 30 nM in this assay. By comparison, cyclosporin A (CsA) inhibited T-cell proliferation less potently, displaying an IC50 value of 136 nM under the same conditions.
T-cell proliferation and cytokine production induced by TCR/CD3 engagement plus CD28 costimulation is potently inhibited by dasatinib. PBMCs were loaded with CFSE and then stimulated with anti-CD3/CD28 beads in the presence or absence of 10 nM dasatinib. (A) Microscopic analysis of PBMC cultures at 72 hours. (B) CFSE staining was analyzed by flow cytometry at 120 hours. Numbers on plots are the percentage of proliferating T cells. (C) Cytokine production was analyzed in supernatants at 120 hours. (D) Peripheral blood T cells were stimulated with plate-bound anti-CD3 plus soluble anti-CD28 antibody for 3 days in the presence of various concentrations of dasatinib or cyclosporin A, or in the presence of DMSO vehicle alone. T-cell proliferation was measured by [3H] thymidine incorporation. Assays were performed in triplicate, and the percentage inhibition relative to controls treated with DMSO vehicle was calculated.
T-cell proliferation and cytokine production induced by TCR/CD3 engagement plus CD28 costimulation is potently inhibited by dasatinib. PBMCs were loaded with CFSE and then stimulated with anti-CD3/CD28 beads in the presence or absence of 10 nM dasatinib. (A) Microscopic analysis of PBMC cultures at 72 hours. (B) CFSE staining was analyzed by flow cytometry at 120 hours. Numbers on plots are the percentage of proliferating T cells. (C) Cytokine production was analyzed in supernatants at 120 hours. (D) Peripheral blood T cells were stimulated with plate-bound anti-CD3 plus soluble anti-CD28 antibody for 3 days in the presence of various concentrations of dasatinib or cyclosporin A, or in the presence of DMSO vehicle alone. T-cell proliferation was measured by [3H] thymidine incorporation. Assays were performed in triplicate, and the percentage inhibition relative to controls treated with DMSO vehicle was calculated.
Dasatinib can potentiate CsA or rapamycin inhibition of T-cell proliferation
Because current therapeutic practice with immunomodulatory drugs often involves a combination of agents in an effort to minimize toxicities associated with individual agents, we examined the ability of suboptimal concentrations of dasatinib to potentiate the effects of CsA or rapamycin in a T-cell proliferation assay (Figure 7A). PBMCs were stimulated in the usual fashion with 100 ng/mL OKT3, and the effects of low-dose dasatinib (1 nM), CsA (10 ng/mL; 8.3 nM), and rapamycin (0.1 ng/mL; 0.11 nM) were investigated alone or in combination. Dose-titration studies of each drug on T-cell proliferation were performed to identify concentrations, resulting in partial inhibition (data not shown). We then determined if greater inhibition of T-cell proliferation could be achieved by combination treatment. As shown for each of the 2 representative donors, there is significantly enhanced inhibition of T-cell proliferation when dasatinib is combined with CsA or rapamycin (Figure 7A, bottom panels). This was evident by both the fewer number of cells entering the cell cycle as well as the markedly reduced number of divisions for the limited cells that were able to proliferate.
Dasatinib inhibits in vivo T-cell proliferation and in combination with cyclosporine A or rapamycin leads to enhanced inhibition of T-cell proliferation in vitro. (A) PBMCs were loaded with CFSE and stimulated with OKT3 (100 ng/mL) in the presence or absence of dasatinib (1 nM), cyclosporine A (CsA; 10 ng/mL [8.3 nM]), or rapamycin (0.1 ng/mL [0.11 nM]). Proliferating T cells were analyzed after 120 hours. Data from 2 representative donors are shown to illustrate moderate interindividual differences in response to each drug. The percentage of proliferating T cells is indicated in each panel. (B) PBMCs were loaded with CFSE and stimulated with OKT3 (100 ng/mL) in the presence or absence of dasatinib (1 or 2 nM), or rapamycin (0.1 or 0.2 ng/mL). Proliferating T cells were analyzed after 120 hours. (C) Spleen and lymph nodes cell from C57BL/6 mice and loaded with CFSE and adoptively transferred by tail vein injection to lethally irradiated C3H/HeJ recipient mice (40 × 106 cells per recipient; n = 3 mice/group). Lethally irradiated C57BL/6 recipient mice were used as syngeneic controls. Compounds were administered by oral gavage once per day at the doses indicated in a vehicle consisting of 50% propylene glycol and 50% water. Lymphocytes were harvested from the spleens of recipient mice on day 3 after transfer and analyzed by flow cytometry for the CFSE content of the donor CD4+ lymphocytes. The response index relative to the syngeneic control group is indicated. Comparison of compound treated groups' response index to the vehicle-treated group was performed by Student t test (**P < .01). Error bars represent SEM.
Dasatinib inhibits in vivo T-cell proliferation and in combination with cyclosporine A or rapamycin leads to enhanced inhibition of T-cell proliferation in vitro. (A) PBMCs were loaded with CFSE and stimulated with OKT3 (100 ng/mL) in the presence or absence of dasatinib (1 nM), cyclosporine A (CsA; 10 ng/mL [8.3 nM]), or rapamycin (0.1 ng/mL [0.11 nM]). Proliferating T cells were analyzed after 120 hours. Data from 2 representative donors are shown to illustrate moderate interindividual differences in response to each drug. The percentage of proliferating T cells is indicated in each panel. (B) PBMCs were loaded with CFSE and stimulated with OKT3 (100 ng/mL) in the presence or absence of dasatinib (1 or 2 nM), or rapamycin (0.1 or 0.2 ng/mL). Proliferating T cells were analyzed after 120 hours. (C) Spleen and lymph nodes cell from C57BL/6 mice and loaded with CFSE and adoptively transferred by tail vein injection to lethally irradiated C3H/HeJ recipient mice (40 × 106 cells per recipient; n = 3 mice/group). Lethally irradiated C57BL/6 recipient mice were used as syngeneic controls. Compounds were administered by oral gavage once per day at the doses indicated in a vehicle consisting of 50% propylene glycol and 50% water. Lymphocytes were harvested from the spleens of recipient mice on day 3 after transfer and analyzed by flow cytometry for the CFSE content of the donor CD4+ lymphocytes. The response index relative to the syngeneic control group is indicated. Comparison of compound treated groups' response index to the vehicle-treated group was performed by Student t test (**P < .01). Error bars represent SEM.
The utility of targeting 2 fairly discrete signaling pathways, characterized by Lck and mTOR, was further investigated by comparing combined low doses of dasatinib and rapamycin with a doubling of the individual agents (Figure 7B). Increasing dasatinib from 1 nM to 2 nM, which is near the IC50 value, led to less T-cell proliferation (85% vs 53% proliferating cells, respectively). Increasing rapamycin from 0.1 ng/mL to 0.2 ng/mL had a modest effect on T-cell proliferation (51% vs 43% proliferating cells, respectively). When 1 nM dasatinib was again combined with 0.1 ng/mL rapamycin, there was much greater inhibition of T-cell proliferation (19% proliferating cells) than with either agent alone, or doubling of the individual agents.
In vivo inhibition of T-cell proliferation
The potent inhibition of T-cell proliferation by dasatinib in vitro led us to examine the potential of dasatinib to inhibit the proliferation of T cells in vivo (Figure 7C). Adoptive transfer of allogeneic CFSE labeled spleen and LN cells from C57BL/6 mice to lethally irradiated C3H/HeJ mice results in significant expansion of the transferred donor CD4 + T cells in an allogeneic response, as measured by flow cytometric analysis. In this experimental system, we did not observe homeostatic proliferation when C57BL/6 donor cells were transferred into lethally irradiated C57BL/6 syngeneic mice. Administration of dasatinib once daily by oral gavage to the recipient mice inhibited this T-cell proliferation in a dose-dependent manner. Significant suppression of the proliferative response was observed at doses of 50 and 20 mg/kg (MPK) but not at 5 MPK. The suppression observed at 50 and 20 MPK (99% and 91%, respectively) was comparable with CsA dosed orally at 50 MPK once daily (97%).
Discussion
The protein tyrosine kinase inhibitor dasatinib has demonstrated efficacy for the treatment of CML and Philadelphia chromosome-positive ALL, particularly in patients resistant to imatinib.9 In addition, given the unique property of dasatinib to inhibit Lck at picomolar concentrations, it is possible that this agent may be exploited for immunomodulatory activity as well.10 Imatinib, another protein tyrosine kinase inhibitor, is also capable of inhibiting T-cell activation. However, the required concentrations are significantly higher than dasatinib.11-14 Thus, dasatinib appears to be a much more potent Lck inhibitor, a feature potentially allowing for immunomodulatory effects at therapeutically achievable levels previously used to treat CML. In this regard, the range of concentrations attained in CML patients who responded to dasatinib also corresponds to the range at which dasatinib inhibits T-cell activation.33 It is therefore plausible that this drug could be useful as an immunomodulatory agent in humans.
In an effort to establish a potential role for dasatinib as an immunomodulatory agent, we set out to examine its effect on 3 critical parameters of T-cell responses: TCR signaling and expression of activation markers, cytokine production, and proliferation. All of these processes were inhibited by dasatinib. Interestingly, although concentrations of dasatinib capable of inhibiting cytokine production and proliferation also suppressed TCR signaling, induction of the TCR proximal signaling apparatus was not completely inhibited. Indeed, quantitative and qualitative aspects of TCR engagement can significantly impact the kinetics and duration of second messenger pathways that ultimately guide effector function.20,34,35 Partial induction of TCR-based signaling has been exploited with altered peptide ligands to induce functional immune tolerance.36 Furthermore, altered peptide ligands capable of tolerance induction have been shown to have partial agonist activity, with a distinct signal transduction “signature.”1 We are currently investigating the ability to pharmacologically achieve a tolerogenic signal transduction signature by targeting key elements of the TCR pathway, such as Lck or Abl.37
The inability of dasatinib to inhibit IL-2 receptor signaling, consistently observed both biochemically (phospho-STAT5, STAT5 DNA binding) and functionally (proliferation) was in accord with previously published results showing that IL-2–induced STAT5 activity is independent of Lck.38 These results with IL-2 signaling along with the ability of PMA to bypass the effects of dasatinib confirm the high specificity of this agent against TCR signaling. Furthermore, in combination with agents targeting IL-2 signaling (JAK3 inhibitor, anti-IL-2 receptor antibody), dasatinib may synergistically inhibit T-cell activation and proliferation.
Cytokine production is a key feature by which T cells direct immune responses through induction of immunoproteaosomes, MHC class II expression, immunoglobulin class switching, eliciting the acute phase response, directing cellular migration, and a host of other immunoregulatory functions.39 The ability of dasatinib to inhibit proinflammatory cytokine production could play an important role in reducing ongoing inflammation, leukocyte recruitment, and host or allograft tissue destruction. Our data suggest that ongoing activity of SFK activity contributes to optimal TCR-mediated proliferation because addition of dasatinib, even several days after the initial stimulus, still resulted in significant inhibition of proliferation. Similar results were previously attained with imatinib as well, strengthening the argument that ongoing SFK activity contributes to optimal TCR-mediated proliferation.11 Thus, it will be important to determine whether SFK inhibition can diminish ongoing cytokine and chemokine production from inflammatory sites in vivo.
Interestingly, at the standard dosing level in CML patients (70 mg twice a day), dasatinib does not appear to have significant immunomodulatory activity. This observation is consistent with the ability to deliver a relatively low dose of an agent targeting Bcr-Abl and still achieve therapeutic efficacy because of “oncogenic addiction.”40-42 This well-characterized phenomenon, in which the tumor cells become increasingly dependent on a single oncogenic activity for survival and proliferation, allows for the unforeseen increase in therapeutic efficacy for kinase inhibitors, such as imatinib and dasatinib in CML. Thus, the exquisite sensitivity of the CML tumor cells for inhibition of Bcr-Abl and SFK allows for a relatively lower dose of dasatinib to achieve efficacy without significantly affecting immune function. Interestingly, a dose of 10 mg/kg twice a day in the mouse gives an exposure similar to the approved dose of 70 mg/kg twice a day in humans; however, higher doses have been used in humans and higher doses are currently being examined in solid organs, such as the SARC 009 study (a phase 2 trial of dasatinib in advanced sarcoma, NCT00464620). Because we observed almost complete inhibition of the alloimmune response in an in vivo mixed lymphocyte reaction (MLR) at 20 mg/kg once daily, there is considerable opportunity to alter the dosing in humans to achieve an immunomodulatory effect, particularly in combination with other therapeutic agents targeting additional accessory or costimulatory pathways.
We were able to show enhanced inhibition of T-cell proliferation by combining dasatinib and CsA or rapamycin. This may be the most relevant finding in our results because it opens the opportunity to titrate concentrations of each individual drug below toxicity levels while maintaining the ability to quell aberrant T-cell activation. Further studies combining Lck inhibitors, such as dasatinib or the recently described A-770041,43,44 with costimulatory blockade may also provide new routes to achieve desired levels of immunomodulation while minimizing side effects. Therefore, new dosing schedules could be developed to optimize SFK inhibition in the context of concurrent NFAT, mTOR, or JAK inhibition.
In conclusion, our results provide compelling evidence that the tyrosine kinase inhibitor dasatinib, at low nanomolar concentrations, can effectively inhibit T-cell activation and proliferation. Thus, dasatinib would join a novel class of immunomodulatory agents targeting the antigen specific immune receptor. The ability to combine with and enhance currently available immunosuppressive agents, such as CsA and rapamycin, could help to translate these observations into a meaningful clinical trial. Therefore, future studies will examine dasatinib and related compounds for in vivo efficacy alone and in combination with agents targeting other aspects of T-cell activation, such as costimulatory blockade in addition to CsA and rapamycin. Finally, although not addressed in this manuscript, it would be reasonable to examine the ability of dasatinib to inhibit B cell receptor signaling because B cells use the SFK Lyn at a very proximal step in activation, analogous to Lck in T cells.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 USC section 1734.
Acknowledgments
The authors thank Bristol-Myers Squibb for kindly providing us with dasatinib for use in our studies as well as Dr Joe Bauer for assistance with EMSA.
This work was supported in part by National Institutes of Health grants R01 HL73429-01, R01 CA113972-01, and K24 HL0077522-01 (J.P.M.).
National Institutes of Health
Authorship
Contribution: A.E.S. designed and performed experiments, analyzed data, and wrote the paper; G.L.S., R.T., V.S., and R.Z. designed and performed experiments and analyzed data; A.M.J. and H.S. performed experiments and analyzed data; and J.P.M. analyzed data and wrote the paper.
Conflict-of-interest disclosure: G.L.S., R.Z., R.T., and V.S. are employees of Bristol-Myers Squibb. The remaining authors declare no competing financial interests.
Correspondence: Jaroslaw P. Maciejewski, Taussig Cancer Center/R40, 9500 Euclid Avenue, Cleveland, OH 44195; e-mail: maciejj@ccf.org.

![Figure 6. T-cell proliferation and cytokine production induced by TCR/CD3 engagement plus CD28 costimulation is potently inhibited by dasatinib. PBMCs were loaded with CFSE and then stimulated with anti-CD3/CD28 beads in the presence or absence of 10 nM dasatinib. (A) Microscopic analysis of PBMC cultures at 72 hours. (B) CFSE staining was analyzed by flow cytometry at 120 hours. Numbers on plots are the percentage of proliferating T cells. (C) Cytokine production was analyzed in supernatants at 120 hours. (D) Peripheral blood T cells were stimulated with plate-bound anti-CD3 plus soluble anti-CD28 antibody for 3 days in the presence of various concentrations of dasatinib or cyclosporin A, or in the presence of DMSO vehicle alone. T-cell proliferation was measured by [3H] thymidine incorporation. Assays were performed in triplicate, and the percentage inhibition relative to controls treated with DMSO vehicle was calculated.](https://ash.silverchair-cdn.com/ash/content_public/journal/blood/111/3/10.1182_blood-2007-04-084814/6/m_zh80050813120006.jpeg?Expires=1769090155&Signature=Uoz4KLmJzeZgXmRbE9aOIWM2C2Go7zz7hyPjr2uZDcXr2diD-YIBuM-YVWKQ0NSO2cyeIfjh0GnyCWNxMqWz~r3aIYPtKvIZNWI~DPh1rUJOmtZTlqLpycArBCuCT1XNvrxoMYaATU5IA7uxjuj0OWfXmtB391wW2U5-u2jMwPOZRoucPq5paa~AvaX3E6DakHxyMLShwrQImvEgVdgXH9YuAv4yzsGDU8vJhus6XUO9-G4BzW0XtjCzlGjVgnNyBRgxO67q2caabZmIDXbKaiu7BzFWkOSlfg~CgGtCr4NwTRrgjf07TIWuNUCvj3SZUyshieoWog7IFUN1Gza1yg__&Key-Pair-Id=APKAIE5G5CRDK6RD3PGA)
![Figure 7. Dasatinib inhibits in vivo T-cell proliferation and in combination with cyclosporine A or rapamycin leads to enhanced inhibition of T-cell proliferation in vitro. (A) PBMCs were loaded with CFSE and stimulated with OKT3 (100 ng/mL) in the presence or absence of dasatinib (1 nM), cyclosporine A (CsA; 10 ng/mL [8.3 nM]), or rapamycin (0.1 ng/mL [0.11 nM]). Proliferating T cells were analyzed after 120 hours. Data from 2 representative donors are shown to illustrate moderate interindividual differences in response to each drug. The percentage of proliferating T cells is indicated in each panel. (B) PBMCs were loaded with CFSE and stimulated with OKT3 (100 ng/mL) in the presence or absence of dasatinib (1 or 2 nM), or rapamycin (0.1 or 0.2 ng/mL). Proliferating T cells were analyzed after 120 hours. (C) Spleen and lymph nodes cell from C57BL/6 mice and loaded with CFSE and adoptively transferred by tail vein injection to lethally irradiated C3H/HeJ recipient mice (40 × 106 cells per recipient; n = 3 mice/group). Lethally irradiated C57BL/6 recipient mice were used as syngeneic controls. Compounds were administered by oral gavage once per day at the doses indicated in a vehicle consisting of 50% propylene glycol and 50% water. Lymphocytes were harvested from the spleens of recipient mice on day 3 after transfer and analyzed by flow cytometry for the CFSE content of the donor CD4+ lymphocytes. The response index relative to the syngeneic control group is indicated. Comparison of compound treated groups' response index to the vehicle-treated group was performed by Student t test (**P < .01). Error bars represent SEM.](https://ash.silverchair-cdn.com/ash/content_public/journal/blood/111/3/10.1182_blood-2007-04-084814/6/m_zh80050813120007.jpeg?Expires=1769090155&Signature=T6H5e42YNoq4Uja0nN41IFPGN69nEydXIUrcIcHFu2Vjjm4m7rIW94jFQWN5RrVTYpPi6jrAGxji0CqLVcvSmTAd8cSzQgoav52JOhNgiQl99T1EvHqJ0kVdRMsPscGLPRxCiIf~iF-1e1CvuWX7jDYZsk68Q-ZH1DGtz9SCJZmaxsg0nKrezok2C3XrULCQRQIOtocZmllXrkDtFUCIG5n5hK0FEqT54lhSc~WMVXkJ1JPgUs54jORgAwVTVKrbq6FlFhrLHkyzB0LAifQkwbMlO577~ItvLz2JdeFGZL2mFc7zFn9-C8qSXPQjjhWBW2TXeIbqH4S4fLZE5O0SDw__&Key-Pair-Id=APKAIE5G5CRDK6RD3PGA)
This feature is available to Subscribers Only
Sign In or Create an Account Close Modal