Chronic myeloproliferative neoplasms (CMPNs) are clonal disorders originating from the transformation of a single cell through one or more genetic events. Thus, clonality has been used to distinguish CMPNs from reactive conditions such as secondary erythrocytosis or thrombocytosis. As most CMPN patients lack cytogenetic markers of clonal hematopoiesis, the diagnosis of clonality relied mainly on X chromosome inactivation assays and was limited to female patients. The discovery of the JAK2 V617F mutation in most cases of polycythemia vera (PV) and some 50% of patients with essential thrombocythemia (ET) and primary myelofibrosis (PMF), collectively referred to as the “classic” Philadelphia chromosome–negative CMPN,2 seemed to provide a convenient marker of monoclonal hematopoiesis. Now we learn that, as always, things are more complicated than first thought.
Independent JAK2 V617F mutations occur in ET but not in PV. In ET, the presence of JAK2 does not suppress JAK2 WT hematopoiesis. The JAK2 WT progenitors are susceptible to independent JAK2 V617F mutations.
Independent JAK2 V617F mutations occur in ET but not in PV. In ET, the presence of JAK2 does not suppress JAK2 WT hematopoiesis. The JAK2 WT progenitors are susceptible to independent JAK2 V617F mutations.
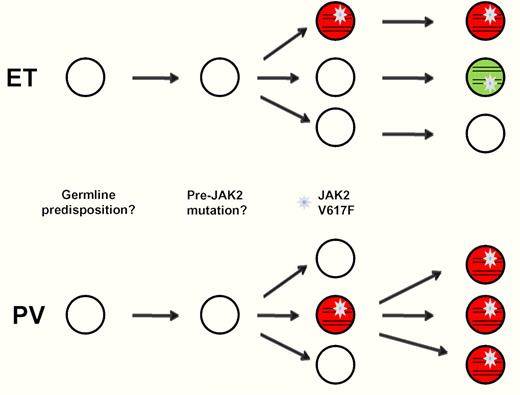
It had been noted several years ago that some patients with the clinical features of ET display polyclonal hematopoiesis, suggesting they may have a reactive condition that phenocopies ET.3 However, the presence of JAK2 V617F in these patients is not consistent with this concept. So what is happening?
To solve the mystery, Lambert et al took advantage of a frequent single SNP in exon 19 of JAK2, 3′ of V617F. Using a combination of sequencing and restriction enzyme digestion, they found the V617F mutation on both JAK2 alleles in 10 of 11 informative patients, indicative of at least 2 independent JAK2 mutation events. In contrast, none of 5 informative PV patients showed this polyclonal pattern. While this finding needs to be confirmed in more patients, it suggests a fundamental pathogenetic difference between PV and ET. The authors speculate that in ET, in contrast to PV, the cell with the initial JAK2 V617F mutation does not inevitably achieve full clonal dominance, since (unlike erythropoietin in PV) thrombopoietin remains unsuppressed. This allows JAK2 V617F–negative progenitor cells to continue proliferating—and acquire additional JAK2 V617F mutations. Why this hypothetical first JAK2 V617 mutation leads to ET in some patients but to PV or PMF in others remains an open question. Several lines of evidence indicate that JAK2 V617F is not the primary pathogenic event in CMPNs. On transformation to acute myeloid leukemia of JAK2 V617F–positive CMPN, the leukemic blasts are JAK2 V617F–negative in approximately 50% of cases, consistent with a pre–JAK2 V617F event.4,5 In some patients with JAK2 V617F–positive PV, a subset of the erythropoietin-independent erythroid colonies display wild-type (WT) JAK2.6 Finally, the recent identification of TET2 mutations in some JAK2 V617F–negative myeloid precursors of JAK2 V617F–positive patients indicates that the acquisition of TET2 mutations may predate the acquisition of JAK2 V617F in a subset of CMPN patients, and, therefore, could be the initiating mutation.7 Thus, it will be interesting to study the relationship between TET2 and polyclonal JAK2 V617F–positive ET. The prediction is that these patients will have either no or different TET2 mutations. Moreover, CMPNs display a familial predisposition, suggesting that a heritable factor can predispose to the acquisition of JAK2 V617F. Consistent with this, 3 groups independently identified a specific JAK2 haplotype as a major risk factor for the development of MPN and showed that the JAK2 V617F mutation preferentially arises on the allele bearing this susceptibility haplotype.8,–10
Lambert et al propose that polyclonal ET should not be considered a malignancy. In fact, their previous work has shown that ET patients with polyclonal hematopoiesis have a decreased risk of thrombosis, suggesting that polyclonality may be a marker of a more benign disease course.3 We think it is important to point out that polyclonality on its own is not synonymous with nonmalignancy. Thus, the independent acquisition of identical oncogenic mutations in the same gene has been observed in highly malignant multifocal solid tumors, such as lung and pancreatic cancers.11,12 In ET, the fact that JAK2 V617F may arise independently in multiple progenitor cells suggests that this progenitor cell pool may still be susceptible to acquiring additional mutations, such as TET2, which could lead to leukemic transformation.13
That a single type of point mutation is present in the majority of CMPN patients has been puzzling to the field ever since its discovery.14 Clearly, there must be a selective advantage to the acquisition of JAK2 V617F, and the observation that this particular mutation can occur at multiple independent times in the same individual makes this argument even more compelling. The big question is now what this selective advantage may be. The race is on.
Conflict-of-interest disclosure: The authors declare no competing financial interests. ■

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal