Abstract
Neutralizing autoantibodies to type I, but not type II, interferons (IFNs) are found at high titers in almost every patient with autoimmune polyendocrinopathy candidiasis ectodermal dystrophy (APECED), a disease caused by AIRE gene mutations that lead to defects in thymic T-cell selection. Combining genome-wide expression array with real time RT-PCR assays, we here demonstrate that antibodies against IFN-α cause highly significant down-regulation of interferon-stimulated gene expression in cells from APECED patients' blood by blocking their highly dilute endogenous IFNs. This down-regulation was lost progressively as these APECED cells matured in cultures without neutralizing autoantibodies. Most interestingly, a rare APECED patient with autoantibodies to IFN-ω but not IFN-α showed a marked increase in expression of the same interferon-stimulated genes. We also report unexpected increases in serum CXCL10 levels in APECED. Our results argue that the breakdown of tolerance to IFNs in AIRE deficiency is associated with impaired responses to them in thymus, and highlight APECED as another autoimmune disease with associated dysregulation of IFN activity.
Introduction
Type I interferons (IFNs) are cytokines with pleiotropic activities that contribute to early defense against pathogens, development of adaptive immunity, and protective antitumor responses. The human type I IFN gene family consists of 13 distinct functional IFN-α, and single IFN-β, IFN-ϵ, IFN-κ, and IFN-ω genes; the respective IFN molecules all use the same cell surface receptor complex, IFN-α receptor.1,2 Although seminal studies reported the expression of type I IFNs by monocytes,3 IFN-α, -β, and -ω are secreted in much larger amounts by dendritic cells (DCs), above all by plasmacytoid DCs.4,5 However, virtually all nucleated cells can produce some type I IFNs after viral infection. The activation of IFN genes in DCs depends on IFN regulatory factors 7 (IRF7) and 3 (IRF3), the former termed “master regulator of type I IFN synthesis.”6 After secretion and receptor binding, membrane-proximal immediate signaling is initiated through the catalytic activation of receptor-associated JAK1 and TYK2 tyrosine kinases. Transcription factors in the signal transducer and activator of transcription family members (STAT1 and STAT2) are then attached to the activated receptor complex via phosphotyrosine recruitment motifs and then undergo phosphorylation on tyrosine and, in complex with IRF9 protein, are translocated to the nucleus to up-regulate the expression of IFN-stimulated genes (ISGs).1 Type I IFNs are deeply implicated in pathogenesis of certain autoimmune diseases. In particular, in the chronic systemic autoimmune disease, systemic lupus erythematosus (SLE),2,5,7-9 IFN-α serum levels are elevated in patients with severe SLE and associated with the frequent up-regulation of ISGs, the so-called IFN signature in their peripheral blood mononuclear cells (PBMCs).
Recently, we reported high titer neutralizing autoantibodies to type I, but not type II, IFNs in autoimmune polyendocrinopathy candidiasis ectodermal dystrophy (APECED or APS1) patients,10 a recessive disorder resulting from mutations in the autoimmune regulator (AIRE) gene (7). Although AIRE has been detected in monocyte-derived DCs,11 the protein is principally expressed in medullary thymic epithelial cells (mTECs) where it is thought to control autoimmunity by regulating the expression of peripheral tissue-restricted antigens that induce self-tolerance in developing T cells.11-16 Most APECED patients develop multiple endocrine autoimmune diseases, often with high levels of serum autoantibodies against components of the affected organs. Highly variable clinically, APECED usually begins in infancy with chronic Candida infection, followed by autoimmune attack on the parathyroids, adrenal cortex, and/or gonads, endocrine cells in the gut, pancreatic islets, thyroid gland, and others.17 The prevalence of organ-specific autoantibodies in APECED patients varies between 8% and 66%.18 For those against IFN-ω or IFN-α, it reaches 100% or more than 95%, respectively.10,19 However, anti-IFN antibodies have not been reported in Aire-deficient mice, which do not precisely reproduce many characteristics of APECED, notably the candidiasis.20 Similar anti-IFN autoantibodies are also found in many patients with thymoma, especially those with myasthenia gravis.21
Here we present new evidence supporting our hypothesis that, by specifically neutralizing IFN-α, these autoantibodies decrease expression of ISGs in APECED peripheral blood cells. The down-regulation of ISGs was not caused by any inherent defect in the patients' monocyte-derived DCs or plasmacytoid DCs, as it was reversible if they were cultured without neutralizing autoantibodies. We also report increase in serum chemokine (C-X-C motif) ligand 10 (CXCL10; alias IP-10) levels in APECED that most probably reflect increased production of proinflammatory cytokines in the target tissues.
Methods
Patients
With local approval from the Ethics Review Committee on Human Research of the University of Tartu, Estonia, and informed consent in accordance with the Declaration of Helsinki, we studied 8 APECED patients and age-matched controls. Their ages at sample collection, APECED features, and organ-specific autoantibodies and AIRE genotypes are given in Table 1. They all had high titers of neutralizing autoantibodies against IFN-ω, and the majority against the IFN-α in addition (Table 2), but patient A3 proved to be negative and patient A2 weakly positive in antiviral neutralization assays.10 None of the patients was taking systemic immunosuppressive treatment at the time of the sampling. Sera from Norwegian APECED and Addison disease collection, the Finnish APECED collection, a Sardinian APECED and unaffected heterozygous relative cohort, and some U.S. APECED patient sera and SLE sera from Tartu University Clinic serum bank were used for cytokine measurements.
Disease characteristics, autoantibodies, and AIRE mutations of APECED patients of this study
| Patient no. . | Sex . | Age, y . | Autoantibodies . | Manifestations . | AIRE mutations . |
|---|---|---|---|---|---|
| A1 | M | 58 | 21OH, SCC, AADC, GAD | C, HP, A, V, Al, | c.769C>T/c.769C>T |
| A2 | M | 47 | AADC, GAD, TPH | HP, D, V | c.879+1G>A/c.879+1G>A |
| A3 | F | 54 | 21OH, GAD | C, A, HP, G, AT, Al, E, D | c.769C>T /c.1336T>G |
| A4 | F | 25 | 21OH, 17OH | A | c.769C>T / c.1242_1243insA |
| A5 | F | 20 | 21OH, SCC, 17OH | C, A, V, N, G | c.769C>T/c.1242_1243insA |
| A6 | M | 21 | 21OH, SCC, 17OH, AADC | C, HP, A, K | c.769C>T/c.769C>T |
| A7 | M | 17 | negative | C, HP, M, AT | c.1064-1068dupCCCGG/ c.1064-1068dupCCCGG |
| A8 | F | 56 | 21OH, 17OH | C, HP, A | R257X/967-979del13 |
| Patient no. . | Sex . | Age, y . | Autoantibodies . | Manifestations . | AIRE mutations . |
|---|---|---|---|---|---|
| A1 | M | 58 | 21OH, SCC, AADC, GAD | C, HP, A, V, Al, | c.769C>T/c.769C>T |
| A2 | M | 47 | AADC, GAD, TPH | HP, D, V | c.879+1G>A/c.879+1G>A |
| A3 | F | 54 | 21OH, GAD | C, A, HP, G, AT, Al, E, D | c.769C>T /c.1336T>G |
| A4 | F | 25 | 21OH, 17OH | A | c.769C>T / c.1242_1243insA |
| A5 | F | 20 | 21OH, SCC, 17OH | C, A, V, N, G | c.769C>T/c.1242_1243insA |
| A6 | M | 21 | 21OH, SCC, 17OH, AADC | C, HP, A, K | c.769C>T/c.769C>T |
| A7 | M | 17 | negative | C, HP, M, AT | c.1064-1068dupCCCGG/ c.1064-1068dupCCCGG |
| A8 | F | 56 | 21OH, 17OH | C, HP, A | R257X/967-979del13 |
21OH indicates 21-hydroxylase; SCC, side-chain cleavage enzyme; AADC, aromatic L-amino acid decarboxylase; GAD, glutamic acid decarboxylase; TPH, tryptophan hydroxylase; 17OH, 17α-hydroxylase; A, adrenal insufficiency; HP, hypoparathyroidism; C, mucocutaneous candidiasis; G, primary gonadal insufficiency; V, vitiligo; Al, alopecia; AT, autoimmune thyroid disease; M, malabsorption; N, nail pitting; E, dental enamel hypoplasia; K, keratopathy; and D, type I diabetes.
The titers of anti-IFN neutralizing autoantibodies in APECED patients of this study
| Patient no. . | Neutralizing titer . | |||
|---|---|---|---|---|
| IFN-α 2 . | IFN-α 8 . | IFN-ω . | IFN-β . | |
| A1 | 90 000 | 90 000 | 50 000 | < 40 |
| A2 | 100 | 300 | 30 000 | < 40 |
| A3 | 60 | 40 | 22 500 | < 40 |
| A4 | 110 000 | 256 000 | 90 000 | < 40 |
| A5 | 64 000 | 45 000 | 50 000 | < 40 |
| A6 | > 256 000 | > 256 000 | 256 000 | < 40 |
| A7 | > 256 000 | 180 000 | 20 000 | < 40 |
| A8 | 500 000 | 500 000 | 120 000 | 550 |
| Patient no. . | Neutralizing titer . | |||
|---|---|---|---|---|
| IFN-α 2 . | IFN-α 8 . | IFN-ω . | IFN-β . | |
| A1 | 90 000 | 90 000 | 50 000 | < 40 |
| A2 | 100 | 300 | 30 000 | < 40 |
| A3 | 60 | 40 | 22 500 | < 40 |
| A4 | 110 000 | 256 000 | 90 000 | < 40 |
| A5 | 64 000 | 45 000 | 50 000 | < 40 |
| A6 | > 256 000 | > 256 000 | 256 000 | < 40 |
| A7 | > 256 000 | 180 000 | 20 000 | < 40 |
| A8 | 500 000 | 500 000 | 120 000 | 550 |
Cell isolation, dendritic cell generation, and cell stimulation
PBMCs were separated from heparinized blood samples on Ficoll-Paque PLUS (GE Healthcare, Little Chalfont, United Kingdom). The percentages of plasmacytoid DCs in PBMCs were determined using blood DC antigen 2 (BDCA2 or CD303) antibody and FACSCalibur (both BD Biosciences, San Jose, CA). The plasmacytoid DCs were obtained by positive sorting using anti–BDCA-4–conjugated magnetic microbeads (Miltenyi Biotec, Auburn, CA). The purity of the isolated cells was greater than 95% to 97%. Monocytes were isolated by positive sorting using anti-CD14–conjugated magnetic microbeads (Miltenyi Biotec) to purities greater than 99% and were cultured in 6-well tissue-culture plates with 50 ng/mL granulocyte-macrophage colony-stimulating factor and 25 ng/mL interleukin-4 (IL-4; both from R&D Systems, Minneapolis, MN) for 6 days at 106 cells/mL in RPMI 1640 supplemented with 2 mM l-glutamine, 100 U/mL penicillin, 100 μg/mL streptomycin, and 10% fetal calf serum (PAA Laboratories, Linz, Austria). By day 6, the monocyte-derived DCs stained negative with anti-CD14-allophycocyanin and positive with fluorescein isothiocyanate–anti-DC-specific intercellular adhesion molecule 3 grabbing nonintegrin (DC-SIGN; BD Biosciences). Plasmacytoid DCs were stimulated with Toll-like receptor 9 (TLR9) ligand CpG ODN (2.5 μM, M362; InvivoGen, San Diego, CA) or live influenza virus A/PR/8/34 (a gift from Dr Jim Robertson, National Institute for Biological Standards and Control) 1:100 for 6 hours. Monocyte-derived DCs were stimulated with Toll-like receptor 3 ligand polyinosinic:polycytidylic acid (25 μg/mL, poly(I:C); InvivoGen) for 18 hours.
Affymetrix GeneChips
Buffy coats from APECED patient A8 with high-titer anti–type I IFN autoantibodies, and 2 healthy blood donors were cryopreserved in liquid nitrogen. Cells were thawed and cultured in RPMI 1640 containing 10% fetal calf serum for 72 hours. Monocyte isolation and DC differentiation were carried out as described in the previous section to yield “immature DCs.” On day 7, some of the DCs were matured (mDCs) for 3 days with monocyte-conditioned medium prepared as previously described.22 RNA was isolated using Trizol reagent (Invitrogen, Carlsbad, CA). RNA labeling and Affymetrix GeneChip hybridizations were done by the Australian Genome Research Facility. Briefly, 100 ng total RNA was amplified using T7-oligo dT and the Megascript T7 kit (Ambion, Austin, TX) following the Affymetrix manual (701725 rev5). A second round of cDNA synthesis was performed using some or all of the first round amplified RNA as indicated. Amplified RNA was biotin-labeled using the GeneChip IVT Labeling Kit; 15μg of labeled RNA was then fragmented as described in the Affymetrix manual (701725 rev5). The Human Genome U133 Plus 2.0 array slides arrays were hybridized overnight, washed as described in the Affymetrix manual (701725 rev5), and scanned using a GeneChip scanner 3000 (Affymetrix). For analysis of the Affymetrix GeneChips, the intensities for each probe set were normalized and summarized using the Robust Multi-array Analysis algorithm.23 Differential expression was assessed using empirical Bayes moderated t-statistics from the LIMMA package.24 Statistical analysis was performed by Ken Simpson. Further data analysis was performed using Microarray Data Analysis System, version 2.19 (MIDAS 2.19), and Multi Experiment Viewer, version 4.0 (MEV 4.0). The data obtained for the microarray analyses are deposited at http://biomedicum.ut.ee/mp/research/ and can be accessed under “Gene expression profile of APCED monocytes and dendritic cells.”
Real-time RT-PCR
RNA was extracted from the cell pellets using the RNeasy Micro Kit (QIAGEN, Hilden, Germany) with DNase treatment according to the manufacturer's instructions, or Trizol reagent (Invitrogen). Total RNA was reverse-transcribed to cDNA using SupersScript III (Invitrogen), 10 mM dNTP Mix, oligo(dT)18 primer, and RiboLock RNase inhibitor (Fermentas, Vilnius, Lithuania). The cDNA obtained was used for real-time quantitative polymerase chain reaction (RT-PCR) using ABI Prism 7900 sequence Detection System (Applied Biosystems, Foster City, CA) and qPCR SYBR Green Core Kit (Eurogentec, Seraing, Belgium) according to the manufacturer's instructions with 2 mM MgCl2. SYBR Green fluorescence was measured after each extension step, and the specificity of amplification was evaluated by melting curve analysis. The relative gene expression levels were calculated using the comparative Ct (ΔΔCt) method (according to Applied Biosystems), where the relative expression is calculated as 2−ΔΔCt, where Ct represents the threshold cycle. Every sample was run in 3 parallel reactions. Primers for all target genes and housekeeping β-actin (ACTb) gene were designed to span exon boundaries in all multiexon genes with Primer Express (Applied Biosystems) and are listed in Table S1 (available on the Blood website; see the Supplemental Materials link at the top of the online article).
Cytokine measurement
IP-10 (CXCL10) Duo Elisa kit and Quantikine HS human IL-6 ELISA kit (both from R&D Systems) were used to detect their concentrations in patient and control sera. Human Th1/Th2 Cytokine Kit (Cytometric Bead Array, BD Biosciences) was used according to the manufacturer's instructions to measure IFN-γ, tumor necrosis factor (TNF)-α, IL-10, IL-5, IL-4, and IL-2 concentrations in patient and control serum samples with the help of FACSCalibur.
STAT1 phosphorylation
Normal human PBMCs or U937 monocytic cell line were used. PBMCs were rested for 2 hours in the medium described in the second section and U937 cells were kept in serum-free medium for 12 hours. The cells were pelleted 2 × 105 per tube and resuspended in 100 μL of medium with the indicated concentrations of recombinant human IFN-α2 (PBL Biomedical Laboratories, Piscataway, NJ) and various concentrations of test sera. After 15 minutes at 37°C and 5% CO2, the cells were fixed immediately with an equal volume of Cytofix buffer and permeabilized in cold Perm Buffer III for 30 minutes on ice. The cells were stained with 5 μL anti–phospho-STAT1 (Y701)-Alexa Fluor 488 at room temperature for 1 hour, washed, and analyzed using FACSCalibur. The mean Alexa Fluor 488-fluorescence intensity (MFI) of the cells was analyzed using CELLQuest software (everything from BD Biosciences). To detect serum IFN-α activity, the cells were incubated in 50% serum for 15 minutes. In some tubes, blocking IFN-α antibody at 10 μg/mL (clone 9D3, Abcam, Cambridge, United Kingdom) or isotype control antibody (BD Biosciences) was added. Data were expressed as MFI units (MFI units = MFIt − MFIb) where MFIt represents value for test sample and MFIb value for background staining.
Statistical analysis
Statistical analysis was done with the help of GraphPad Prism software (GraphPad Software, San Diego, CA). The mean values of different groups were compared using t test in the case of gene expression data and Mann-Whitney test in the case of cytokine concentrations. Bonferroni correction was used for multiple comparisons.
Results
Interferon-regulated gene expression is decreased in APECED blood cells
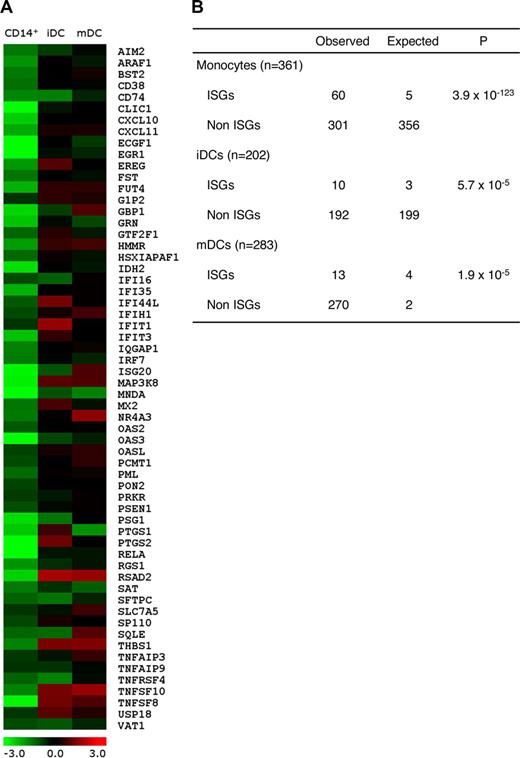
High-titer anti–type I IFN autoantibodies are found in nearly 100% of AIRE-mutant APECED patients.10,19,25 They neutralize the antiviral actions of most type I IFNs, especially IFN-ω, but IFN-β less frequently and the type II IFN-γ almost never. To assess their potential effects on ISG expression patterns, we performed Affymetrix Human Genome U133 Plus 2.0 Array analyses on monocytes and monocyte-derived DCs. Readily isolated in large numbers from APECED and control subjects' blood, these cells are known type I IFN producers and responders. After normalization, the genes showing more than 1.5-fold changes in each of 2 experiments were considered to be differentially expressed. Using literature surveys, we selected a total of 285 genes (Table S2) that are up-regulated by IFNs.26,27 Strikingly, 61 of these genes were down-regulated for at least 1.5-fold (Figure 1A), and the number of down-regulated ISGs was highly significantly changed in APECED CD14+ monocytes compared with healthy controls (P = 3.9 × 10−123; Figure 1B). Notably, however, the differences (and their significances) waned as the monocytes matured in culture into immature dendritic cells (iDCs; P = 5.7 × 10−5; Figure 1B) and mDCs (P = 1.9 × 10−5; Figure 1B) as visualized in Figure 1A.
Microarray analysis of APECED and control monocytes and monocyte-derived DCs. (A) The gene expression profiles (log2 of fold change between APECED patient A8 and control 2) of 61 differentially expressed ISGs in monocytes, iDCs, and mDCs. Most of the ISGs lost their differential expression by the iDC stage. (B) The differential expression of ISGs in APECED patient monocytes, iDCs, and mDCs. n indicates the total number of genes down-regulated 1.5-fold in each microarray data set. ISGs and non-ISGs indicate the number of genes, respectively, characterized or not characterized as ISGs. The expected numbers of ISGs and non-ISGs were calculated assuming the random selection. χ2 test was used to calculate the P values to determine whether the deviation is significant.
Microarray analysis of APECED and control monocytes and monocyte-derived DCs. (A) The gene expression profiles (log2 of fold change between APECED patient A8 and control 2) of 61 differentially expressed ISGs in monocytes, iDCs, and mDCs. Most of the ISGs lost their differential expression by the iDC stage. (B) The differential expression of ISGs in APECED patient monocytes, iDCs, and mDCs. n indicates the total number of genes down-regulated 1.5-fold in each microarray data set. ISGs and non-ISGs indicate the number of genes, respectively, characterized or not characterized as ISGs. The expected numbers of ISGs and non-ISGs were calculated assuming the random selection. χ2 test was used to calculate the P values to determine whether the deviation is significant.
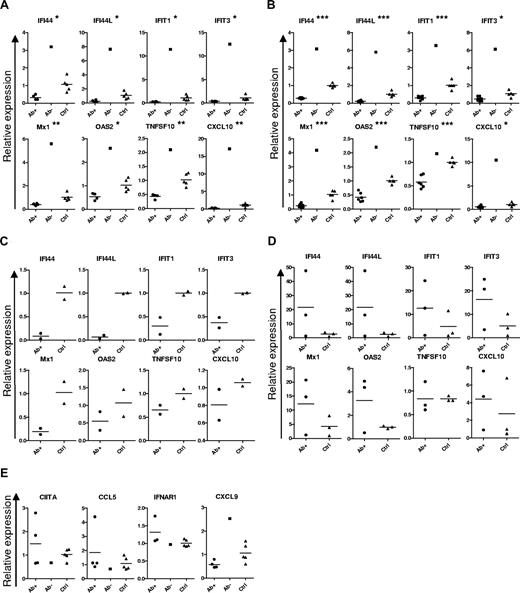
To confirm this ISG down-regulation independently in more APECED patients, we next analyzed the expression of some well-known ISGs that showed reduced expression in our array experiments, using RT-PCR to quantitate transcripts in PBMCs freshly isolated ex vivo. The down-regulation clearly correlated with the presence of neutralizing autoantibodies specific for the IFN-α because it was evident in all the APECED patients with high titers of autoantibodies against several IFN-α subtypes (Figure 2A). In contrast, expression of ISGs was strongly increased in one Norwegian APECED patient with neutralizing autoantibodies against IFN-ω only (patient A3, Table 2; Figure 2A). Furthermore, patient A2 with low titers of anti–IFN-α antibodies showed the highest ISG expression levels among APECED patients with neutralizing autoantibodies against IFN-α for 7 of the 8 ISGs tested (data not shown).
Decreased expression of ISGs in APECED cell populations. Expression of ISGs (A) and other genes (E) in freshly isolated PBMCs from IFN-α antibody-positive (Ab+, 4 cases) and -negative (Ab−, 1 case) APECED patients and healthy controls (Ctrl, 5 cases). (C) Expression of ISGs in freshly isolated plasmacytoid DCs (2 cases in each group). (B) Expression of ISGs in freshly isolated monocytes from IFN-α antibody-positive (6 cases) and -negative (1 case) APECED patients and healthy controls (7 cases). (D) Expression of ISGs in monocyte-derived DCs from APECED patients and healthy controls (3 cases in each group). *P < .05, **P < .001, ***P < .001, comparison of the Ab+ APECED patient and control blood cells. Bars represent group averages.
Decreased expression of ISGs in APECED cell populations. Expression of ISGs (A) and other genes (E) in freshly isolated PBMCs from IFN-α antibody-positive (Ab+, 4 cases) and -negative (Ab−, 1 case) APECED patients and healthy controls (Ctrl, 5 cases). (C) Expression of ISGs in freshly isolated plasmacytoid DCs (2 cases in each group). (B) Expression of ISGs in freshly isolated monocytes from IFN-α antibody-positive (6 cases) and -negative (1 case) APECED patients and healthy controls (7 cases). (D) Expression of ISGs in monocyte-derived DCs from APECED patients and healthy controls (3 cases in each group). *P < .05, **P < .001, ***P < .001, comparison of the Ab+ APECED patient and control blood cells. Bars represent group averages.
To identify the cell types affected, we then analyzed freshly isolated monocytes and plasmacytoid DCs. Notably, both cell types also showed similar down-regulation of the same ISGs (Figure 2B,C). Moreover, the down-regulated ISG pattern had normalized in monocyte-derived DCs that had differentiated for 6 days in culture with fetal calf serum (Figures 1A,B, 2D). In contrast with the ISGs, we saw no differences between the groups in the expression patterns of genes that are selectively (CIITA) or preferentially (CXCL9) regulated by IFN-γ; nor for CCL5, a chemokine induced more by proinflammatory cytokines than type I IFNs, or IFNAR1 (Figure 2E).
Taken together, these results show clearly decreased expression of all the classic ISGs tested in all the APECED patients with neutralizing antibodies against IFN-α, whereas, in one patient who has only anti–IFN-ω autoantibodies, it appeared to be strongly enhanced.
APECED sera with anti–IFN-α inhibit ISG expression
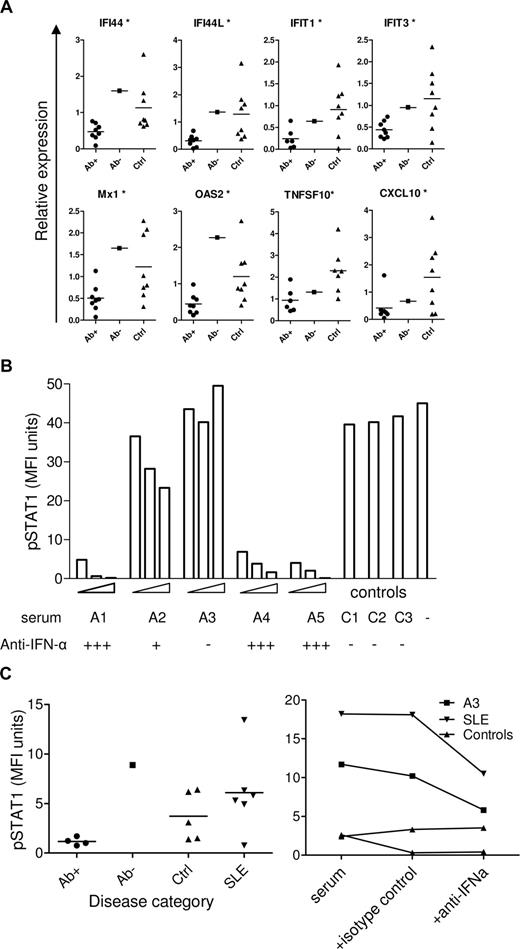
We next tested for acute effects of the neutralizing autoantibodies after incubating monocytes from healthy donors in medium containing 20% autologous plasma and 2% APECED or control sera for 18 hours. Expression of all the ISGs tested was significantly down-regulated by all the APECED sera that contained anti–IFN-α neutralizing autoantibodies, but not by the one specific for IFN-ω (Figure 3A). If fetal calf serum was used instead of autologous serum, no differences in ISG expression were seen in monocytes cultured in the presence of APECED or control sera (data not shown). Evidently, human plasma contains low levels of type I IFNs that can be blocked by the patients' neutralizing autoantibodies.
Effect of APECED sera on ISG expression and STAT1 phosphorylation. (A) Expression of ISGs in control monocytes incubated in 20% autologous sera with the addition of 2% APECED sera-positive (Ab+) or -negative (Ab−) for IFN-α antibodies, or with control serum (Ctrl) for 18 hours. *P < .05 between IFN-α antibody–positive APECED patients and healthy controls. (B) U937 cells were treated with 1000 U/mL IFN-α for 15 minutes or with the same concentration of IFN-α preincubated with 2%, 5%, or 10% of APECED (A1-A5) or 10% of control (C1-C3) sera, stained for intacellular pSTAT1 and measured by flow cytometry. (C) Control PBMCs were incubated with 50% of serum samples from APECED patients positive (Ab+) or negative (Ab−) for IFN-α antibodies, healthy controls (Ctrl), or SLE patients for 15 minutes and stained for intracellular pSTAT1 to test for IFN activity in the sera (left panel). Control PBMCs were incubated with 50% of serum samples from an APECED patient negative for IFN-α autoantibodies (A3), an SLE patient with IFN activity, or two healthy controls for 15 minutes with or without the addition of neutralizing anti–IFN-α antibody or isotype control antibody as indicated, and stained for intracellular pSTAT1 (right panel).
Effect of APECED sera on ISG expression and STAT1 phosphorylation. (A) Expression of ISGs in control monocytes incubated in 20% autologous sera with the addition of 2% APECED sera-positive (Ab+) or -negative (Ab−) for IFN-α antibodies, or with control serum (Ctrl) for 18 hours. *P < .05 between IFN-α antibody–positive APECED patients and healthy controls. (B) U937 cells were treated with 1000 U/mL IFN-α for 15 minutes or with the same concentration of IFN-α preincubated with 2%, 5%, or 10% of APECED (A1-A5) or 10% of control (C1-C3) sera, stained for intacellular pSTAT1 and measured by flow cytometry. (C) Control PBMCs were incubated with 50% of serum samples from APECED patients positive (Ab+) or negative (Ab−) for IFN-α antibodies, healthy controls (Ctrl), or SLE patients for 15 minutes and stained for intracellular pSTAT1 to test for IFN activity in the sera (left panel). Control PBMCs were incubated with 50% of serum samples from an APECED patient negative for IFN-α autoantibodies (A3), an SLE patient with IFN activity, or two healthy controls for 15 minutes with or without the addition of neutralizing anti–IFN-α antibody or isotype control antibody as indicated, and stained for intracellular pSTAT1 (right panel).
To test whether these anti–IFN-α autoantibodies also inhibit IFN signaling, we assessed phosphorylation of STAT1 protein, a crucial early event after stimulation of the IFN receptor complex. Cells treated with IFN-α showed significant phosphorylation of STAT1 protein that was again inhibited, indeed, down to baseline levels, but only by APECED sera containing high titers of neutralizing autoantibodies to IFN-α (Figure 3B). By contrast, the IFN-ω–specific serum A3, and another from one SLE patient, induced STAT1 phosphorylation; that induction was specifically neutralized by anti–IFN-α antibody (Figure 3C). Together, the down-regulation of ISG expression and of STAT1 phosphorylation in the presence of APECED sera shows that the changes observed in the APECED cells are the result of autoantibody neutralization of IFN-α.
APECED patients' plasmacytoid DCs and monocyte-derived DCs express normal levels of type I IFNs
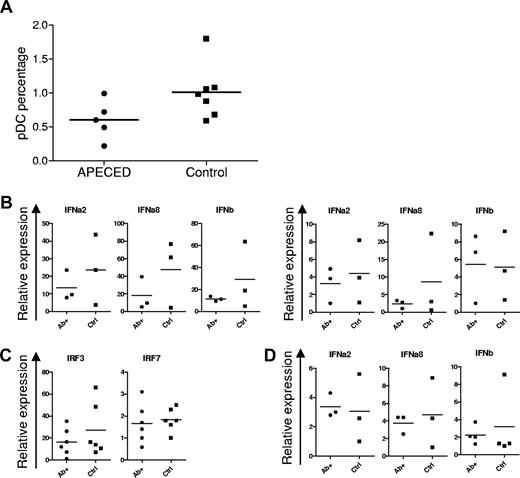
In response to viral stimulation, plasmacytoid DCs produce 10- to 100-fold more type I IFNs than monocytes. When PBMCs were stained for BDCA-2, a C-type lectin receptor that is specific for plasmacytoid DCs, their frequencies were slightly lower in APECED patients than in healthy controls, but not significantly (P = .079, Figure 4A).
Normal plasmacytoid DC numbers and dendritic cell IFN production in APECED. (A) Percentages of plasmacytoid DCs (pDCs) among PBMCs in APECED and control groups (P = .079, t test). (B) Expression of type I IFN genes after plasmacytoid DC stimulation for 6 hours with live influenza virus (left panel) or 2.5 μM CpG (right panel) in IFN-α–positive APECED (Ab+) or control persons (Ctrl). (C) IRF3 and IRF7 expression in stimulated plasmacytoid DCs from APECED or control individuals. (D) Type I IFN gene expression was studied after 18 hours of stimulation of monocyte-derived DCs from APECED patients (Ab+) and healthy controls (Ctrl) with 25 μg/mL of poly(I:C).
Normal plasmacytoid DC numbers and dendritic cell IFN production in APECED. (A) Percentages of plasmacytoid DCs (pDCs) among PBMCs in APECED and control groups (P = .079, t test). (B) Expression of type I IFN genes after plasmacytoid DC stimulation for 6 hours with live influenza virus (left panel) or 2.5 μM CpG (right panel) in IFN-α–positive APECED (Ab+) or control persons (Ctrl). (C) IRF3 and IRF7 expression in stimulated plasmacytoid DCs from APECED or control individuals. (D) Type I IFN gene expression was studied after 18 hours of stimulation of monocyte-derived DCs from APECED patients (Ab+) and healthy controls (Ctrl) with 25 μg/mL of poly(I:C).
To assess the potential of these plasmacytoid DCs to autoimmunize against IFNs in APECED, we next measured their IRF and IFN expression after short-term stimulation with influenza virus or CpG. We found no significant differences in transcript levels of their IFN-α2, IFN-α8, or IFN-β (Figure 4B) or of interferon-regulatory factors IRF3 and IRF7 (Figure 4C): indeed, the levels were even slightly lower in APECED than control plasmacytoid DCs. Because myeloid DCs also contribute significantly to the overall production of type I IFNs, we stimulated (precultured) monocyte-derived DCs for 18 hours with poly(I:C). Again, however, no differences were seen in expression of these type I IFN genes (Figure 3D).
In conclusion, the numbers of plasmacytoid DCs and levels of type I IFN mRNA expression by dendritic cells were comparable in APECED patients and healthy controls, which argues against any inherent hyperactivation in circulating APECED DCs that could lead to the breakdown of tolerance to type I IFNs.
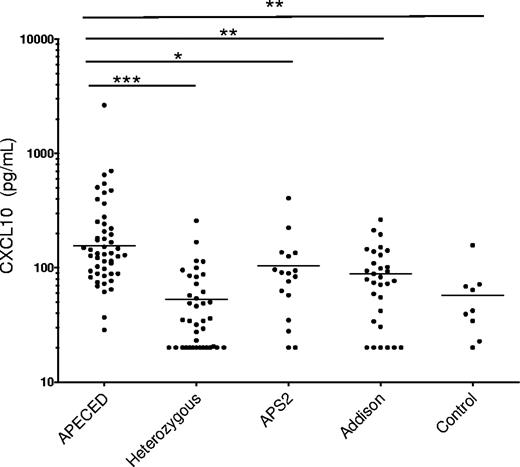
Elevation of serum CXCL10 levels in APECED patients
There are several recent reports of elevated levels of the pro-inflammatory chemokine CXCL10 in endocrine autoimmune diseases.28-30 Our studies here indicate that the neutralizing autoantibodies to IFN-α decrease the expression of CXCL10 in blood cells from APECED patients (Figure 2A-C). This prompted us to measure their serum CXCL10 levels; they were assessed by quantitative immunoassay in serum samples available from 49 APECED patients, 39 unaffected AIRE heterozygous relatives, and 9 healthy controls. In contrast with the decreased mRNA levels in blood cells, the APECED patients had significantly higher serum CXCL10 protein levels than the unaffected relatives (Figure 5, P < .001). Levels were even higher than in patients with isolated Addison's disease or autoimmune polyendocrine syndrome type 2 (APS2; P < .01 and P < .05, respectively). In the APECED patients, the CXCL10 levels did not correlate with the exact AIRE mutations, disease-onset ages, or numbers or types of clinical manifestations. Although the values spanned a wide range in each group, nearly all APECED patients were above the medians of the controls or relatives but did vary substantially in serial samples from the same individual Norwegian patients (data not shown).
CXCL10 levels in APECED and control sera. CXCL10 levels are significantly higher in APECED than in unaffected AIRE-heterozygous relatives, patients with APS2, Addison disease, or healthy controls. Geometric means of groups are indicated on plots. *P < .05, **P < .01, ***P < .001.
CXCL10 levels in APECED and control sera. CXCL10 levels are significantly higher in APECED than in unaffected AIRE-heterozygous relatives, patients with APS2, Addison disease, or healthy controls. Geometric means of groups are indicated on plots. *P < .05, **P < .01, ***P < .001.
We next investigated serum levels of proinflammatory cytokines in APECED patients and healthy controls. As with CXCL10, we observed a tendency to higher levels of IL-6 (P = .053) and TNF-α (P < .05) in APECED than in healthy controls (Table S3). However, the levels of IFN-γ, IL-10, IL-5, IL-4, and IL-2 showed no significant changes (Table S3 and data not shown).
Discussion
Our experiments clearly demonstrate down-regulated ISG expression in APECED patients' PBMCs. In sharp contrast, the expression of ISGs is higher in SLE patients, which is consistent with their well-known elevations in serum IFN-α levels.2,5,7-9,31 The lower expression of ISGs in APECED patients is apparently caused by neutralizing autoantibodies to the IFN-α rather than IFN-β or IFN-ω. Moreover, anti–IFN-α antibody–containing sera clearly down-regulated ISG expression in control monocytes ex vivo and blocked the key IFN-induced early signaling event, STAT1 phosphorylation. Importantly, these effects were reversible; after maturation in fetal calf serum (instead of APECED plasma), these patients' monocyte-derived DCs showed normal expression of ISGs. We propose that deprivation of the normal low levels of circulating IFNs2,32-34 by the neutralizing anti–IFN-α antibodies induces a state of type I IFN underactivity that can readily be reversed in culture.
Interestingly, we found markedly elevated levels of the IFN-induced chemokine CXCL10 in APECED sera, averaging approximately 5-fold higher than in healthy controls. In apparent contrast, its mRNA expression was decreased in blood cells. Approximately 5-fold higher serum CXCL10 protein levels have also been described in SLE, with similarly poor correlations with gene expression in blood cells.35 Modest increases in CXCL10 have been described in other organ-specific diseases, such as Hashimoto thyroiditis,28 Graves disease,29 myasthenia gravis,36 and Addison disease30 and also during type I IFN treatment.37,38 In APECED, the autoimmune processes in endocrine tissues are most probably accompanied by IFN-γ production that is a strong inducer of CXCL10. We propose that chemokines measured in serum are derived not from blood cells but rather from the extravascular target tissues, where they are responding to inflammatory stimuli and thus chemoattracting immune cells out of the circulation. That might explain the slightly decreased percentages of plasmacytoid DCs in APECED blood (Figure 4A), which have also been noted in SLE.39
Type I IFNs are produced in large quantities during many acute viral infections. In physiologic situations, their actions may largely be localized to infected tissues and operate mainly via autocrine and paracrine stimulation. Although the autoantibodies can effectively neutralize IFNs in the bloodstream, they may not reach sufficient levels to do so in the tissues, which might help explain the surprising rarity of viral infections in APECED patients despite their decreased expression of ISGs in blood cells. In addition, there may be compensation by IFN-β and/or IFN-λ, which are neutralized much less in most patients.
The consistently increased expression of ISGs in APECED patient A3, whose neutralizing autoantibodies only recognize IFN-ω (and not IFN-α), also hints at an underlying local overproduction of type I IFNs in APECED patients who are masked by the anti–IFN-α autoantibodies in the typical cases. Most ISGs are up-regulated by both type I IFNs and IFN-γ. The normal expression of CIITA, which is selectively up-regulated by IFN-γ, argues that it does not contribute substantially and thus implicates the IFN-α most strongly. The CIITA level was even lower in patient A3, and her serum did contain IFN-α because it evoked significant STAT1 phosphorylation in control cells and could be neutralized by anti–IFN-α antibodies. Notably, one side effect of treatment with IFN-α is thyroid autoimmunity.40 Interestingly, the one patient with detectable serum IFN-α and with neutralizing autoantibodies only against IFN-ω (patient A3) also has thyroiditis, which is otherwise rare among Norwegian APECED patients. Similarly, the other serologically similar patient (A2) had thyroid peroxidase autoantibodies.
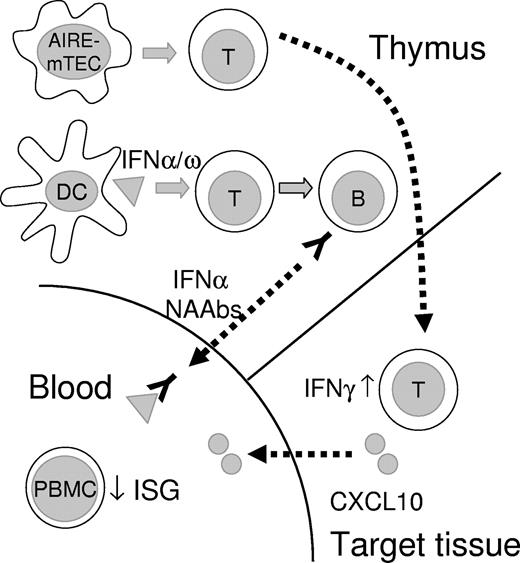
After virus- or TLR-stimulation, plasmacytoid DCs produce a mixture of type I IFNs, but predominantly IFN-α, whereas, after TLR 4 or TLR 3 stimulation, monocyte-derived DCs secrete mainly IFN-β,41 which is recognized infrequently (∼ 20%) by the neutralizing autoantibodies in APECED.10 Moreover, the type I IFNs show distinct actions; the chemokine-stimulating and DC-maturing properties of IFN-ω seem particularly restricted.42 This is consistent with the observed ISG up-regulation by the APECED sera with neutralizing autoantibodies against only IFN-ω, in contrast to ISG down-regulation by the APECED sera with autoantibodies to IFN-α (and IFN-ω). Although our results do not indicate any abnormal IFN overproduction in APECED peripheral DCs, the human thymus contains not only numerous DCs but also many plasmacytoid DCs, which can produce large amounts of type I IFNs. Moreover, they are predominantly located in the medulla and corticomedullary junction,43 where type I IFNs appear to be constitutively secreted.10 Type I IFN overproduction by thymic plasmacytoid DCs impairs thymic T-cell development.44 It is also involved in terminal differentiation and subsequent apoptosis of thymic epithelial cells,45 and so, interestingly, is wild-type AIRE.46 The coincidence of highly prevalent, high-titer IFN autoantibodies in patients with thymic epithelial tumors or genetic defects in AIRE suggests parallel mechanisms of autoimmunization in the APECED thymus and thymomas. Indeed, the neoplastic epithelial cells in thymomas almost always fail to express AIRE.47 We propose that aberrant cell death or some other so far unknown danger signal renders the APECED thymic environment prone to autoimmunize responsive T cells against locally abundant type I IFNs (Figure 6).
Schematic illustration of thymic and peripheral events in APECED. Tissue-restricted antigen-specific T cells are not deleted in APECED thymus because of the deficient function of AIRE in medullary thymic epithelial cells (mTEC), and escape to cause tissue damage in endocrine organs in APECED patients. We speculate that, because of aberrant cell death or alternative danger signal, human AIRE-deficient thymus (and thymomas) assume some functions of secondary lymphoid organs, with associated overproduction of type I IFNs by thymic DCs. These DCs are activated and present a broad spectrum of type I interferons to autoimmunize specific T and then B cells. The resulting high titer neutralizing antibodies inhibit ISG responses of PBMCs to basal circulating levels of type I IFNs. Autoreactive T cells infiltrating target endocrine tissues secrete IFN-γ. This induces the production of CXCL10 and further expansion of the adjacent infiltrates.
Schematic illustration of thymic and peripheral events in APECED. Tissue-restricted antigen-specific T cells are not deleted in APECED thymus because of the deficient function of AIRE in medullary thymic epithelial cells (mTEC), and escape to cause tissue damage in endocrine organs in APECED patients. We speculate that, because of aberrant cell death or alternative danger signal, human AIRE-deficient thymus (and thymomas) assume some functions of secondary lymphoid organs, with associated overproduction of type I IFNs by thymic DCs. These DCs are activated and present a broad spectrum of type I interferons to autoimmunize specific T and then B cells. The resulting high titer neutralizing antibodies inhibit ISG responses of PBMCs to basal circulating levels of type I IFNs. Autoreactive T cells infiltrating target endocrine tissues secrete IFN-γ. This induces the production of CXCL10 and further expansion of the adjacent infiltrates.
In conclusion, we show that ISGs are down-regulated in APECED patients' PBMCs by their autoantibodies against type I IFNs and demonstrate marked increase in serum CXCL10 levels in APECED. Our results suggest that AIRE deficiency causes increased production of IFNs, possibly through aberrant cell death in the APECED thymus,46 which results in highly specific autoantibodies to type I IFNs and down-regulation of ISGs in APECED blood cells.
The online version of this article contains a data supplement.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 USC section 1734.
Acknowledgments
The authors thank the patients and controls for so kindly providing the generous samples on which this study crucially depends; Dr Kylie E. Webster and Dr Ken Simpson for help in sample preparation and analysis, respectively, of the Affymetrix chips; Ulla Kiiskinen and Elisabeth Halvorsen for technical assistance; and Jim Robertson for influenza virus.
This work was supported by the Wellcome Trust Senior Fellowship grant, EU Framework program 6 (Thymaide and Euraps; P.P.), the European Regional Fund and Archimedes Foundation, the Estonian Science Foundation (grants 6663, 6514 and 7197; K.K., P.P., M.L.), Slovenian Agency for Research (J3-9663; K.T.P. and T.B.), National Health and Medical Research Council (NHMRC) fellowships (171601 and 461204), and National Health and Medical Research Council (NHMRC) program grants (257501, 264573 and 406700; H.S.S.).
Howard Hughes Funding
Authorship
Contribution: K.K., M.L., A.S.B.W., A. Meager, L.T., A. Murumägi, and H.S.S. performed experiments and analyzed data; K.K. and T.O. performed statistical analysis; A.S.B.W., E.S.H., K.L., K.T.P., T.B., A.L., O.K., A. Meloni, B.E.-L., N.K.M., J.P., K.J.E.K., and R.U. were responsible for collection of clinical material and data; and K.K., N.W., and P.P. designed the research and wrote the paper.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: Kai Kisand, Institute of General and Molecular Pathology, University of Tartu, Ravila Str 19, Tartu 50411, Estonia; e-mail: kai.kisand@ut.ee.