Abstract
Adenosine deaminase (ADA) deficiency is a disorder of the purine metabolism leading to combined immunodeficiency and systemic alterations, including skeletal abnormalities. We report that ADA deficiency in mice causes a specific bone phenotype characterized by alterations of structural properties and impaired mechanical competence. These alterations are the combined result of an imbalanced receptor activator of nuclear factor-κB ligand (RANKL)/osteoprotegerin axis, causing decreased osteoclastogenesis and an intrinsic defect of osteoblast function with subsequent low bone formation. In vitro, osteoblasts lacking ADA displayed an altered transcriptional profile and growth reduction. Furthermore, the bone marrow microenvironment of ADA-deficient mice showed a reduced capacity to support in vitro and in vivo hematopoiesis. Treatment of ADA-deficient neonatal mice with enzyme replacement therapy, bone marrow transplantation, or gene therapy resulted in full recovery of the altered bone parameters. Remarkably, untreated ADA–severe combined immunodeficiency patients showed a similar imbalance in RANKL/osteoprotegerin levels alongside severe growth retardation. Gene therapy with ADA-transduced hematopoietic stem cells increased serum RANKL levels and children's growth. Our results indicate that the ADA metabolism represents a crucial modulatory factor of bone cell activities and remodeling. The trials were registered at www.clinicaltrials.gov as #NCT00598481 and #NCT00599781.
Introduction
Genetic defects in the adenosine deaminase (ADA) gene are among the most common causes for severe combined immunodeficiency (SCID).1 Lack of ADA causes accumulation of purine metabolites in plasma, lymphoid tissues, and red blood cells. ADA-SCID patients have lymphopenia, absent cellular and humoral immunity, failure to thrive, and recurrent infections.2 The additional presence of skeletal, hepatic, renal, lung, and neurologic abnormalities underlines that ADA deficiency is a multiorgan pathology.1,3
Approximately 50% of early-onset ADA-deficient patients exhibit radiologically detectable bone defects.4 Lack of organized cartilage columnar formation, large lacuni containing hypertrophied cells, lack of trabecular formation with uninterrupted areas of calcified cartilage, as well as few osteoblasts (OBs) and osteoclasts (OCs) with normal mineralizing osteoid have been reported.4-6 Nonetheless, the underlying cellular and molecular mechanisms have remained unclear because of the complexity of the skeletal phenotype and the fact that bone abnormalities are observed also in other immunodeficiencies.7,8 Increasing evidence underlines the importance of an intense crosstalk between immune and bone cells regulating not only bone remodeling but also hematopoiesis.9 Therefore, ADA deficiency represents an important model to study both the impact of altered purine metabolism and immunodeficiency on bone and bone marrow (BM) stroma.
Bone is a highly complex organ that participates in mineral metabolism,10,11 provides structural integrity for the body, and supports hematopoiesis. Through a well-organized balance of bone resorption and formation in a time- and space-dependent manner, bone remodeling enables bone mechanical competence and adaptation to various mechanical demands.12 This complex process requires interaction between different cell types and is regulated by a variety of mechanical and molecular factors. OCs derived from monocyte/macrophage precursor cells differentiate into multinucleated giant cells specialized in bone resorption.13 Monocyte/macrophage function has been described to be modulated by adenosine receptor activation.14
OBs are bone-forming cells derived from mesenchymal origin. They secrete an extracellular matrix consisting mainly of type I collagen, which they later mineralize. Human OBs possess all 4 adenosine receptor subtypes and produce extracellular adenosine, which modulates their secretion of interleukin-6 (IL-6) and osteoprotegerin (OPG).15 Adenosine exhibits a potent mitogenic effect on murine calvarial OBs,16 whereas adenosine triphosphate is a known inhibitor of bone formation.17
Besides their established role in bone remodeling, OBs are a crucial component of the hematopoietic stem cell (HSC) niche.18-20 The interaction of HSCs with OBs is critical for maintaining stem cell properties, including self-renewal capacity and the ability to differentiate into multiple lineages.21
OBs and OCs interact closely to maintain bone homeostasis. Their crosstalk is mediated by RANKL and its decoy receptor OPG. RANKL is produced by OBs but also activated T and B lymphocytes. The source of OPG had historically been attributed to OBs; until recently, BM B cells have been described as a major source of BM OPG.22 Because the RANKL-to-OPG ratio regulates OC activity and formation, it is conceivable that T−B− immunodeficiencies, such as ADA-SCID, are accompanied by a general misbalance of bone homeostasis.
In ADA-deficient patients, as in other forms of SCID, bone marrow transplantation (BMT) is an effective treatment.23 Enzyme replacement therapy (ERT) with polyethylene glycol-conjugated bovine ADA (PEG-ADA) provides metabolic detoxification but often insufficient immune reconstitution.24 Recently, gene therapy (GT) with BM CD34+ cells transduced with a γ-retroviral vector has been shown to correct both the immune and metabolic defects of ADA-SCID children pretreated with low-intensity conditioning.25,26 However, little information is available on the corrective effects of these treatments on the associated bone defects.7,27,28
Because of the complexity of the interactive scenario described herein and the limited availability of biologic materials obtained from ADA-deficient patients, we first focused our study on the bone phenotype of the ADA−/− mouse model. ADA−/− mice retain many features associated with ADA deficiency in humans, including T- and B-cell lymphopenia and a profound metabolic defect.29 Elevated adenosine levels cause abnormal alveolar development, leading ADA−/− mice to die postnatally within 3 weeks.
We hypothesized that the altered purine metabolism in ADA deficiency impairs OBs and OCs genesis and activity through immunodependent and -independent processes resulting in a specific bone phenotype. We characterized the in vivo bone phenotype of ADA−/− mice and evaluated possible OB and/or OC defects in vitro. In parallel, we analyzed the ADA−/− BM stromal cell compartment and assessed its capacity to support hematopoiesis. We extended our study to 15 ADA-SCID patients, either naive for treatment, under ERT or after GT to assess bone parameters. Results were discussed in light of potential correction of the ADA bone phenotype by current treatment regimens.
Methods
Mice
ADA-deficient mice have been described previously.29 Breeding pairs for FVB;129-Adatm1MW-TgN(PLADA)4118Rkmb were purchased from The Jackson Laboratory. Rag2−/−γc−/− mice30 on BALB/c background were obtained from the Central Institute for Experimental Animals. Matched wild-type controls (BALB/c) were purchased from The Jackson Laboratory. All animals were bred and maintained in a specific pathogen-free animal facility. Procedures were performed according to protocols approved by the Committee for Animal Care and Use of San Raffaele Scientific Institute (Institutional Animal Care and Use Committee 318).
Patients and clinical trials
Patients were enrolled in 2 subsequent phase 1/2 clinical protocols approved by San Raffaele Scientific Institute's Ethical Committee and Italian National Regulatory Authorities (www.clinicaltrials.gov, #NCT00598481 and #NCT00599781). Orphan Drug Status to ADA vector-transduced CD34+ cells was granted by the European Medicines Agency (EMEA/OD/053/05) to the Italian Telethon Foundation. Patients' parents signed informed consent to experimental treatment. GT treatment was performed as described.31 Only patients not displaying any other congenic or endocrine diseases were included. In case of healthy donors or patients on ERT not enrolled in either trial, parents or adult subjects signed informed consent in accordance with the Declaration of Helsinki for research studies on peripheral blood.
Peripheral quantitative computed tomography
Measurements were performed using a Stratec Research SA+ peripheral quantitative computed tomography (pQCT) scanner (Stratec) at voxel size of 0.070 mm3 and scan speed of 3 mm/second. All images were obtained with 360 projections and section thickness of 500 or 100 μm.
To separate muscle from bone, scans were analyzed twice with pQCT software 6.00B, using contour mode 1 and peel mode 2, threshold of −50 mg/cm3, inner threshold of 40 mg/cm3, filter 2 F03F04 for calculation of total area, and threshold of 280 mg/cm3, inner threshold of 400 mg/cm3 for the calculation of bone area. Bones alone were analyzed using contour mode 2 and peel mode 2 with a threshold of 350 mg/cm3 for the calculation of trabecular and total bone parameters in metaphysis and with a threshold of 600 mg/cm3 for cortical bone parameters in diaphysis.
Histomorphometry
Femora from 19-day-old mice were fixed, dehydrated, and embedded in methyl methacrylate without decalcification. Histomorphometric analysis was performed on toluidine blue– and Alizarin Red S–stained sections using a light microscope (Nikon Axiophot) equipped with an image analysis system (Nikon DS-5Mc Videocamera; NIS Elements AR 2.20 Nikon Software).
Calvarial OB cultures and lentiviral transduction
Calvaria from newborn mice were explanted, digested with collagenase, and grown until confluence. In the absence of stimuli, ADA−/− OBs were transduced with pCCLsin.cPPT.hPGK.hADA.Wpre (PGK-ADA) at a multiplicity of infection of 100 as described previously.32 Median ADA activity was 13 460.5 plus or minus 1904 nmol/h per milligram of protein. At days 3, 5, 8, 10, 12, and 15, replated (15 000/well) cells were harvested and counted using an automated cell counter (Coulter Counter ZM, Electronics).
Human OB-like cultures
Human bone cell cultures were established using trabecular bone samples obtained from waste materials during orthopedic surgery.33 Cells were tested for alkaline phosphatase and osteocalcin production after 1,25(OH)2D3 10−8 M to ensure that they were endowed with OB characteristics.
In vitro osteoclastogenesis
Flushed total BM was plated in presence of 100 ng/mL macrophage colony-stimulating factor (M-CSF; PeproTech) for 3 days. Cells were replated at 5000/well in 96-well-plates and cultured in presence of 25 ng/mL M-CSF and 100 ng/mL RANKL (PeproTech). Cultures were fixed, thrombin receptor activating peptide (TRAP) staining was performed according to the manufacturer's instructions (Sigma-Aldrich), and multinucleated (3 or more nuclei) TRAP+ cells were scored.
Alamar Blue viability assay
OBs, M-CSF–dependent BM macrophages, and stromal layers for long-term culture-initiating cell (LTC-IC) assays were isolated as described in “Calvarial OB cultures,” “In vitro osteoclastogenesis,” and “Stromal cultures (CFU-F, LTC-IC)” and plated in 96-well-plates; 10 μL Alamar Blue (Biosource) reagent was added per well. After 3 hours, viability was measured (excitation 530 nm, emission 590 nm) using a Victor3 Microplate Reader (PerkinElmer Life and Analytical Sciences).
ADA enzymatic activity
Gene expression analyses
RNA was extracted using EUROzol (Euroclone) and transcribed into cDNA using a High-Capacity cDNA Archive Kit (Applied Biosystems). Real-time polymerase chain reactions were carried out using Assay-on-Demand gene expression arrays (Applied Biosystems). The relative expression of each gene was normalized to hypoxanthine phosphoribosyl transferase (HPRT) as endogenous control. mRNA levels were quantified using the comparative threshold-cycle method. Arrays used included the following: Mm01187117_m1, Mm00801666_g1, Mm01337566_m1, Mm00485009_m1, Mm00435452_m1, Mm00441908_m1, Mm03003491_m1.
FACS analyses
Staining for annexin V and 7-amino-actinomycin D was performed on cultured OBs according to manufacturer's instructions (both BD Biosciences PharMingen). Stainings for OC precursors from flushed total BM were performed as described previously.32 Rat anti–mouse monoclonal antibodies used included the following: fluorescein isothiocyanate–conjugated anti-CD117 (2B8; BD Biosciences PharMingen), phycoerythrin-conjugated anti-F4/80 (Serotec), allophycocyanin-conjugated anti-CD11b (M1/70; BD Biosciences PharMingen), and Pacific Blue–conjugated anti-CD48 (HM48-1; BioLegend). Samples were analyzed using a BD fluorescence-activated cell sorter (FACS) Canto and DiVa software (BD Biosciences PharMingen).
ELISA
Murine RANKL, OPG (R&D Systems), and murine N-terminal propeptide of type I procollagen (PINP; iDS) were assayed on sera from 19-day-old male ADA+/+ and ADA−/−, Rag2γc+/+ and Rag2γc−/− or 12-week-old ADA+/+ and rescued ADA−/− mice. Enzyme-linked immunosorbent assay (ELISA) for fragments of the type I collagen (CTX; RatLaps ELISA; Nordic Biosciences Diagnostics) was performed according to the manufacturer's instructions on serum samples from 12-week-old ADA+/+ or rescued ADA−/− mice starved for 6 hours. Assessment of CTX in untreated ADA−/− mice of 19 days of age was not feasible because of the starvation protocol. Increased sensitivity sRANKL and human OPG ELISA (Biomedica) were performed on plasma from patients and pediatric normal donors according to the manufacturer's instructions.
Stromal cultures (CFU-F, LTC-IC)
Fibroblast colony-forming unit (CFU-F) assays were performed using MesenCult medium (StemCell Technologies) according to the manufacturer's protocol. Stromal feeder layers for LTC-IC assays were established from flushed BM using MyeloCult medium (StemCell Technologies) according to the manufacturer's instructions. Within 1 week after irradiation (15 Gy), lineage-negative cells isolated using StemSep Murine Progenitor Enrichment separation (StemCell Technologies) were added. ADA−/− cells showed comparable numbers and viability after irradiation at 15 Gy and 30 Gy, excluding a potential bias because of differential irradiation sensitivity36 (data not shown). After 4 weeks of coculture, adherent and nonadherent cells from each well were transferred into semisolid MethoCult (StemCell Technologies). CFU were scored after 10 days.
Proteome profile
Supernatants from LTC-IC cultures established as described in “Stromal cultures (CFU-F, LTC-IC)” were analyzed using Proteome Profiler Arrays (R&D Systems) according to the manufacturer's instructions. Densitometric analysis was carried out using ImageQuant software (GE Healthcare).
5-Fluorouracil treatment
Neonatal ADA+/+ and ADA−/− mice were transplanted into the temporal vein with 3000 ADA+/+ lineage-negative cells, as described.32 Transplanted cells were purified by StemSep Murine Progenitor Enrichment separation (StemCell Technologies). Starting from 6 weeks of age, surviving mice received weekly intraperitoneal injections with 150 mg/kg 5-fluorouracil (Sigma-Aldrich).
ERT, BMT, and GT treatment
ADA−/− were rescued by weekly intraperitoneal injections (1000 U/kg) with PEG-ADA (Adagen, Enzon), by BMT or GT as described previously.32 A total of 5 × 106 ADA+/+ or transduced ADA−/− BM cells were infused by injection through the temporal vein of irradiated neonatal ADA−/− mice.
Statistical analyses
For comparisons between groups, an unpaired 2-tailed Student t test was used. A P value less than .05 was considered significant. For analyses of LTC-IC assays, the difference in scored colonies was evaluated fitting a generalized linear model based on Poisson distribution, taking into account possible differences between experiments and first-order interactions. For Proteome Profile analyses, a linear model was used to assess differences in protein expression, taking into account possible differences between experiments. P values were adjusted for multiplicity by the Benjamini-Hochberg false discovery rate method. All analyses were performed with R software (Version 2.8.0, Fedora Project).
Results
ADA-deficient mice display a specific bone phenotype
Because bone defects resulting from alterations of crosstalk between hematopoietic and bone cells have been described in other immunodeficient mice,37 we studied the ADA−/− bone phenotype compared with double-mutant Rag2γc−/− mice.30 The latter lack T, B, and NK cells but do not have the profound metabolic defect typical for ADA−/− mice. Comparing both immunodeficient models allowed us to distinguish the contribution of immunodeficiency from that of the metabolic disease to the ADA bone phenotype. Because of strain differences, ADA−/− and Rag2γc−/− mice were compared with their respective sex- and age-matched wild-type controls.
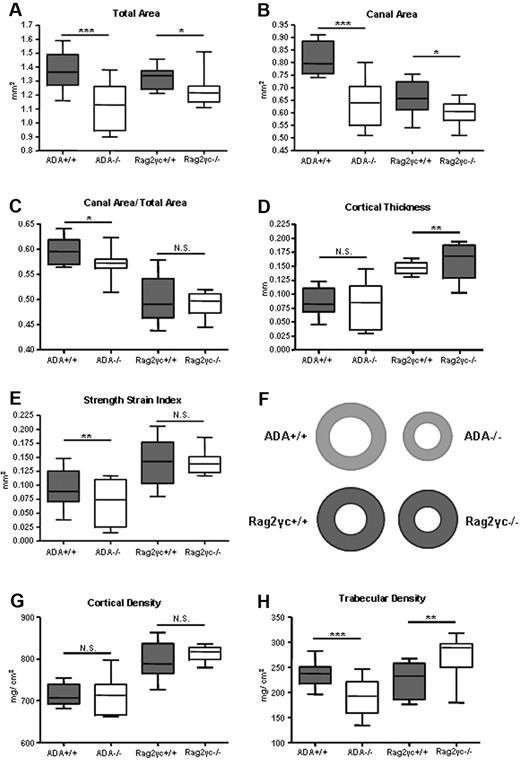
ADA−/− mice were analyzed at 19 days of age, when they are generally smaller, whereas Rag2γc−/− mice (x-ray not shown) are comparable with wild-type controls (supplemental Figure 1A, available on the Blood website; see the Supplemental Materials link at the top of the online article). To characterize bone size and structural parameters pQCT was performed on tibiae and femora retrieved from ADA+/+, ADA−/−, Rag2γc+/+, and Rag2γc−/− mice. As shown in Figure 1A-B, a significantly lower total area and medullary canal area was found in both immunodeficient mouse models. This difference was more pronounced in the ADA−/− compared with Rag2γc−/− mice, resulting in significant reduction in the canal area to total area ratio (Figure 1C). Consequently, cortical thickness (Figure 1D) was found to be significantly larger in Rag2γc−/− mice, whereas no difference was measured in ADA−/− mice. The strength-strain index, direct estimate of bone strength, was significantly reduced only in ADA−/− mice (Figure 1E). The observed structural differences were not accompanied by changes in cortical bone density (Figure 1G). Interestingly, the trabecular density was significantly reduced in ADA−/− mice but significantly increased in Rag2γc−/− mice (Figure 1H).
Ex vivo femural pQCT analyses of ADA+/+ and ADA−/− compared with Rag2γc+/+ and Rag2γc−/− mice. (A) Total area (mg/cm3). (B) Medullary canal area (mm2). (C) Cortical thickness (mm). (D) Canal area/total area ratio (AU). (E) Strength-strain index (mm3). (F) Schematic view representing cross-sectional bone sections in diaphysis of all 4 groups of mice analyzed. (G) Cortical density (mm/cm3). (H) Trabecular density (mm/cm3). Box and whiskers graphs. ***P < .001. **P < .001-.005. *P = .005-.05. ADA+/+ (n = 22), ADA−/− (n = 18), Rag2γc+/+ (n = 10), and Rag2γc−/− (n = 14).
Ex vivo femural pQCT analyses of ADA+/+ and ADA−/− compared with Rag2γc+/+ and Rag2γc−/− mice. (A) Total area (mg/cm3). (B) Medullary canal area (mm2). (C) Cortical thickness (mm). (D) Canal area/total area ratio (AU). (E) Strength-strain index (mm3). (F) Schematic view representing cross-sectional bone sections in diaphysis of all 4 groups of mice analyzed. (G) Cortical density (mm/cm3). (H) Trabecular density (mm/cm3). Box and whiskers graphs. ***P < .001. **P < .001-.005. *P = .005-.05. ADA+/+ (n = 22), ADA−/− (n = 18), Rag2γc+/+ (n = 10), and Rag2γc−/− (n = 14).
ADA−/− mice have significant weight loss during the last days of life (supplemental Figure 1B). Because developmental changes in bone strength are secondary to increasing loads imposed by larger muscle forces,38 the functional muscle-bone unit was evaluated to distinguish whether any observed bone defect is specifically related to insufficient muscle mass. We found a large reduction in the muscle cross-sectional area, whereas bone mineral content remained unchanged, resulting in a significantly higher proportion between bone mineral content and muscle cross-sectional area in ADA−/− mice (supplemental Figure 1C).
ADA−/− mice display low trabecular bone volume
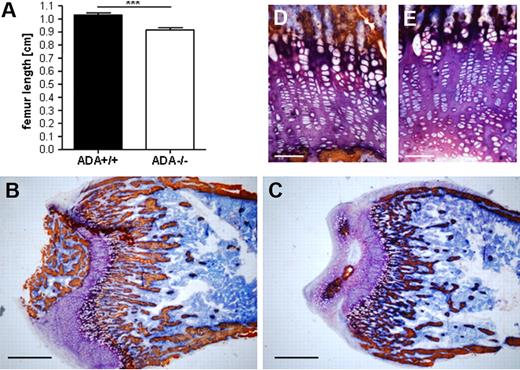
The low trabecular density, detected by pQCT, was confirmed at static histomorphometric evaluation. As shown in Figure 2A, longitudinal growth of ADA−/− femora is impaired compared with controls. To assess defects in the trabecular bone network, metaphyseal sections were analyzed by image analysis software (Figure 2B-C). As summarized in supplemental Table 1, the trabecular bone volume and trabecular number in ADA−/− mice were significantly lower compared with wild-type. Consequently, trabecular separation was significantly increased, whereas trabecular thickness was not different between both groups. Despite early reports on a selective toxicity of ADA substrates for chondrocytes,6 we did not detect any gross differences between the growth plates of ADA+/+ and ADA−/− mice (Figure 2D-E).
Reduced longitudinal growth and trabecular bone in ADA−/− mice. (A) ADA−/− femora and tibiae (not shown) are significantly shorter compared with wild-type (cm). ***P < .001. Metaphyseal bone sections of ADA+/+ (B) and ADA−/− (C) femora (original magnification ×2.5). Scale bar represents 600 μm and original magnification ×20 of ADA+/+ (D) and ADA−/− (E). Scale bar represents 50 μm; indicates defects in ossification but not in cartilage formation. Histomorphologic analyses were performed on ADA+/+ (n = 5) and ADA−/− (n = 5) femora. BV TV indicates trabecular bone volume (%); Tb Th, trabecular thickness (μm); Tb N, trabecular number (1/mm); Tb S, trabecular separation (mm).
Reduced longitudinal growth and trabecular bone in ADA−/− mice. (A) ADA−/− femora and tibiae (not shown) are significantly shorter compared with wild-type (cm). ***P < .001. Metaphyseal bone sections of ADA+/+ (B) and ADA−/− (C) femora (original magnification ×2.5). Scale bar represents 600 μm and original magnification ×20 of ADA+/+ (D) and ADA−/− (E). Scale bar represents 50 μm; indicates defects in ossification but not in cartilage formation. Histomorphologic analyses were performed on ADA+/+ (n = 5) and ADA−/− (n = 5) femora. BV TV indicates trabecular bone volume (%); Tb Th, trabecular thickness (μm); Tb N, trabecular number (1/mm); Tb S, trabecular separation (mm).
Reduced RANKL/OPG ratio and reduced bone formation in ADA−/− mice
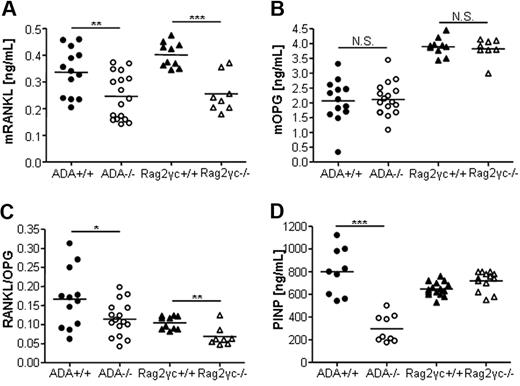
RANKL and OPG, well-characterized key players of crosstalk between OBs or stromal cells with hematopoietic OC precursors, synchronize osteoclastogenesis, and bone resorption. As shown in Figure 3A, both immunodeficient models showed significantly reduced RANKL levels, whereas serum OPG levels were in the normal range for both groups (Figure 3B). Consequently, the RANKL/OPG ratio, indicator of in vivo bone turnover, was reduced in ADA−/− and Rag2γc−/− mice (Figure 3C). To assess new bone formation, we measured the levels of the PINP. As shown in Figure 3D, PINP levels were significantly reduced in sera of ADA−/− mice, although comparable in Rag2γc−/− mice and controls.
Reduced RANKL/OPG ratio and PINP levels in ADA-deficient mice. (A) Murine serum RANKL (ng/mL) in ADA+/+ (n = 22) and ADA−/− (n = 18), Rag2γc+/+ (n = 10), and Rag2γc−/− (n = 14) mice. (B) Murine OPG (ng/mL). (C) Murine RANKL/OPG ratio (AU). ***P < .001. **P = .001-.005. *P = .005-.05. N.S. indicates not significant. (D) In vivo bone formation rate as assessed by PINP levels are significantly lower in ADA−/− mice compared with wild-type controls. Serum PINP levels (ng/mL), ADA+/+ (n = 9), ADA−/− (n = 9), Rag2γc+/+ (n = 15), and Rag2γc−/− (n = 13).
Reduced RANKL/OPG ratio and PINP levels in ADA-deficient mice. (A) Murine serum RANKL (ng/mL) in ADA+/+ (n = 22) and ADA−/− (n = 18), Rag2γc+/+ (n = 10), and Rag2γc−/− (n = 14) mice. (B) Murine OPG (ng/mL). (C) Murine RANKL/OPG ratio (AU). ***P < .001. **P = .001-.005. *P = .005-.05. N.S. indicates not significant. (D) In vivo bone formation rate as assessed by PINP levels are significantly lower in ADA−/− mice compared with wild-type controls. Serum PINP levels (ng/mL), ADA+/+ (n = 9), ADA−/− (n = 9), Rag2γc+/+ (n = 15), and Rag2γc−/− (n = 13).
ADA activity in bone cells
ADA is ubiquitously expressed, but little information is available on the specific levels of enzymatic activity in bone cells. Remarkably, ADA activity in wild-type OBs was found 3-fold higher compared with mesenchymal progenitor cells, 2-fold higher compared with OC precursors and therefore lies in the range of other lymphoid organs, such as the spleen or BM (supplemental Table 2).
ADA−/− OB activity is impaired
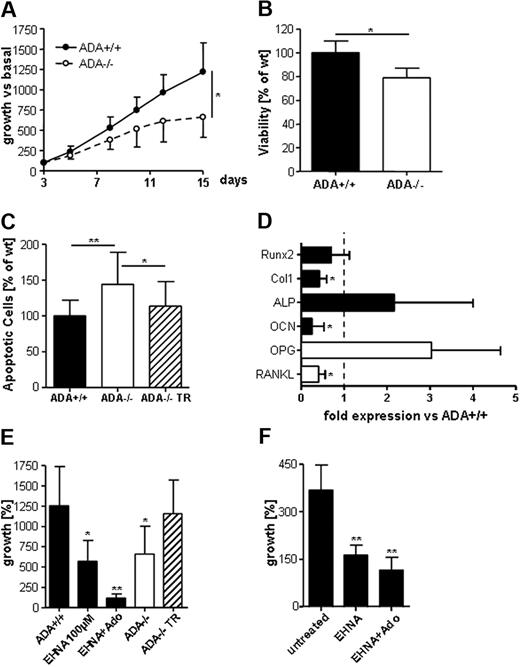
The reduced periosteal bone apposition in vivo and the high ADA activity detected in wild-type OBs in vitro suggested defects of the osteoblastic compartment in ADA−/− mice. ADA−/− OBs showed a significantly lower proliferation rate compared with wild-type cells in vitro (Figure 4A). This was associated with a significant decrease in viability and a significant increase in the percentage of apoptotic cells (Figure 4B-C). Importantly, transduction with a lentiviral vector encoding for ADA rescued both the growth defects and increased sensitivity to apoptosis observed in ADA−/− cells (Figure 4C-E). We performed quantitative gene expression arrays designed to assess whether OB differentiation is altered in ADA−/− mice (Figure 4D). Runx2, known to contribute to early osteogenic differentiation, was expressed at comparable levels.39 Although expression levels of collagen type 1- and osteocalcin-recognized markers of OB activity were significantly decreased in ADA−/− OBs,39 alkaline phosphatase, essential for matrix mineralization, revealed 2-fold increased expression levels. Interestingly, RANKL expression levels were significantly reduced, whereas OPG levels were 3-fold increased. ADA inhibition with erythro-9-(2-hydroxy-3-nonyl)-adenine (EHNA; 100 mM) is a commonly used method to mimic ADA deficiency in vitro. For unavailability of sufficient patient material, we used EHNA to block ADA activity in primary human OBs. As demonstrated in murine OBs (Figure 4E), EHNA efficiently blocked ADA activity, reducing OBs proliferation to levels comparable with ADA−/− cells. Proliferation of human OBs was significantly inhibited when cultured with EHNA (Figure 4F), culturing with EHNA and adenosine completely ablated proliferation.
Defects in ADA−/− OB activity. (A) In vitro ADA−/− OBs grow significantly less than wild-type cells. *P = .005-.05. (B) Viability as assessed by the Alamar Blue staining is significantly lower in cultured ADA−/− OBs. (C) The percentage of apoptotic cells as assessed by annexin V+/7-amino-actinomycin D− staining is significantly higher in ADA−/− OBs but normalizes in cells transduced with a lentiviral vector encoding for ADA (ADA−/− TR). **P = .001-.005. (D) TaqMan gene expression analyses for osteoblastic differentiation markers Runx2, type 1 collagen, alkaline phosphatase, and osteocalcin as well as RANKL and OPG; fold expression versus wild-type (represented by dashed line), normalized for HPRT endogenous control; average of 3 independent experiments ± SD. (E) Growth of murine ADA+/+ OB after 12 days in vitro is reduced when cultured with EHNA (100 μM) and abolished when cultured with EHNA and adenosine (1 mM). ADA−/− OBs regain growth after lentiviral vector transduction. Growth as percentage of initially plated OBs ± SD. (F) Growth of human OB after 13 days of in vitro is reduced when cultured with EHNA (100 μM). **P < .01.
Defects in ADA−/− OB activity. (A) In vitro ADA−/− OBs grow significantly less than wild-type cells. *P = .005-.05. (B) Viability as assessed by the Alamar Blue staining is significantly lower in cultured ADA−/− OBs. (C) The percentage of apoptotic cells as assessed by annexin V+/7-amino-actinomycin D− staining is significantly higher in ADA−/− OBs but normalizes in cells transduced with a lentiviral vector encoding for ADA (ADA−/− TR). **P = .001-.005. (D) TaqMan gene expression analyses for osteoblastic differentiation markers Runx2, type 1 collagen, alkaline phosphatase, and osteocalcin as well as RANKL and OPG; fold expression versus wild-type (represented by dashed line), normalized for HPRT endogenous control; average of 3 independent experiments ± SD. (E) Growth of murine ADA+/+ OB after 12 days in vitro is reduced when cultured with EHNA (100 μM) and abolished when cultured with EHNA and adenosine (1 mM). ADA−/− OBs regain growth after lentiviral vector transduction. Growth as percentage of initially plated OBs ± SD. (F) Growth of human OB after 13 days of in vitro is reduced when cultured with EHNA (100 μM). **P < .01.
No intrinsic defect of ADA−/− OCs
Consistent with the small medullary canal area observed in ADA−/− mice, serum RANKL levels were found to be significantly decreased, suggesting a block in osteoclastogenesis. To exclude a lack of OC precursors, we assessed their percentage in the BM by FACS staining for CD11b, cKit, CD48, and F4/80. The CD11b+/cKit− population was selected as myeloid precursors; within this subpopulation, we distinguished committed myeloid precursors from macrophages by CD48 and F4/80 staining. The relative percentages of OC precursors of both the monocyte and macrophage lineages were increased in the BM of ADA−/− mice (supplemental Figure 2A). This significant difference was maintained when the percentage of OC precursors was normalized for the absolute number of total BM cells and the median size of the BM cavity (supplemental Figure 2B). No differences in viability of OC precursors and in vitro osteoclastogenesis from M-CSF and RANKL-stimulated BM were detected (supplemental Figure 2C-D).
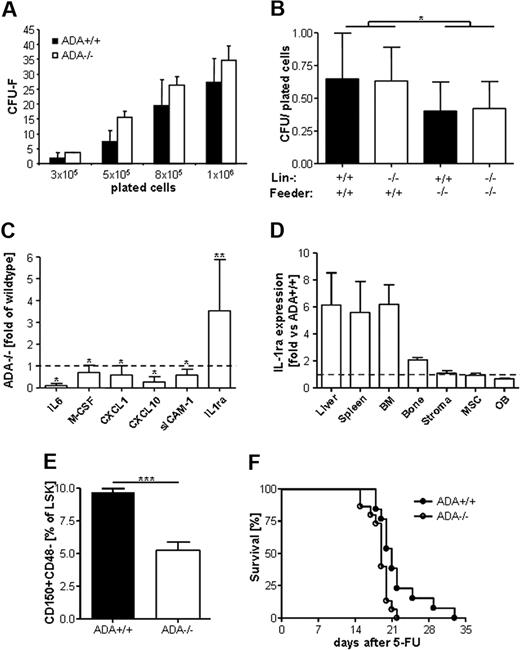
Reduced hematopoietic support of ADA−/− stroma
Given the importance of OB for the maintenance of the HSC niche, profound defects of the osteoblastic compartment in ADA−/− mice described herein imply a potential impact on the HSC niche and/or hematopoiesis itself. Using CFU-F assays, we showed that ADA+/+ and ADA−/− mesenchymal progenitor cells grow equally in vitro (Figure 5A). Nonetheless, when cocultured with ADA+/+ or ADA−/− lineage-negative cells, ADA−/− stromal cells supported colony formation less efficiently than wild-type cells (Figure 5B). This difference was significantly different considering wild-type versus ADA−/− feeder layers (P = .012), but independent from the cocultured lineage-negative cells. Proteome profiling of total cytokine and chemokine production from the supernatant of LTC-IC cultures revealed reduced levels of M-CSF, IL-6, CXCL1/10, and sICAM-1 (Figure 5C). Interestingly, only the IL-1 receptor antagonist (IL-1ra) was expressed at higher levels compared with wild-type. The secreted form of the IL-1ra is an acute-phase protein intervening in the counterregulation of inflammatory processes and is highly expressed in liver and spleen.40 Gene expression arrays for IL1-ra revealed 5.6- to 6.2-fold increased expression levels in spleen, liver, and BM from ADA−/− mice. Normal IL-1ra expression was detected in cultured OBs, stromal, and mesenchymal cells (Figure 5D), indicating that hematopoietic cells are probably the source of increased IL-1ra levels detected in proteome profiles from ADA−/− LTC-IC assays. To assess potential defects of the HSC niche in situ, we assessed the percentage of CD150+CD48− within the lineage-negative, Sca1+ and cKit+ compartment, which are generally thought to be quiescent HSC with a frequency of 50% repopulating capacity.41 As shown in Figure 5E, the percentage of LSK CD150+CD48− HSC is significantly lower in ADA−/− mice compared with wild-type. In addition, we transplanted neonatal ADA+/+ and ADA−/− mice with wild-type lineage-negative cells. Hematopoietic reconstitution was assessed by monitoring survival on weekly injections of the cell-cycle-dependent myelotoxic agent 5-fluorouracil.42 As shown in Figure 5F, because of hematopoietic failure, ADA−/− mice transplanted with wild-type cells died earlier compared with ADA+/+ mice (P = .011).
Stromal insufficiency to support hematopoiesis. (A) CFU-F assays show comparable frequency of mesenchymal progenitor cells in ADA+/+ or ADA−/− total BM. (B) LTC-IC assays of ADA+/+ or ADA−/− feeder layers cocultured with ADA+/+ or ADA−/− lineage− cells. ADA+/+ or ADA−/− LTC-IC grow significantly less when cocultured with ADA−/− feeder layers (P = .012). (C) Proteome profile from supernatants of ADA+/+ and ADA−/− cocultures, performed in triplicate; average ± SD. **P = .001-.005. *P = .005-.05. (D) TaqMan gene expression analyses for IL-1ra. Spleen, liver, BM, and bone in vivo. Stromal cells as used for LTC-IC, mesenchymal progenitor cells as cultured for CFU-F assays, OBs in vitro; fold expression versus wild-type (represented by dashed line), normalized for HPRT endogenous control (average of 3 experiments ± SD. (E) The percentage of quiescent HSCs, as assessed by FACS staining for lineage−, Sca1+, cKit+, CD150+, CD48− cells is significantly lower in ADA−/− (n = 8) compared with ADA+/+ mice (n = 8). ***P < .001. (F) Survival curves of ADA+/+ and ADA−/− mice transplanted with ADA+/+ lineage− cells after multiple 5-fluorouracil injections at days 7, 14, 21, and 28. P = .011.
Stromal insufficiency to support hematopoiesis. (A) CFU-F assays show comparable frequency of mesenchymal progenitor cells in ADA+/+ or ADA−/− total BM. (B) LTC-IC assays of ADA+/+ or ADA−/− feeder layers cocultured with ADA+/+ or ADA−/− lineage− cells. ADA+/+ or ADA−/− LTC-IC grow significantly less when cocultured with ADA−/− feeder layers (P = .012). (C) Proteome profile from supernatants of ADA+/+ and ADA−/− cocultures, performed in triplicate; average ± SD. **P = .001-.005. *P = .005-.05. (D) TaqMan gene expression analyses for IL-1ra. Spleen, liver, BM, and bone in vivo. Stromal cells as used for LTC-IC, mesenchymal progenitor cells as cultured for CFU-F assays, OBs in vitro; fold expression versus wild-type (represented by dashed line), normalized for HPRT endogenous control (average of 3 experiments ± SD. (E) The percentage of quiescent HSCs, as assessed by FACS staining for lineage−, Sca1+, cKit+, CD150+, CD48− cells is significantly lower in ADA−/− (n = 8) compared with ADA+/+ mice (n = 8). ***P < .001. (F) Survival curves of ADA+/+ and ADA−/− mice transplanted with ADA+/+ lineage− cells after multiple 5-fluorouracil injections at days 7, 14, 21, and 28. P = .011.
Recovery of altered bone parameters in rescued ADA−/− mice
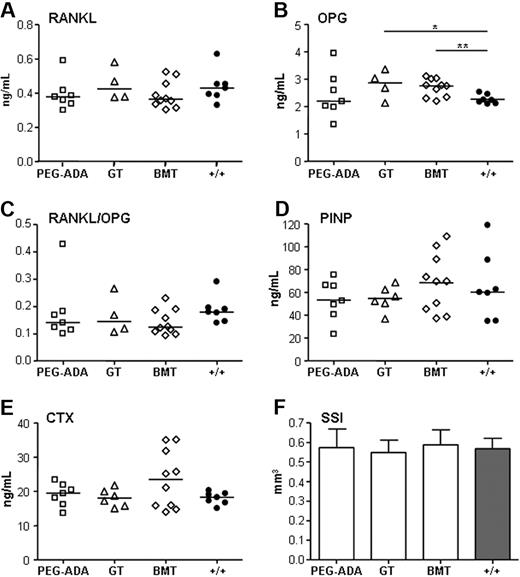
To assess the effect of different treatments on the ADA bone phenotype, we rescued newborn ADA−/− mice with ERT, BMT, or ex vivo lentiviral-mediated GT. Serum concentrations of RANKL, OPG, PINP, and CTX were assessed in surviving mice at 12 weeks of age. As shown in Figure 6, mice rescued by all 3 different treatments displayed full correction of serum RANKL and, consequently, RANKL/OPG levels (Figure 6A-C). Serum PINP levels were completely rescued, whereas CTX levels were comparable with controls (Figure 6D-E). pQCT analyses confirmed that all bone parameters previously observed to be altered in naive ADA−/− mice recovered to normal (Figure 6F, supplemental Figure 3).
Rescue of the ADA bone phenotype by ERT, GT, or BMT. Comparable serum levels of (A) RANKL (ng/mL), (B) OPG (ng/mL), *P = .005-.05; **P = .001-.005. (C) RANKL/OPG ratio (AU), (D) PINP (ng/mL), and (E) CTX (ng/mL) at 12 weeks of age. Scatter plots plus average. PEG-ADA (n = 7), GT (n > 4), BMT (n = 10), and wild-type controls (n = 7). (F) Strength-strain index (mm3); average ± SD; PEG-ADA (n = 4), GT (n = 5), BMT (n = 5), and wild-type controls (n = 4).
Rescue of the ADA bone phenotype by ERT, GT, or BMT. Comparable serum levels of (A) RANKL (ng/mL), (B) OPG (ng/mL), *P = .005-.05; **P = .001-.005. (C) RANKL/OPG ratio (AU), (D) PINP (ng/mL), and (E) CTX (ng/mL) at 12 weeks of age. Scatter plots plus average. PEG-ADA (n = 7), GT (n > 4), BMT (n = 10), and wild-type controls (n = 7). (F) Strength-strain index (mm3); average ± SD; PEG-ADA (n = 4), GT (n = 5), BMT (n = 5), and wild-type controls (n = 4).
ADA-SCID patients show delayed growth and altered bone remodeling
From the few case reports in ADA-SCID patients, it has remained unclear whether their severe growth delay is associated with specific defects in bone remodeling. To assess whether bone turnover is impaired, we measured RANKL and OPG ratio in plasma of 5 ADA-deficient patients naive for treatment and displaying severe growth retardation (Table 1). In all patients, RANKL levels were severely reduced or undetectable (P = .005). Interestingly, serum OPG levels were significantly increased (P = .003), resulting in RANKL versus OPG ratio significantly reduced compared with age-matched normal donors (P = .011).
ADA-SCID patients naive for therapy display significantly reduced growth
| . | Age, y . | Percentile height, Z-score . | RANKL, pg/mL . | OPG, pg/mL . | RANKL/OPG, × 102 . |
|---|---|---|---|---|---|
| Patient no. 1 | 1 | −1.66 | 1.42 | 181.9 | 0.78 |
| Patient no. 2 | 1.9 | −3.91 | 1.03 | 129.6 | 0.79 |
| Patient no. 3 | 1.8 | −1.83 | 0 | 181.9 | 0 |
| Patient no. 4 | 1.6 | −1.90 | 0 | 145.2 | 0 |
| Patient no. 5 | 0.5 | −2.21 | 3.08 | 257.3 | 1.19 |
| ND 1-5 years of age (n = 10), mean (SEM) | 11.49 (3) | 104.1 (7) | 13.4 (5) | ||
| P | .005 | .003 | .011 |
| . | Age, y . | Percentile height, Z-score . | RANKL, pg/mL . | OPG, pg/mL . | RANKL/OPG, × 102 . |
|---|---|---|---|---|---|
| Patient no. 1 | 1 | −1.66 | 1.42 | 181.9 | 0.78 |
| Patient no. 2 | 1.9 | −3.91 | 1.03 | 129.6 | 0.79 |
| Patient no. 3 | 1.8 | −1.83 | 0 | 181.9 | 0 |
| Patient no. 4 | 1.6 | −1.90 | 0 | 145.2 | 0 |
| Patient no. 5 | 0.5 | −2.21 | 3.08 | 257.3 | 1.19 |
| ND 1-5 years of age (n = 10), mean (SEM) | 11.49 (3) | 104.1 (7) | 13.4 (5) | ||
| P | .005 | .003 | .011 |
ND indicates normal donor.
GT but not enzyme replacement normalizes RANKL in ADA-SCID patients
As reported in Table 2, patients receiving ERT long-term (1-21 years), displayed low or undectable serum RANKL levels (P = .004), whereas OPG was in the normal range (P = .504). This resulted in a significantly reduced RANKL/OPG ratio (P = .033). All patients displayed significantly reduced height compared with age-matched standards. We next analyzed RANKL and OPG in patients treated with HSC-GT combined to reduced intensity conditioning.31 As reported in Table 3, in patients treated with GT, RANKL, and consequently the RANKL/OPG ratio significantly increased one to 2 years after treatment (P = .005; P = .031). Importantly, both serum RANKL (P = .979) and the RANKL/OPG ratio (P = .515) were not significantly different from the range observed in pediatric normal donors 1 to 5 years of age (Table 1). Consistently, 8 of 9 patients displayed an increase in percentile of height at the last follow-up after treatment. However, the percentile of height and bone age (Greulich/Pyle) was not fully normalized in all patients.
ADA-SCID patients on ERT with PEG-ADA display reduced growth
| . | Age at last follow-up, y . | PEG-ADA, y . | Percentile height, Z-score . | RANKL, pg/mL . | OPG, pg/mL . | RANKL/OPG, × 102 . |
|---|---|---|---|---|---|---|
| Patient no. 6 | 5.6 | 5 | −1.69 | 0 | 78.1 | 0 |
| Patient no. 7 | 1.5 | 1 | −2.06 | 0 | 120.6 | 0 |
| Patient no. 8 | 1.4 | 1 | −0.29 | 1.61 | 153.2 | 1.05 |
| Patient no. 9 | 1.3 | 1 | 1.29 | 0 | 145.2 | 0 |
| Patient no. 10 | 6.2 | 6 | −0.38 | 2.85 | 18.4 | 15.48 |
| Patient no. 11 | 2.5 | 1 | −2.14 | 2.57 | 173.8 | 1.48 |
| Patient no. 12 | 12.5 | 10 | −3.61 | 0 | 93.3 | 0 |
| Patient no. 13 | 22.5 | 21 | −0.93 | 0 | 167.5 | 0 |
| Patient no. 14 | 19 | 17 | −1.98 | 0 | 135.3 | 0 |
| ND 1-15 years of age (n = 14), mean (SEM) | 8.06 (1.6) | 106.9 (5.6) | 8.4 (1.7) | |||
| P | .004 | .504 | .033 |
| . | Age at last follow-up, y . | PEG-ADA, y . | Percentile height, Z-score . | RANKL, pg/mL . | OPG, pg/mL . | RANKL/OPG, × 102 . |
|---|---|---|---|---|---|---|
| Patient no. 6 | 5.6 | 5 | −1.69 | 0 | 78.1 | 0 |
| Patient no. 7 | 1.5 | 1 | −2.06 | 0 | 120.6 | 0 |
| Patient no. 8 | 1.4 | 1 | −0.29 | 1.61 | 153.2 | 1.05 |
| Patient no. 9 | 1.3 | 1 | 1.29 | 0 | 145.2 | 0 |
| Patient no. 10 | 6.2 | 6 | −0.38 | 2.85 | 18.4 | 15.48 |
| Patient no. 11 | 2.5 | 1 | −2.14 | 2.57 | 173.8 | 1.48 |
| Patient no. 12 | 12.5 | 10 | −3.61 | 0 | 93.3 | 0 |
| Patient no. 13 | 22.5 | 21 | −0.93 | 0 | 167.5 | 0 |
| Patient no. 14 | 19 | 17 | −1.98 | 0 | 135.3 | 0 |
| ND 1-15 years of age (n = 14), mean (SEM) | 8.06 (1.6) | 106.9 (5.6) | 8.4 (1.7) | |||
| P | .004 | .504 | .033 |
ADA-SCID patients treated with hematopoietic stem cell gene therapy display a significant increase in RANKL
| Patient no. . | Age at therapy, y . | Age at last follow-up, y . | Percentile height, Z-score . | RANKL, pg/mL . | OPG, pg/mL . | RANKL/OPG, × 102 . | Percentage of bone age/ actual age, y (post GT) . | ||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Pre . | Post (at last follow-up) . | Pre . | Post . | Pre . | Post . | Pre . | Post . | ||||
| 1 | 1 | 7.6 | −1.66 | −1.12 | 1.4 | 3.65 | 181.9 | 113.1 | 0.78 | 3.23 (2) | 96% (6) |
| 2 | 1.9 | 8 | −3.91 | −1.48 | 1.0 | 16.1 | 129.6 | 62.3 | 0.79 | 25.95 | 109% (6) |
| 4 | 1.6 | 3.3 | −1.9 | −2.11 | 0 | 0.82 | 195.7 | 172.8 | 0 | 0.47 (1) | 96% (1) |
| 5 | 0.5 | 1.5 | −2.21 | −2.07 | 3.0 | 18.3 | 257.3 | 191.5 | 1.19 | 9.59 (1) | ND |
| 6 | 5.6 | 10.7 | −1.69 | −1.22 | 0 | 2.3 | 78.1 | 157.5 | 0 | 1.46 (1) | 81% (4) |
| 7 | 1.5 | 4.6 | −2.06 | −0.69 | 0 | 3.54 | 120.6 | 412.9 | 0 | 0.85 (2) | 65% (3) |
| 8 | 1.4 | 3.6 | −0.29 | 1.46 | 1.6 | 9.1 | 153.2 | 198.6 | 1.05 | 4.58 (1) | 74% (2) |
| 9 | 1.3 | 2.9 | 1.29 | 0.20 | 0 | 6.84 | 145.2 | 148.1 | 0 | 4.61 (1) | 64% (1) |
| 15 | 1.6 | 6.3 | −3.11 | −0.99 | 2.2 | 8.94 | 121.2 | 64.2 | 1.87 | 13.93 | ND |
| P | .005 | .698, NS | .031 | ||||||||
| Patient no. . | Age at therapy, y . | Age at last follow-up, y . | Percentile height, Z-score . | RANKL, pg/mL . | OPG, pg/mL . | RANKL/OPG, × 102 . | Percentage of bone age/ actual age, y (post GT) . | ||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Pre . | Post (at last follow-up) . | Pre . | Post . | Pre . | Post . | Pre . | Post . | ||||
| 1 | 1 | 7.6 | −1.66 | −1.12 | 1.4 | 3.65 | 181.9 | 113.1 | 0.78 | 3.23 (2) | 96% (6) |
| 2 | 1.9 | 8 | −3.91 | −1.48 | 1.0 | 16.1 | 129.6 | 62.3 | 0.79 | 25.95 | 109% (6) |
| 4 | 1.6 | 3.3 | −1.9 | −2.11 | 0 | 0.82 | 195.7 | 172.8 | 0 | 0.47 (1) | 96% (1) |
| 5 | 0.5 | 1.5 | −2.21 | −2.07 | 3.0 | 18.3 | 257.3 | 191.5 | 1.19 | 9.59 (1) | ND |
| 6 | 5.6 | 10.7 | −1.69 | −1.22 | 0 | 2.3 | 78.1 | 157.5 | 0 | 1.46 (1) | 81% (4) |
| 7 | 1.5 | 4.6 | −2.06 | −0.69 | 0 | 3.54 | 120.6 | 412.9 | 0 | 0.85 (2) | 65% (3) |
| 8 | 1.4 | 3.6 | −0.29 | 1.46 | 1.6 | 9.1 | 153.2 | 198.6 | 1.05 | 4.58 (1) | 74% (2) |
| 9 | 1.3 | 2.9 | 1.29 | 0.20 | 0 | 6.84 | 145.2 | 148.1 | 0 | 4.61 (1) | 64% (1) |
| 15 | 1.6 | 6.3 | −3.11 | −0.99 | 2.2 | 8.94 | 121.2 | 64.2 | 1.87 | 13.93 | ND |
| P | .005 | .698, NS | .031 | ||||||||
NS indicates not significant; and ND, not done.
Numbers in parentheses indicate year after GT.
Discussion
Skeletal defects have been reported to be a common feature of ADA deficiency in humans3-5,8,27,28 ; nonetheless, it has remained unclear whether they are a consequence of SCID or caused by alterations in purine metabolism. The present study has filled this gap by providing evidence that ADA deficiency in mice is associated with a specific bone phenotype, characterized by alterations in structural properties and mechanical competence. However, these structural alterations being only partially superimposable to those observed in T−B− Rag2γc−/− mice, could not be ascribed to immunodeficiency per se. While the small medullary canal area appeared a joint feature in both immunodeficient models, low bone mass and strength-strain index were observed only in ADA−/− mice. The reduced trabecular density, specifically affected by ADA deficiency, was related to a low trabecular number, rather than reduced trabecular thickness. Because fetal bone development predicts bone mass accrual as a consequence of increasing trabecular bone volume in function of growth,43 the decreased trabecular number might be the expression of impaired ossification of the cartilaginous template with subsequent deterioration of bone structural properties. However, no mineralization defects were measured in the cortical bone of ADA−/− mice, and no osteoid seams were observed in static histomorphometric evaluation, indicating an appropriate acquisition of tissue material properties. Although no gross alteration of the growth plate morphology could be observed, the contribution of reduced growth plate activity for the determination of the bone phenotype is plausible because both ADA-deficient mice and patients display significantly reduced longitudinal growth. Contrarily, the contribution of muscle dysfunction can be considered minimal because the higher muscle-bone-unit here observed was related to the faster loss of muscle than bone mass because of metabolic toxification at a later stage of survival.
The described ADA bone phenotype is consistent with the hypothesis that the altered purine metabolism impairs OC and OB genesis, proliferation and activity through immuno-dependent and -independent processes.14-17 Because of reduced RANKL levels, the serum RANKL/OPG ratio was significantly reduced in both ADA−/− and Rag2γc−/− mice. The small medullary canal area fits this observation, suggesting a shift of the bone remodeling sequence toward reduced osteoclastogenesis. Interestingly, proteome profiling of cytokine and chemokine production from LTC-IC cultures revealed reduced levels of M-CSF and IL-6, factors involved in OC activity and generation.9 It is doubtful that this shift is the result of an intrinsic defect of OC precursors because in the presence of M-CSF and RANKL they equally form TRAP+ cells in vitro.
The high ADA activity measured in wild-type OBs indicates a strong dependency of this cell type on the ADA metabolic pathway. Interestingly, enzymatic activity measured in mesenchymal progenitor cells was 3-fold lower, indicating that differentiating OBs must up-regulate ADA expression considerably. It is therefore conceivable that, with increasing dependency on the ADA enzyme during proliferation or differentiation, OB function and viability become severely affected.
The reduced outward displacement of the thin cortex as well as the lower serum PINP levels in ADA−/− mice are in agreement with the hypothesis that OBs are the major casualty of ADA substrates' toxicity. The lower proliferation rate of ADA−/− OBs observed in vitro, associated with decreased viability and increased apoptosis, confirms this hypothesis and underlines the importance of purine metabolism in OB function. Importantly, correction of ADA expression by lentiviral vector transduction leads to full correction of ADA−/− OB growth defects and apoptosis, providing direct evidence that the OBs insufficiency state is cell intrinsic. Proliferation of primary murine and human OBs was similarly reduced, when mimicking ADA deficiency using the ADA inhibitor EHNA. Thus, low OB numbers may contribute to the bone phenotype observed in both ADA-deficient mice and patients. In accordance with impaired bone formation, the expression profile of murine ADA−/− OBs is characterized by low collagen type 1 and osteocalcin levels. However, it is likely that OBs compensate for their low numbers expressing elevated levels of alkaline phosphatase, so that no differences of bone material properties were detected. Similar to the low RANKL/OPG ratio observed in ADA−/− sera, their respective RNA expression levels were altered also in ADA−/− OBs. Being the dominant mediators of osteoclastogenesis, their misbalance confirms the central role of OBs determining the ADA bone phenotype and suggests the activation of a compensatory mechanism for bone mass preservation.9
Because RANKL ligand is also produced by B and T cells, its reduced serum levels can be partially attributed to the lymphopenia in both ADA−/− and Rag2γc−/− mice. Nonetheless, lymphocytes are severely reduced but not completely absent in BM and thymus of both immunodeficient models, which probably accounts for the residual serum RANKL levels. In addition, RANKL expression, in contrast to OPG, is down-regulated in ADA−/− OBs. Its reduced expression might be induced by the increased levels of IL-1ra, detected in ADA−/− mice, which may act by preventing IL-1 signaling, a potent stimulator of bone resorption. IL-1 modulates OC activity directly44 or indirectly through its ability to stimulate RANKL production by OBs45 ; therefore, IL-1ra secreted at elevated levels in ADA−/− mice probably restricts IL-1–induced bone resorption and RANKL expression. It can be speculated that the reduced endosteal resorption might counterbalance the low periosteal bone apposition to fulfill the physical requisite of bone strength for loading and lightness for mobility.46 Considering that BM B cells are a major producer of OPG,22 it was unexpected to observe normal serum OPG levels in ADA−/− mice. Nonetheless, OPG may be produced by other cell sources; and indeed, we found a 3-fold up-regulation of OPG mRNA levels in ADA−/− OBs.
These data suggest an OB insufficiency state as consequence of ADA deficiency that, given the interplay between osteogenesis and hematopoiesis, implies an impact on the BM microenvironment and the HSC niche.18-20 In vivo depletion of OBs caused loss of HSCs, followed by a marked decrease in BM cellularity.20 Consistently, the OB insufficiency in ADA−/− mice is associated with a reduced BM cellularity (not shown). The capacity of ADA−/− stromal cells to support colony formation from lineage-negative cells was significantly lower compared with wild-type. This reduced capacity might be associated with the low production of IL-6, crucial survival factor for HSCs and is in agreement with the defective hematopoietic support reported in IL-6–deficient mice.47 The hypothesis that the HSC niche in ADA deficiency is specifically affected was further supported by the reduced percentage of LSK CD150+CD48− HSCs in ADA−/− BM and by the hematopoietic failure and premature death of ADA−/− mice transplanted with wild-type HSC when challenged with 5-fluorouracil. Our data may provide a possible explanation for the higher toxicity of pretransplantation conditioning and transplantation failures observed in ADA-SCID patients treated with BMT24,48 as well as for frequent findings of hematopoietic abnormalities1 (and A. Aiuti, unpublished results, January 2009). Future studies will be needed to assess whether current therapeutic approaches may be improved by simultaneous targeting of the niche. In this regard, transplantation of normal or ADA engineered mesenchymal stem cells may facilitate HSC engraftment or contribute to the reconstitution of the bone cell compartment.
Comparative analyses of adult ADA−/− mice treated with ERT, GT, or BMT showed full correction of RANKL, RANKL/OPG, and PINP as well as recovery of bone growth. We observed a significant increase of serum OPG levels in mice treated with GT or BMT, a phenomenon that has also been reported in allotransplanted patients.49,50 This is probably related to pretransplantation conditioning rather than the transplantation procedure itself. Consistently nonirradiated PEG-ADA–treated mice displayed normal OPG levels.
The results obtained in 15 ADA-SCID patients, either naive or under different treatments, have confirmed that bone defects are a common feature associated with ADA deficiency in humans and extended the relevance of our findings to the human disease phenotype. RANKL and OPG levels in naive ADA-deficient patients, displaying serious growth retardation, were significantly lower compared with healthy controls. GT with autologous gene-corrected stem cells, but not ERT, resulted in a significant increase in serum RANKL and the RANKL/OPG ratio. The less efficient correction of bone parameters in ADA-SCID patients compared with mice might be because all mice received treatment within the first 5 days of life, whereas patients were treated at variable ages. Moreover, ERT could be more efficient to correct the murine ADA bone phenotype because to rescue ADA−/− mice up to 50 times higher doses of PEG-ADA are required compared with standard doses in humans. Because OBs are not derived from HSCs, they are not expected to be corrected by either treatment. It is possible that endogenous ADA production by resident hematopoietic cells is more efficient to cross-correct OB function compared with circulating PEG-ADA. Moreover, patients treated with GT show superior immune reconstitution and lymphocyte counts with respect to patients treated with ERT,31 representing an important source of RANKL.
In conclusion, we report an ADA-specific bone phenotype, characterized by low bone mass accrual, size acquisition, and impaired mechanical competence, which is the result of an OB insufficiency with subsequent impact on the HSC niche. Our results emphasize the role of the ADA metabolism in modulating bone cell activities and add a stromal component to the series of immunologic defects described in ADA deficiency. The present data show that correction of the ADA bone phenotype is feasible with current treatment options, but longer follow-up in these patients will be needed to assess whether bone defects are resolvable over a longer period of time.
The online version of this article contains a data supplement.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 USC section 1734.
Acknowledgments
The authors thank Immacolata Brigida, Nicola Carriglio, Antonella Tabucchi, and Bernhard Gentner for technical assistance and helpful suggestions, Luciano Callegaro for patient data management, and the physicians and nurses of the Pediatric Clinical Research Unit, Italian Telethon Foundation.
This work was supported by the Italian Telethon Foundation (HSR-TIGET grant), the independent drug research program of the Italian Medicines Agency (grant FARM5JRXRM), and the European Commission: Concerted Safety and Efficiency Evaluation of Retroviral Transgenesis in Gene Therapy of Inherited Diseases (grant LSBH-CT-2004-005242).
Authorship
Contribution: A.V.S. designed and performed most research, analyzed data, and wrote the paper; E.M. designed and performed in vitro experiments and analyzed data; R.J.H. performed animal experiments; E.Z. performed pQCT analyses; F.C. performed histomorphometric analyses; M.C. collected patient data; E.G., C.M.R., and M.C.C. followed patients; A. Ambrosi performed the statistical analysis; F.C. conducted the biochemical studies; M.G.R. and A.V. contributed to the study design; A.R. designed the research and wrote the paper; A. Aiuti designed the research, analyzed data, and revised the paper; and all authors checked the final version of the manuscript.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: Alessandro Aiuti, San Raffaele Telethon Institute for Gene Therapy, Via Olgettina 58, 20132 Milano, Italy; e-mail: a.aiuti@hsr.it.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal