Granulocyte colony-stimulating factor receptor (GCSFR) signaling participates in the production of neutrophilic granulocytes during normal hematopoietic development, with a particularly important role during emergency hematopoiesis. This study describes the characterization of the zebrafish gcsf and gcsfr genes, which showed broad conservation and similar regulation to their mammalian counterparts. Morpholino-mediated knockdown of gcsfr and overexpression of gcsf revealed the presence of an anterior population of myeloid cells during primitive hematopoiesis that was dependent on GCSF/GCSFR for development and migration. This contrasted with a posterior domain that was largely independent of this pathway. Definitive myelopoiesis was also partially dependent on a functional GCSF/GCSFR pathway. Injection of bacterial lipopolysaccharide elicited significant induction of gcsf expression and emergency production of myeloid cells, which was abrogated by gcsfr knockdown. Collectively, these data demonstrate GCSF/GCSFR to be a conserved signaling system for facilitating the production of multiple myeloid cell lineages in both homeostatic and emergency conditions, as well as for early myeloid cell migration, establishing a useful experimental platform for further dissection of this pathway.
Introduction
Granulocyte colony-stimulating factor (GCSF) and its cognate receptor granulocyte colony-stimulating factor receptor (GCSFR) form part of the class I cytokine receptor signaling system, members of which play important roles in hematopoiesis.1,2 GCSF expression is normally very low, but during emergency conditions such as the presence of bacterial lipopolysaccharide (LPS), strong expression is induced in monocytes, fibroblasts, and endothelial cells.3,4 GCSFR is principally expressed in hematopoietic cells, including myeloid progenitors, neutrophils, and monocytes, as well as certain nonhematopoietic cells.5 Stimulation of cell-surface GCSFR leads to activation of downstream intracellular signaling cascades, including the JAK/STAT/SOCS pathway.6
The function of GCSF/GCSFR signaling in granulopoiesis is reflected by the severe neutropenia and reduced survival of neutrophils observed in both GCSF−/− and GCSFR−/− knockout mice, although both also showed granulocyte-macrophage progenitor (GMP) defects.7,8 Indeed, bone marrow from GCSFR−/− mice exhibited reduced capacity to repopulate GMPs compared with that from wild-type mice, suggesting a wider role for GCSF/GCSFR signaling at the GMP level.9 In addition, GCSF/GCSFR signaling in trans mediates effective mobilization of hematopoietic progenitor cells (HPCs) from the bone marrow.10 The importance of GCSF/GCSFR clinically is highlighted by the broad application of GCSF to the treatment of neutropenic conditions, as well as the association of GCSFR mutants with severe congenital neutropenia and its associated sequelae.11
Hematopoiesis is largely conserved between mammals and zebrafish, being divided into distinct primitive and definitive stages in each case. Primitive hematopoiesis in zebrafish is initiated within the lateral mesoderm that becomes split into separate anterior (rostral blood island [RBI]) and posterior (intermediate cell mass [ICM]) domains with distinct functions.12 Macrophages originate in the RBI then later migrate posteriorly, whereas erythrocytes and at least some granulocytes originate from the pICM.13 Definitive hematopoiesis commences in the ventral wall of the dorsal aorta, a region analogous to the mammalian aorta-gonad-mesonephros.12,13 It later relocates, first to the caudal hematopoietic tissue, analogous to mammalian fetal liver, and ultimately to the primordium of the kidney that continues to be the site of adult hematopoiesis in a manner analogous to the mammalian bone marrow.12,14,15 Hematopoietic correspondence in zebrafish extends to the presence of a multilineage myeloid compartment16 and a conserved response to bacterial infection.17
A range of studies have also highlighted the conservation of key hematopoietic transcription factors, such as scl, lmo2, gata1, ikaros, and spi1,18,,,–22 and functional markers, such as fms, lyz, lplastin, mpo, and mmp9 in zebrafish.23,,,–27 Increasingly the hematopoietic cytokine/cytokine receptor repertoire and its downstream signaling pathways are also being explored in this model, providing new insights into hematopoiesis.28,–30 In this study, orthologues for zebrafish gcsf and gscfr were extensively characterized, revealing broad conservation to their mammalian counterparts at the genomic, transcriptional regulation, and protein levels. Early detection of gcsfr expression was limited to the RBI, with morpholino-mediated targeting of gcsfr significantly reducing general myelopoiesis, with the greatest effects in the RBI where it also reduced myeloid cell migration. Injection of gcsf mRNA increased myelopoiesis within the anterior domain in a gcsfr-dependent manner. Ablation of gcsfr translation also reduced definitive myelopoiesis, which in contrast was augmented by enforced gcsf expression. Finally, injection of LPS induced gcsf expression and emergency myelopoiesis, again in a gcsfr-dependent manner. This study demonstrates a functional GCSF/GCSFR signaling program participating widely in zebrafish myelopoiesis, providing a useful experimental platform for further studies.
Methods
Fish maintenance
Wild-type or Tg(lyz::EGFP) fish were manually spawned and embryos collected at appropriate stages31 for manipulation or analysis. All experiments were performed under appropriate Deakin University and University of Auckland Animal Welfare Committee guidelines.
Functional assays
To simulate an emergency hematopoietic response, embryos were anesthetized with 0.1 mg/mL benzocaine and injected with 5 μg/mL LPS into the venous return at 48 hours postfertilization (hpf) and embryos fixed at 56 hpf. Wound-healing assays were performed on 3-day postfertilization (dpf) Tg(lyz::EGFP) embryos, as previously described.24 To analyze phagocytosis, Molecular Probes pHrodo Escherichia coli BioParticle conjugates (Invitrogen), were resuspended at 200 μg/mL in PBS/20 mM HEPES (pH 7.4) and microinjected into the circulation of anaesthetized 2-dpf Tg(lyz::EGFP) embryos, which were incubated for 30 minutes in the dark before confocal imaging. Fluorescence-activated cell sorting on labeled cells from 4-dpf Tg(lyz::EGFP) larvae was performed as described,32 with minor modifications.
In silico data mining and analysis
Sequence searching was performed using BLAST (http://www.ncbi.nlm.nih.gov/blast/)33 with Genomescan (Massachusetts Institute of Technology, Cambridge, MA) used to predict protein coding sequences from genomic DNA.34 Subsequent multiple sequence alignments were performed using AlignX 9 (Invitrogen) and ClustalX 1.83.35,36 Phylogenetic analysis was performed using NJ-plot (Pôle Bioinformatique Lyonnais, http://pbil.univ-lyon1.fr/).37 Synteny analysis was performed using Ensembl (http://www.ensembl.org)38 with version 7 of the zebrafish assembly (Zv7). Superfamily HMM was used to analyze protein domains,39,40 and SWISS-MODEL was used to predict structure.41,42 The nomenclature for the zebrafish genes followed the conventions of the Zebrafish Information Network (http://www.zfin.org). Genes were submitted to EMBL43 using Webin (European Bioinformatics Institute, http://www.ebi.ac.uk/embl/Submission/webin.html) with the following accession numbers: gcsf (FM174388) and gcsfr (AM157796).
RT-PCR
RNA isolation and reverse transcription–polymerase chain reaction (RT-PCR) were performed essentially as described,29 using the following primers: gcsf forward: 5′-CTACTGGAGTCTGGTGATTGTGC, gcsf reverse: 5′-AGACAGCGACATTTACACAGAGC, gcsfr forward: 5′-GGGAATGTCTACACGCCTGC, gcsfr reverse: 5′-CTGTGCTTCTGTAATCTGTCCAATG, ef1α forward: 5′-AAGGAGAAGATCGACCGTCG, ef1α reverse: 5′-TGATTGAGGGAAATTCACTTGG, lyz forward: 5′-TCTCCATTGGCAACTATGTGTGC, and lyz reverse: 5′-CTTTGTTTGCGCTGCTCACAGC. For 5′RACE, the following primers were used according to the manufacturer's protocol (Invitrogen): gcsfr 1: 5′-AACACTTTGCCTTTGGTC, gcsfr 2: 5′-GATGGAGCAGGACACAGAAACC, and gcsfr 3: 5′-GAGATCCAGCCAAAACCACAG. Cloning and sequencing of relevant RT-PCR products were performed as previously described.29
Morpholino and mRNA injection
Two antisense splice-site targeting morpholinos (Gene Tools, Philomath, OR) were used, gcsfrMo1: 5′-TTTGTCTTTACAGATCCGCCAGTTC; gcsfrMo2: 5′-CAGTTAAATGACCCATTTACCTGTG; as well as a scrambled morpholino (scramMo): 5′-CCTCTTACCTCAGTTACAATTTATA control. Aliquots of morpholino (1 mM) or in vitro–transcribed gcsf mRNA (100 ng/μL) and 1% (wt/vol) phenol red in Danieau solution were injected into embryos between the 1-cell and 4-cell developmental stages.
Whole-embryo staining and analysis
Whole-mount in situ hybridization (WISH) was performed as described.27 A TUNEL apoptosis assay was performed using the In Situ Cell Death Detection Kit (Roche, Indianapolis, IN), following the manufacturer's protocol. The O-dianisidine (Sigma-Aldrich, St Louis, MO) staining was performed as described previously.25 Whole-mount myeloperoxidase staining was performed using the manufacturer's protocol (Sigma-Aldrich), except embryos were fixed in 4% PFA and the incubation time was 5 minutes. Statistical analysis of cell counts after WISH were performed using GraphPad Prism software (version 4; San Diego, CA). The unpaired independent t test was used to determine the statistical significance of various treatments, typically with sample populations of approximately 20 to 30 embryos obtained from several independent manipulations.
Photography
A SZX-ILLK200 microscope (Olympus, Melville, NY) was used to visualize embryos, and digital images recorded using DP Controller 2.1.1.183 software and DP90 camera (Olympus). Stage lighting was provided using a Microlight 150 (Fiberoptic Lightguides; Fiberoptic Components, Sterling, MA).
Time-lapse confocal microscopy
Manipulated Tg(lyz::EGFP) and Tg(lyz::EGFP_CAAX) embryos were anaesthetized using tricaine44 and imaged using an Olympus FV1000 confocal laser scanning microscope equipped with an incubation chamber kept at 28.5°C, as described.23 Videos were generated using ImageJ64 (National Institutes of Health, Bethesda, MD),45 whereas isosurface reconstruction and subsequent 4D analysis was performed using Imaris 5.7.2 (Bitplane, St Paul, MN) image analysis software.
Results
Analysis of the putative zebrafish gcsfr gene and its encoded protein
As part of a comprehensive analysis of class I cytokine receptor evolution, a single putative GCSFR orthologue was previously identified in zebrafish on the basis of sequence homology and conserved synteny.29 The 5′-untranslated region (UTR) of the zebrafish gcsfr was characterized and putative transcription start sites were identified using 5′-RACE and comparison with corresponding genomic sequences extracted from relevant databases. In silico analysis of the upstream gcsfr promoter region indicated the presence of putative transcription factor binding sites for HOXA5, SPI1, and members of the C/EBP family, which were also conserved in the human GCSFR promoter (Figure 1A). Importantly, HOXA5, C/EBPα, and SPI1 have each been shown to facilitate myelopoiesis,46,47 with both C/EBPα and SPI1 capable of regulating mammalian GCSFR promoters.48
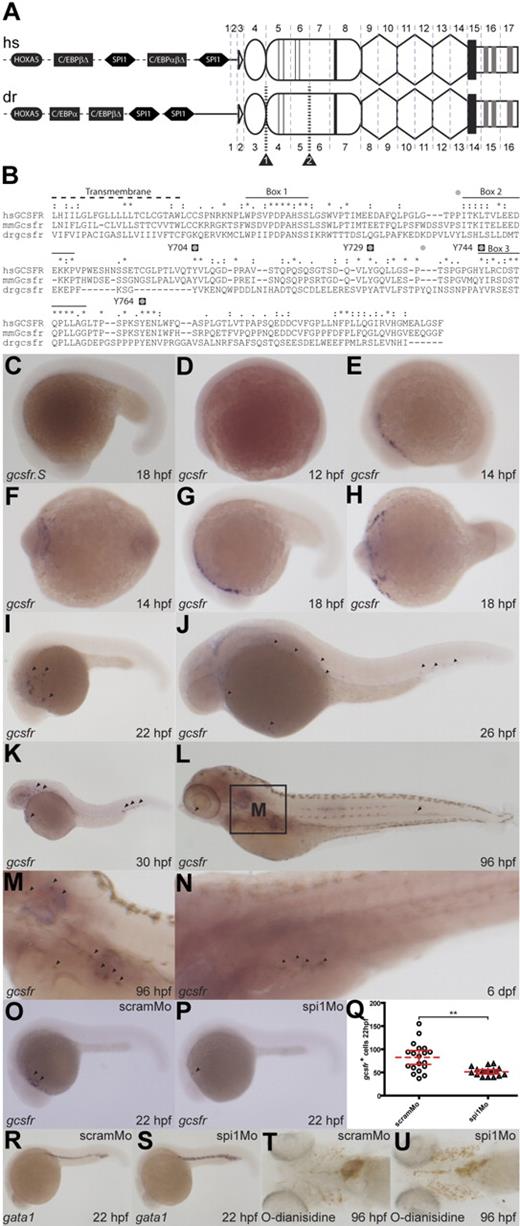
Characterization of zebrafish gcsfr. (A) Analysis of the zebrafish gcsfr gene locus. Schematic representation of human (hs) GCSFR and zebrafish (dr) gcsfr genes: showing conserved transcription factor binding sites for HOXA5 (black ovals), SPI1 (black hexagons), and members of the C/EBP family (black rectangles) within the promoter region (dashed line) upstream of the 5′UTR (black line). The domain structure of the encoded protein is superimposed onto the respective splicing pattern: leader sequence (white triangle), Ig-like domain (oval), CHD (round rectangle), with thin lines indicating cysteines and thick lines indicating WSXWS motif, FBN-like domains (hexagons), transmembrane domain (black rectangle), and intracellular domain (white rectangle), containing boxes 1, 2, and 3 (gray rectangles). Splice sites are shown as vertical gray dashed lines, with exons numbered, whereas splice site–targeted morpholinos are indicated by striped black lines and labeled black triangles. (B) Alignment of human (hs), mouse (mm), and zebrafish (dr) GCSFR intracellular domains. Identical (*), strongly conserved (:), and weakly conserved (.) residues are displayed. Conserved motifs are indicated: Transmembrane (dotted line), Boxes 1, 2, and 3 (black lines), mammalian intracellular tyrosines (black squares labeled with human residue numbers), and zebrafish intracellular tyrosines (gray circles). (C-N) Expression of zebrafish gcsfr during development. Whole-mount in situ hybridization of staged wild-type embryos with sense (C) or antisense (D-N) gcsfr probes at the times indicated. (O-Q) Role of spi1 in regulation of gcsfr-expressing cells. Embryos injected with scramMo (O,R,T) or spi1Mo (P,S,U) were probed with antisense gcsfr (O,P) and positive cells enumerated (Q; dashed red line indicates mean; red lines, 95% confidence interval; **P < .01 level of statistical significance), or with antisense gata1 (R,S), or stained with O-dianisidine (T,U).
Characterization of zebrafish gcsfr. (A) Analysis of the zebrafish gcsfr gene locus. Schematic representation of human (hs) GCSFR and zebrafish (dr) gcsfr genes: showing conserved transcription factor binding sites for HOXA5 (black ovals), SPI1 (black hexagons), and members of the C/EBP family (black rectangles) within the promoter region (dashed line) upstream of the 5′UTR (black line). The domain structure of the encoded protein is superimposed onto the respective splicing pattern: leader sequence (white triangle), Ig-like domain (oval), CHD (round rectangle), with thin lines indicating cysteines and thick lines indicating WSXWS motif, FBN-like domains (hexagons), transmembrane domain (black rectangle), and intracellular domain (white rectangle), containing boxes 1, 2, and 3 (gray rectangles). Splice sites are shown as vertical gray dashed lines, with exons numbered, whereas splice site–targeted morpholinos are indicated by striped black lines and labeled black triangles. (B) Alignment of human (hs), mouse (mm), and zebrafish (dr) GCSFR intracellular domains. Identical (*), strongly conserved (:), and weakly conserved (.) residues are displayed. Conserved motifs are indicated: Transmembrane (dotted line), Boxes 1, 2, and 3 (black lines), mammalian intracellular tyrosines (black squares labeled with human residue numbers), and zebrafish intracellular tyrosines (gray circles). (C-N) Expression of zebrafish gcsfr during development. Whole-mount in situ hybridization of staged wild-type embryos with sense (C) or antisense (D-N) gcsfr probes at the times indicated. (O-Q) Role of spi1 in regulation of gcsfr-expressing cells. Embryos injected with scramMo (O,R,T) or spi1Mo (P,S,U) were probed with antisense gcsfr (O,P) and positive cells enumerated (Q; dashed red line indicates mean; red lines, 95% confidence interval; **P < .01 level of statistical significance), or with antisense gata1 (R,S), or stained with O-dianisidine (T,U).
Comparison of cDNA and genomic sequences showed that zebrafish gcsfr was composed of 16 exons. Apart from an additional exon in the 5′-UTR of human GCSFR, the positions of splice sites were identical between zebrafish and human genes. Sequence analysis of human and zebrafish GCSFR proteins revealed conservation of important motifs and domains during evolution (Figure 1A). In the extracellular region, the immunoglobulin-like (Ig), cytokine receptor homology (CHD), and 3 fibronectin type III-like (FBN) domains were maintained, the signature WSXWS motif was present within the CHD, and 2 of the 4 cysteines were conserved. Analysis using SWISS-MODEL42 confirmed structural similarities between zebrafish and mammalian GCSFR proteins (Figure S1, available on the Blood website; see the Supplemental Materials link at the top of the online article).
Sequence alignment of the intracellular regions of zebrafish and mammalian GCSFR proteins revealed comparable hydrophobic transmembrane domains, as well as equivalent box 1, box 2, and box 3 sequences that are also conserved in other members of the GP130 family49 (Figure 1B). Conservation was particularly strong for box 1, the essential motif for Jak kinase docking, and the STQPLL internalization motif found within box 3.2,50 There were 6 intracellular tyrosines in zebrafish GCSFR (Y675, Y694, Y724, Y733, Y743, and Y765), 4 of which aligned with the intracellular tyrosines of human GCSFR (Y704, Y729, Y744, and Y764), including conservation of surrounding residues responsible for docking specificity of downstream signaling molecules. Collectively, these data suggest overall structural and functional conservation of the GCSFR between mammals and zebrafish.
The spatiotemporal expression of zebrafish gcsfr during embryogenesis was examined using WISH (Figure 1C-O). Transcripts for gcsfr were first observed at 14 hpf, although expression was limited to the RBI at this time (Figure 1E,F). The gcsfr+ cells subsequently migrated in a posterior manner over the yolk sac, particularly from 18 hpf until 22 hpf (Figure 1G-I), similar to that observed for several other zebrafish myeloid markers.16,51 The first gcsfr+ cells were detected in the pICM from about 26 hpf (Figure 1J), after the onset of circulation,12 with this population increasing from 30 hpf onward (Figure 1K), suggesting a second wave of production at this location. The gcsfr+ population subsequently decreased to several isolated cells, including within the developing pronephros at 96 hpf (Figure 1L,M) and 6 dpf (Figure 1N), a key site of definitive hematopoiesis at this time.
Mammalian GCSFR expression has previously been reported to be under the control of the transcription factor SPI1,48 which is highly conserved in zebrafish.18,51 Since binding sites for SPI1 were conserved in the zebrafish gcsfr promoter, and both spi118 and gcsfr exhibited similar anterior expression patterns in zebrafish embryos, the role of spi1 in regulating gcsfr+ cells was explored. Injection of a morpholino targeting a splice site within spi1 produced a phenotypically normal embryo. However, WISH analysis of spi1 morphants at 22 hpf with a gcsfr probe revealed significant down-regulation of gcsfr expression compared with scrambled morpholino–injected control embryos (P < .01), although it was not totally abolished (Figure 1O-Q). Consistent with another study,52 progenitor and mature erythroid populations at 22 hpf (Figure 1R,S) and 96 hpf (Figure 1T,U) were also increased.
Role of the zebrafish gcsfr in hematopoiesis
Mice lacking GCSFR,8,53 or patients constitutively expressing hypomorphic GCSFR mutants,11 are moderately to severely neutropenic. Moreover, mouse knockouts of either GCSF or GCSFR show defects in GMPs and impaired hematopoietic stem cell mobilization.7,53 Morpholino-mediated gene knockdown was used to investigate the role of zebrafish gcsfr. Injection of either of 2 independent splice site–targeting morpholinos, gcsfrMo1 and gcsfrMo2 (Figure 1A), produced embryos that were phenotypically identical to embryos injected with a control scrambled morpholino (scramMo; Figures 2, 3). RT-PCR performed using primers on either side of the splice sites targeted by the morpholinos provided evidence of robust blocking of splicing in each case (data not shown).
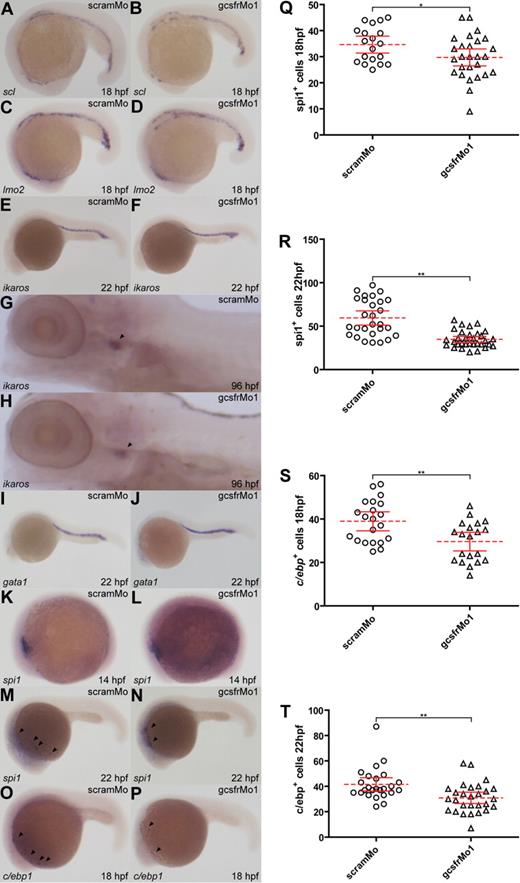
Analysis of early hematopoietic, lymphoid, and erythroid populations in gcsfr morphants. (A-P) WISH analysis of embryos injected with scrambled morpholino (scramMo) or zebrafish GCSFR morpholino 1 (gcsfrMo1) at various time points using antisense probes to specific hematopoietic marker genes, as indicated. (Q-T) The total number of spi1+ or c/ebp+ cells from embryos injected with either scramMo or gcsfrMo1 were counted, with mean (dashed red line), 95% confidence interval (red lines), and level of statistical significance (*P < .05; **P < .01) indicated.
Analysis of early hematopoietic, lymphoid, and erythroid populations in gcsfr morphants. (A-P) WISH analysis of embryos injected with scrambled morpholino (scramMo) or zebrafish GCSFR morpholino 1 (gcsfrMo1) at various time points using antisense probes to specific hematopoietic marker genes, as indicated. (Q-T) The total number of spi1+ or c/ebp+ cells from embryos injected with either scramMo or gcsfrMo1 were counted, with mean (dashed red line), 95% confidence interval (red lines), and level of statistical significance (*P < .05; **P < .01) indicated.
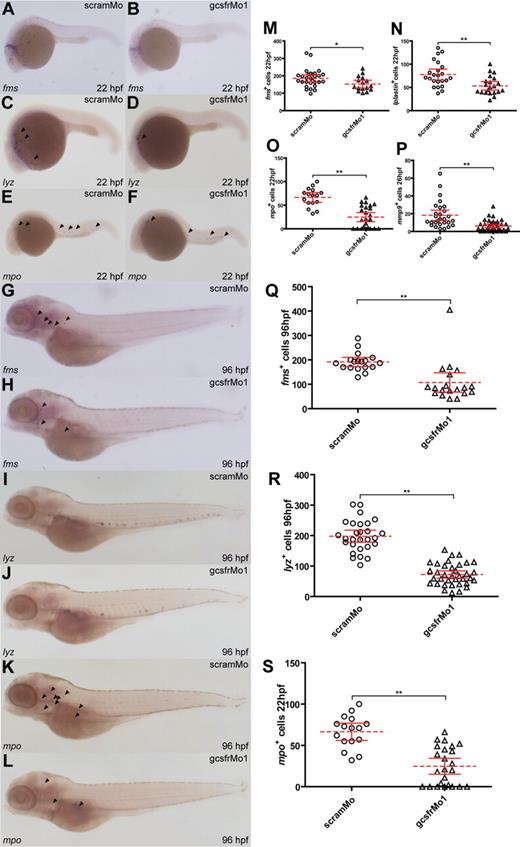
Analysis of myeloid cell populations in gcsfr morphants. (A-L) WISH analysis of embryos injected with scramMo or gcsfrMo1 at various time points using antisense probes to specific myeloid genes, as indicated. (M-S) The total number of cells positive for the indicated myeloid markers from embryos injected with either scramMo or gcsfrMo1 was counted, showing mean (dashed red line), 95% CI (solid red lines), and level of statistical significance (*P < .05; **P < .01).
Analysis of myeloid cell populations in gcsfr morphants. (A-L) WISH analysis of embryos injected with scramMo or gcsfrMo1 at various time points using antisense probes to specific myeloid genes, as indicated. (M-S) The total number of cells positive for the indicated myeloid markers from embryos injected with either scramMo or gcsfrMo1 was counted, showing mean (dashed red line), 95% CI (solid red lines), and level of statistical significance (*P < .05; **P < .01).
Hematopoietic stem cells (HSCs) are derived from the lateral plate mesoderm during zebrafish embryogenesis to commence the process of primitive hematopoiesis in the RBI and pICM from around 12 hpf.12 Given the early expression of gcsfr in the RBI, gcsfr morphants were analyzed with specific HSC makers. Neither scl nor lmo219,20 was affected by gcsfr knockdown at either 18 hpf (Figure 2A-D) or 22 hpf (data not shown). Similarly, expression of ikaros, which marks HSCs during primitive hematopoiesis at 22 hpf, or lymphoid progenitors during definitive hematopoiesis at 96 hpf,21 was unaffected in gcsfr morphants (Figure 2E-H). In addition, there was no overt difference in expression of the early erythroid marker, gata122 (Figure 2I,J). There was also no change in expression of the early myeloid marker spi118 at 14 hpf (Figure 2K,L). However, there was a significant decrease in spi1+ cells at 18 hpf (P < .05; Figure 2Q) that was sustained at 22 hpf (P < .01; Figure 2M,N,R). The spi1+ cells also appeared to be less dispersed posteriorly in gcsfr morphants. There was a similar significant decrease in cells expressing the early myeloid marker, c/ebp1,54 in gcsfr morphants at both 18 hpf (P < .01; Figure 2O,P,S) and 22 hpf (P < .01; Figure 2T).
Distinct populations of mature myeloid cells appear to originate at both anterior, RBI, and posterior, pICM, regions of the embryo.13 Cells expressing fms, which exclusively marks the monocyte/macrophage compartment in the anterior region during early hematopoiesis,26 were reduced both in number (P < .01) and relative dispersal in gcsfr morphants (Figure 3A,B,M). There was a similar reduction in the number and extent of dispersion for the pan-leukocyte markers, lyz24,55 (P < .03; Figure 3C,D) and lplastin26,56 (P < .01; Figure 3N). Finally, there was down-regulation of the number of cells expressing the neutrophil-specific markers, mpo25 (P < .01; Figure 3E,F,O) and mmp927 (P < .01; Figure 3P), with a significantly greater effect seen in the anterior domain. This collectively suggests that zebrafish gcsfr plays a broad role in myeloid cell production, with a possible secondary function in the migration of these cells.
The initiation of definitive hematopoiesis begins from approximately 24 to 48 hpf in zebrafish,57 and thus the myeloid cell populations were also analyzed at later time points. As with primitive hematopoiesis, the populations of fms+ (P < .01; Figure 3G,H,Q), lyz+ (P < .01; Figure 3I,J,R), and mpo+ (P < .01; Figure 3K,L,S) cells were reduced in gcsfr morphants at 96 hpf. This indicates that zebrafish gcsfr also participates in the generation of both granulocytes and monocytes/macrophages in definitive hematopoiesis.
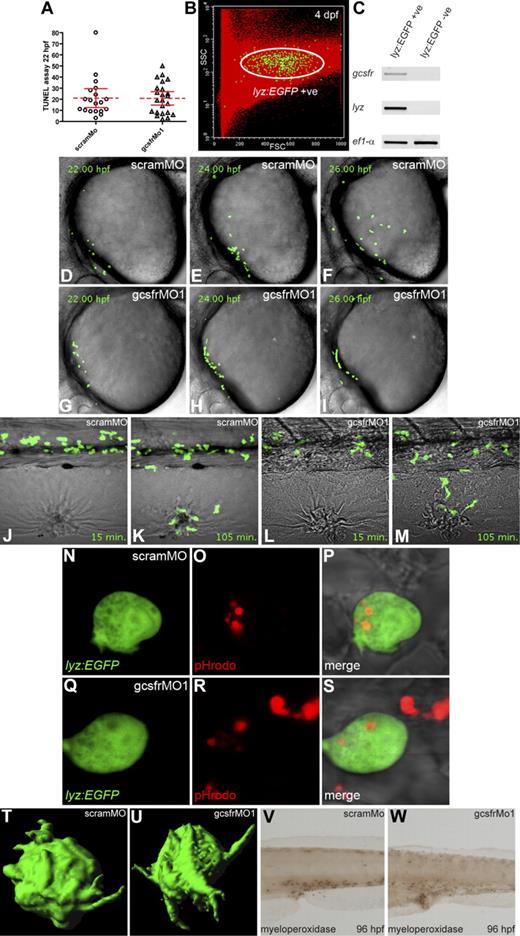
To investigate how gcsfr knockdown affects myelopoiesis, TUNEL assays were performed. No significant difference in apoptotic cells was observed within the RBI of gcsfr morphants compared with scramMo controls (Figure 4A), suggesting the reduced myeloid cell numbers are due to decreased production rather than decreased survival. To further understand the effects on cell migration and function lyz::EGFP transgenic embryos24 were used, having first established gcsfr expression in EGFP+ cells (Figure 4B,C). In vivo time-lapse imaging confirmed the striking inability of these cells to migrate across the yolk sac in gcsfr morphants (Figure 4G-I, Video S2) compared with scramMo controls (Figure 4D-F, Video S1). In contrast, the dorsoventral migration or directional migration in response to wounding (Figure 4J-M, Videos S3–4) was largely unaffected, although retention of EGFP+ cells at the wound was impaired. In addition, EGFP+ cells from gcsfr morphants were equally able to ingest bacteria (Figure 4N-S) and project pseudopodia (Figure 4T,U), whereas myeloperoxidase staining of individual cells was equally strong in gcsfr morphants, although the total number was reduced (P < .01; Figure 4V,W, Figure S2). These data collectively suggest a specific role for GCSFR in early myeloid cell migration and development, but that the myeloid cells produced using GCSFR-independent pathways function normally.
Functional analysis of gcsfr morphants. (A) Quantitation of TUNEL assay performed on 22-hpf embryos previously injected with either scramMo or gcsfrMo1. Only apoptotic cells on the yolk were counted, with the mean (dashed red line) and 95% confidence interval (red lines) indicated. (B) Fluorescence analysis of lyz::EGFP cells from 96-hpf embryos based on EGFP fluorescence, forward scatter (FSC), and side scatter (SSC) characteristics. (C) RT-PCR of various genes from total RNA extracted from lyz::EGFP-expressing fluorescence-activated cell sorting (FACS)–isolated cells. (D-I) In vivo visualization of lyz::EGFP cells at the developmental time points indicated in embryos previously injected with scramMo (D-F) or gcsfrMo1 (G-I). (J-M) Wounding-induced migration of lyz::EGFP cells in 72-hpf embryos that had been previously injected with scramMo (J,K) and gcsfrMo1 (L,M) at 15 minutes and 105 minutes after injury. (N-S) Phagocytosis assay of lyz::EGFP cells in 48-hpf embryos injected with pHrodo E coli BioParticle conjugate that had been previously injected with scramMo (N-P) or gcsfrMo1 (Q-S). Panels show separate EGFP (N,Q), rhodamine (O,R), and merged (P,S) images. (T,U) Three-dimensional isosurface reconstruction of lyz::EGFP cells on the yolk surface of 20-hpf embryos previously injected with scramMo (T) or gcsfrMo1 (U). (V,W) Myeloperoxidase staining of 96-hpf embryos that had been previously injected with scramMo (V) or gcsfrMo1 (W).
Functional analysis of gcsfr morphants. (A) Quantitation of TUNEL assay performed on 22-hpf embryos previously injected with either scramMo or gcsfrMo1. Only apoptotic cells on the yolk were counted, with the mean (dashed red line) and 95% confidence interval (red lines) indicated. (B) Fluorescence analysis of lyz::EGFP cells from 96-hpf embryos based on EGFP fluorescence, forward scatter (FSC), and side scatter (SSC) characteristics. (C) RT-PCR of various genes from total RNA extracted from lyz::EGFP-expressing fluorescence-activated cell sorting (FACS)–isolated cells. (D-I) In vivo visualization of lyz::EGFP cells at the developmental time points indicated in embryos previously injected with scramMo (D-F) or gcsfrMo1 (G-I). (J-M) Wounding-induced migration of lyz::EGFP cells in 72-hpf embryos that had been previously injected with scramMo (J,K) and gcsfrMo1 (L,M) at 15 minutes and 105 minutes after injury. (N-S) Phagocytosis assay of lyz::EGFP cells in 48-hpf embryos injected with pHrodo E coli BioParticle conjugate that had been previously injected with scramMo (N-P) or gcsfrMo1 (Q-S). Panels show separate EGFP (N,Q), rhodamine (O,R), and merged (P,S) images. (T,U) Three-dimensional isosurface reconstruction of lyz::EGFP cells on the yolk surface of 20-hpf embryos previously injected with scramMo (T) or gcsfrMo1 (U). (V,W) Myeloperoxidase staining of 96-hpf embryos that had been previously injected with scramMo (V) or gcsfrMo1 (W).
Identification and analysis of zebrafish gcsf
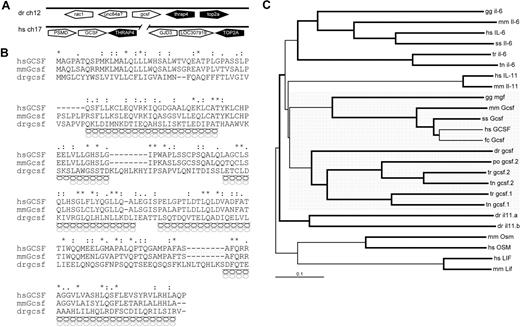
Genes encoding class I cytokines exhibit low overall sequence conservation, although they show conserved synteny and their products display a similar tertiary structure, consisting of 4 α-helices.58 Initial in silico searches for zebrafish gcsf using BLAST data mining failed to return any positive matches. However, a conserved synteny relationship has been reported between the GCSF, THRAP, and TOP2A genes in mammals and teleost fish, with GCSF being tail-to-tail with THRAP in many species.59 The zebrafish thrap gene was identified in a section of zebrafish chromosome 12. Detailed bioinformatic analysis of the genomic region downstream of this gene identified a region with homology to human GCSF. Genomescan was used to predict the putative zebrafish gcsf sequence, which maintained the tail-to-tail arrangement to thrap, with the top2a gene lying adjacent (Figure 5A). Analysis of the conceptual translation of zebrafish gcsf with Superfamily HMM confirmed it to be a member of the long-chain 4-helical cytokine family (data not shown).40 Sequence alignment revealed low but consistent sequence identity between the GCSF sequence of zebrafish and those of humans (16%) and mice (17%; Figure 5B), as well as the chicken myelomonocytic growth factor (MGF; 19%) and pufferfish GCSF.1 and GCSF.2 (both 25%; data not shown). Structural analysis using SWISS-MODEL identified conserved positions of α-helices with mammalian GCSF (Figure 5B),42,60 whereas phylogenetic analysis confirmed the likely evolutionary relationship of the zebrafish sequence with other GCSF sequences, in comparison with the related cytokines, IL-6 and IL-11 (Figure 5C). Expression of zebrafish gcsf was confirmed using RT-PCR at various time points (data not shown), confirming that it was not a pseudogene. However, due to apparently low expression levels, gcsf expression was virtually undetectable using WISH (data not shown), consistent with the normally undetectable levels of human GCSF under homeostatic circumstances.61
Characterization of zebrafish gcsf. (A) Synteny analysis of human GCSF and zebrafish gcsf gene loci. Pentagons represent genes showing their respective orientations: white indicates GCSF/gcsf genes; black, genes showing conserved synteny; and gray, nonsyntenic genes. (B) Alignment of human, mouse, and zebrafish GCSF sequences, as described in Figure 1. Predicted α-helical secondary structure is indicated for mammalian (black helices) and zebrafish (gray helices) sequences. Exons are indicated by alternating gray and white shading. (C) Phylogenetic analysis of GCSF and related cytokines from human (hs), mouse (mm), pig (ss), chicken (gg), cat (fc), fugu (tr), pufferfish (tn), and Japanese flounder (po) using the neighborhood-joining algorithm. Branches with a bootstrap value higher than 80% are bolded. GCSF sequences are highlighted by gray shading.
Characterization of zebrafish gcsf. (A) Synteny analysis of human GCSF and zebrafish gcsf gene loci. Pentagons represent genes showing their respective orientations: white indicates GCSF/gcsf genes; black, genes showing conserved synteny; and gray, nonsyntenic genes. (B) Alignment of human, mouse, and zebrafish GCSF sequences, as described in Figure 1. Predicted α-helical secondary structure is indicated for mammalian (black helices) and zebrafish (gray helices) sequences. Exons are indicated by alternating gray and white shading. (C) Phylogenetic analysis of GCSF and related cytokines from human (hs), mouse (mm), pig (ss), chicken (gg), cat (fc), fugu (tr), pufferfish (tn), and Japanese flounder (po) using the neighborhood-joining algorithm. Branches with a bootstrap value higher than 80% are bolded. GCSF sequences are highlighted by gray shading.
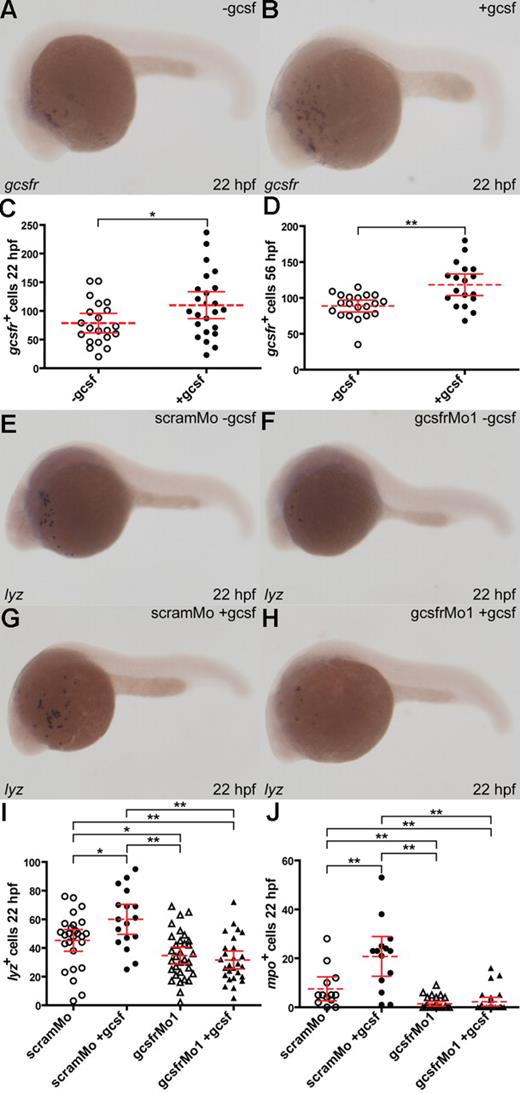
To test for a direct functional relationship between the putative zebrafish gcsf and gcsfr, in vitro–transcribed gcsf mRNA was injected into 1- to 4-cell embryos. This resulted in a significant increase in the number of gcsfr+ cells at both 22 hpf (P < .03; Figure 6A-C) and 56 hpf (P < .01; Figure 6D, and data not shown) compared with sham-injected embryos. To further investigate this interaction, either scramMo or gcsfrMo1 was coinjected with gcsf mRNA. Embryos coinjected with scramMo and gcsf mRNA showed a significant increase in lyz+ (P < .02) cells at 22 hpf compared with those injected only with scramMo (Figure 6E,F,I). As expected, the lyz+ cell population was reduced in gcsfrMo1 morphants compared with scramMo-injected embryos. Crucially, there was no subsequent increase in lyz+ cell populations in embryos coinjected with gcsfrMo1 and gcsf mRNA, compared with those injected with gcsfrMo1 alone (Figure 6G-I). In contrast, no significant difference was observed in total mpo+ cells between embryos injected with scramMo only and those injected with scramMo and gcsf mRNA–coinjected embryos, although there was a significant increase in the small number within the anterior population of mpo+ cells (P < .01; Figure 6J, and data not shown). Injection of gcsfrMo1 reduced the total mpo+ populations compared with scramMo-injected embryos (P < .01; data not shown), whereas coinjection of gcsfrMo1 and gcsf mRNA blocked the gcsf-induced increase in anterior population mpo+ cells (Figure 6J). Therefore, zebrafish gcsf exerts its effects on anterior RBI myeloid cell production in a gcsfr-dependent manner, confirming a likely ligand-receptor relationship.
Functional interaction between zebrafish gcsf and gcsfr. (A-D) Enforced gcsf expression expands the gcsfr+ population. Embryos were either injected with capped gcsf mRNA (+gcsf) or sham injected (−gcsf) and probed with gcsfr (A,B). The total number of gcsfr+ cells with or without gcsf was analyzed at 22 hpf (C) or 96 hpf (D), with mean (dashed red line), 95% confidence interval (red lines), and level of statistical significance (*P < .05; **P < .01) indicated. (E-J) Enforced gcsf expression expands myeloid populations via gcsfr. Embryos were coinjected with combinations of either scamMo or gcsfrMo1 with or without capped gcsf mRNA, as indicated, and probed with lyz (E-H). The total numbers of lyz+ (I) and anterior mpo+ cells (J) at 22 hpf were analyzed as discussed for panels C and D.
Functional interaction between zebrafish gcsf and gcsfr. (A-D) Enforced gcsf expression expands the gcsfr+ population. Embryos were either injected with capped gcsf mRNA (+gcsf) or sham injected (−gcsf) and probed with gcsfr (A,B). The total number of gcsfr+ cells with or without gcsf was analyzed at 22 hpf (C) or 96 hpf (D), with mean (dashed red line), 95% confidence interval (red lines), and level of statistical significance (*P < .05; **P < .01) indicated. (E-J) Enforced gcsf expression expands myeloid populations via gcsfr. Embryos were coinjected with combinations of either scamMo or gcsfrMo1 with or without capped gcsf mRNA, as indicated, and probed with lyz (E-H). The total numbers of lyz+ (I) and anterior mpo+ cells (J) at 22 hpf were analyzed as discussed for panels C and D.
Participation of zebrafish gcsf and gcsfr in emergency hematopoiesis
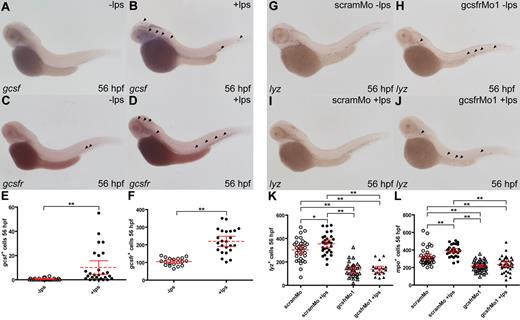
Signaling via the GCSF/GCSFR pathway plays a key role in emergency hematopoiesis.62 Consistent with this role, the expression of GCSF orthologues has been shown to be significantly up-regulated by challenge with bacterial LPS in mammals, birds, and fish, particularly in cells of the monocyte/macrophage lineage.59,62,63 To simulate emergency hematopoiesis, LPS was injected into the venous return of 48-hpf zebrafish embryos. This produced increased expression of gcsf 8 hours later, with a significant increase in both the number of predominantly large vasculature-associated cells exhibiting gcsf expression (P < .01), as well as their relative levels of expression (Figure 7A,B,D). LPS injection also led to a statistically significant increase in the number gcsfr+ cells (P < .01; Figure 7C,D,F). The involvement of zebrafish gcsfr in eliciting this emergency response was further analyzed by injecting LPS into the venous return of 48-hpf embryos that had been previously injected with either scramMo or gcsfrMo1. In scramMo-injected embryos, LPS challenge produced an increase in lyz+ (P < .03; Figure 7G,H,K) and mpo+ (P < .01; Figure 7L, and data not shown) cells compared with unchallenged embryos at 56 hpf. Again there were reduced numbers of lyz+ (P < .01) and mpo+ (P < .01) cells in unchallenged gcsfr morphants compared with their scramMo counterparts, but importantly no increase in gcsfr morphants challenged with LPS (Figure 7I-L). Therefore, zebrafish gcsfr plays an integral role in the emergency response elicited by LPS, presumably as a direct result of the increased gcsf expression induced by LPS.
Zebrafish gcsf and gcsfr participate in emergency myelopoiesis. (A-F) LPS affects gcsf+ and gcsfr+ cell populations. Embryos at 48 hpf were injected with 5 μg/mL LPS (+lps) or not (−lps) and fixed 8 hours later, before WISH analysis with either gcsf (A,B) or gcsfr (C,D). Total populations of gcsf (E) and gcsfr (F) were analyzed with mean (dashed red line), 95% CI (solid red lines), and level of statistical significance (*P < .05; **P < .01) indicated. (G-L) LPS effects require gcsfr. Embryos were injected at 1- to 2-cell stage with either scramMo or gcsfrMo1, injected with or without LPS at 48 hpf, and collected 8 hours later, before probing with lyz (I,J). Cell count data of the total populations of lyz+ (K) and mpo+ (L) cells at 56 hpf were analyzed as described for panels E and F.
Zebrafish gcsf and gcsfr participate in emergency myelopoiesis. (A-F) LPS affects gcsf+ and gcsfr+ cell populations. Embryos at 48 hpf were injected with 5 μg/mL LPS (+lps) or not (−lps) and fixed 8 hours later, before WISH analysis with either gcsf (A,B) or gcsfr (C,D). Total populations of gcsf (E) and gcsfr (F) were analyzed with mean (dashed red line), 95% CI (solid red lines), and level of statistical significance (*P < .05; **P < .01) indicated. (G-L) LPS effects require gcsfr. Embryos were injected at 1- to 2-cell stage with either scramMo or gcsfrMo1, injected with or without LPS at 48 hpf, and collected 8 hours later, before probing with lyz (I,J). Cell count data of the total populations of lyz+ (K) and mpo+ (L) cells at 56 hpf were analyzed as described for panels E and F.
Discussion
Zebrafish are increasingly being harnessed as an alternative model for investigating vertebrate hematopoiesis.64 A considerable literature has already been generated concerning the function of key hematopoietic transcription factors, including scl, lmo2, ikaros, gata1, spi1, and c/ebp1.18,,,–22,52,65 Increasingly, these studies are being extended to hematopoietic cytokines, their receptors, and downstream signaling molecules.28,–30,66,,–69 Collectively, these investigations have confirmed the broad similarity and functional equivalence of the hematopoietic regulatory networks in zebrafish,13 further emphasizing the relevance of the model. This study has characterized the zebrafish gcsfr, identified the gene encoding its cognate ligand, gcsf, and analyzed the involvement of gcsf/gcsfr during primitive, definitive, and emergency hematopoiesis. The data obtained have elucidated an overall conservation of the GCSF/GCSFR pathway, as well as provided new insights into how it regulates the early production and migration of myeloid cells.
Conservation of GCSF and GCSFR
We had previously identified a putative zebrafish gcsfr gene on the basis of overall sequence homology and conserved synteny.29 Further analysis revealed conservation of upstream transcription factor binding sites, splice structure, as well as key domains in the encoded protein, including the presence of Ig, CHD, 3 FBN, transmembrane, and intracellular domains. Within the CHD, the signature domain of the class I cytokine receptors,70 there was conservation of several key residues. This included the WSXWS motif, involved in ligand binding and receptor maturation,71 and the diproline “hinge” between the 2 FBN folds of the CHD, mutation of which affects ligand binding.72 There was also conservation of 2 of the 4 cysteines within the CHD. Although differing from human GCSFR, a similar arrangement of cysteines is seen in several other class I receptors, including LIFR.49 Moreover, our analysis identified conservation of several other sequences, including APXXRLDTM and QL(F/Y)WKP motifs, further analysis of which may prove fruitful.
Key residues important for intracellular signal transduction in mammalian GCSFR have been elucidated, with the majority of these also found to be conserved in zebrafish GCSFR. This included box 1, box 2, and box 3 motifs, that are also conserved within the GP130 family,2 as well as 4 tyrosine residues equivalent to those found in the human GCSFR, Y704, Y729, Y744, and Y764, although the zebrafish GCSFR possesses 2 additional tyrosine residues. Of particular note was the conservation of residues essential for mediating interaction with the JAK/STAT/SOCS pathway, including the box 1 motif and adjacent W residue important for JAK binding,73 the Y704 and Y744 equivalents that mediate STAT3 binding,74 and the Y729 equivalent that serves as a docking site for SOCS3.75 This is important as JAK1, JAK2, and STAT3 have each been shown to play key roles in GCSFR-mediated responses76,77 and SOCS3 is the key negative regulator of this receptor,78 with general conservation of the JAK/STAT/SOCS pathway components in zebrafish.66,,–69 Furthermore, residues involved in other key pathways were also conserved. These include the Y764 equivalent that is important for docking of Shc and 3BP2, as well as concomitant transduction of strong proliferative signals in mammalian systems,76,79 and equivalents of both Y704 and Y764 that mediate binding of SHP-2 and the p85 subunit of PI3-kinase.76 There was also conservation of the dileucine-containing STQPLL sequence within Box 3, which is important for efficient receptor internalization50,80 and found in several related receptors.29,81,82 Finally, the comparative sequence analysis revealed a conserved FPLLQ sequence at the C-termini of mammalian and zebrafish GCSFR sequences, the functional significance of which remains to be investigated.
Conserved synteny relationships were used to identify a putative gcsf gene in zebrafish. Despite low sequence identity, the predicted secondary structure of the encoded zebrafish protein matched that of mammalian GCSF proteins, whereas phylogenic analysis placed it within the clade of GCSF proteins from all other species. Despite a duplication of gcsf reported in other teleost fish,59 only a single gcsf gene was identified in zebrafish. The zebrafish gcsf exhibited low basal expression that was significantly up-regulated after injection of LPS, consistent with that observed with GCSF in other species.59,83 Injection of gcsf mRNA into zebrafish embryos increased the number of gcsfr+ and other myeloid cell populations, an effect that was blocked in gcsfr morphant embryos. This collectively suggests that the gene identified was a GCSF orthologue, with a conserved GCSF/GCSFR signaling pathway existing in zebrafish.
Function of the GCSF/GCSFR pathway in zebrafish
Primitive hematopoiesis in zebrafish begins at approximately 12 hpf,12 with expression of early hematopoietic markers (scl, lmo2, and ikaros), followed by markers for early myeloid (spi1 and c/epb1) and erythroid (gata1) populations.18,,,–22 This initially occurs along the length of the lateral plate mesoderm, coalescing into distinct anterior and posterior domains.13 Monocyte/macrophage (fms), and pan-leukocyte (lplastin and lyz) markers are initially expressed in the anterior domain of the RBI, with these cells subsequently migrating posteriorly.16,84 Conversely, the granulocyte-specific marker mpo is initially expressed in the pICM domain from 18 hpf, with significant expression appearing only much later in the RBI.25 Expression of gcsfr was limited to the RBI from 14 hpf and, as with fms+, lyz+, and lplastin+ cells,16 gcsfr+ cells subsequently migrated over the yolk sac toward the posterior of the embryo. The first gcsfr+ cells were observed in the pICM at approximately 26 hpf, after the commencement of circulation. Analysis of the zebrafish gcsfr promoter revealed the presence of conserved binding sites for SPI1 and C/EBP family members. The anterior expression profile initiating from the RBI of both spi1 and c/ebp1 is similar to that of gcsfr, and both have been implicated in the transcriptional regulation of mammalian GCSFR genes.48 Indeed, knockdown of spi1 led to significant decrease in gcsfr+ expression. Since spi1 is also expressed in the pICM from 14 hpf until 20 hpf,18 spi1 may initiate gcsfr expression in zebrafish, although this needs to be confirmed experimentally.
Morpholino-mediated knockdown of zebrafish gcsfr, targeting 2 independent splice sites within the gene, produced no effect on the expression of early hematopoietic markers, scl, lmo2, and ikaros, or initial spi1 expression. However, from 18 hpf onward, there was a decrease in the numbers of anterior RBI generated spi1+ and c/ebp1+ early myeloid cells. There was also a reduction in fms+ monocyte/macrophages, lyz+ and lplastin+ leukocytes, as well as mpo+ and mmp9+ granulocyte populations, although the latter effects were significantly greater in the respective anterior populations. As a corollary, injection of gcsf mRNA increased the numbers of gcsfr+ and lyz+ myeloid cells, as well as specifically increasing the anterior but not posterior mpo+ population. These data collectively suggest that the zebrafish GCSF/GCSFR pathway acts at the level of the common myeloid progenitor, influencing numbers of both monocyte/macrophages as well as granulocytic cells. It also demonstrates that myelopoiesis within the RBI is gcsfr dependent, whereas that within the pICM is largely gcsfr independent, consistent with the observed RBI expression of gcsfr at the anterior of the embryo. The relatively small, but consistent, effects of gcsfr knockdown on pICM myelopoiesis suggest that some gcsfr expression may also occur in this region that is below the limits of detection using WISH, and under the threshold that can stimulate robust proliferation in response to exogenous gcsf.
The initial wave of definitive hematopoiesis in zebrafish occurs between 24 to 48 hpf with the establishment of markers for definitive HSCs within the dorsal aorta.57 Later, hematopoiesis shifts to the kidney, thymus, and pancreas, the former analogous to the mammalian bone marrow.12 During the phase of definitive hematopoiesis, the gcsfr is expressed in several locations in the zebrafish embryo. From around 24 to 26 hpf gcsfr+ cells are observed in circulation, as well as in increasing numbers within the pICM, suggesting a second wave of production at this location. However, by 96 hpf the number of gcsfr+ cells in circulation decreases markedly, and gcsfr+ cells become limited to the developing kidney by 6 dpf, indicative of ongoing myelopoiesis occurring at this site. Knockdown of gcsfr led to decreased numbers of fms+, lyz+, and mpo+ cells at 96 hpf, suggesting a continued role for the GCSF/GCSFR pathway during definitive myelopoiesis.
Mice lacking GCSFR or patients constitutively expressing hypomorphic GCSFR mutants are neutropenic, but still possess mature granulocytes, indicating that both GCSFR-dependent and GCSFR-independent granulopoiesis occur in mammals.8,11,53 This study confirms that both pathways exist in zebrafish, but that they largely occur in anatomically distinct domains, at least during primitive hematopoiesis. Furthermore, the GCSF/GCSFR pathway functions more widely in myelopoiesis, rather than just granulopoiesis. This is consistent with the defects in both granulocytic and macrophage progenitors seen in GCSF−/− mice,7 as well as the decreased numbers of GMPs and granulocytes seen in competitive repopulation studies between wild-type and GCSFR−/− bone marrow.9 Such a broad functionality also extends to the chicken GCSF equivalent, MGF, which has been shown to promote the proliferation of both monocytes and granulocytes.59
Emergency hematopoiesis, such as that elicited by bacterial infection, leads to an increase in neutrophils to combat infection.62 This is mediated largely by induction of GCSF expression in response to bacterial LPS.62 This process was simulated by injection of LPS into 48-hpf embryos, which led to increased zebrafish gcsf expression, probably within circulating monocyte/macrophage cells. There were concomitant increases in gcsfr+, lyz+, and mpo+ cell populations, which were abrogated in gcsfr morphants, indicating emergency myelopoiesis occurs in a GCSF/GCSFR-dependent manner.
As confirmed by time-lapse confocal imaging on transgenic embryos, an important novel finding of this study was the requirement of gcsfr for the migration of anterior myeloid cells during embryonic development. This was not a migration defect, per se, as myeloid cells were seen to move in gcsfr morphants along the dorsoventral axis at this time or in response to wound healing at later time points, but rather a specific defect in directional migration across the yolk sac, and in retention at sites of wounding—the latter paralleling studies in which microtubule polymerization was inhibited.85 The GCSF/GCSFR pathway is known to participate in HPC and neutrophil mobilization,86 although this is via indirect transacting signals that are independent of GCSFR expression on these cells.86,87 Investigating potential parallels for this in early myeloid cell migration remains an exciting area for further research.
This study has established that a functional GCSF/GCSFR signaling system exists in zebrafish with conserved modes of regulation and function. This provides a validated experimental platform for the further in vivo study of this important ligand-receptor signaling system during both normal development and emergency responses. Further areas for study include a more detailed molecular analysis of the migration defect of myeloid cells in gcsfr morphants, the role of specific intracellular signaling pathways in mediating the effects of the gcsf/gcsfr, and the elucidation of the pathways controlling gcsfr-independent myelopoiesis. Collectively, these investigations will add to our understanding of vertebrate hematopoiesis.
The online version of this article contains a data supplement.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 USC section 1734.
Acknowledgments
The authors thank Dan Merlo, Cuong Huynh, Suzita Noor, and Deakin Animal House staff for zebrafish care, as well as Peter Beech, Liza John, Rowena Lewis, Sarah Shigdar, Lynda O'Sullivan, and Jan West for helpful discussions and advice.
C.L. and B.A.O. were supported by Deakin University Postgraduate Research Awards and the Center for Cellular and Molecular Biology (Burwood, Australia).
Authorship
Contribution: C.L. and A.C.W. conceptualized experiments, interpreted data, and wrote and drafted the paper; C.L. also performed most of the bioinformatics and experimental studies; C.J.H. and P.C. performed the confocal and FACS experiments and contributed to the paper; B.A.O. provided assistance in the analyses and drafting of the paper; and all authors read and approved the final paper.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: Alister C. Ward, School of Medicine, Deakin University, Waurn Ponds, Victoria, Australia, 3217; e-mail: award@deakin.edu.au.