Abstract
Abstract 860
Elderly CML patients treated with Imatinib (IM) in early chronic phase (CP) have similar cytogenetic response and survival compared with younger patients, but they show a lower compliance to standard IM therapy (400 mg/day).
The aim of the study is to investigate if CCgR that has been achieved with standard (daily administration) IM therapy can be maintained with the same dose of IM given intermittently (INTERIM).
The study population is represented by elderly patients (≥ 65 years old) with Ph+ CML and with stable CCgR after at least 2 years of standard IM therapy (daily administration). IM is given at the same dose that was given at the time of enrollment by the following intermittent schedule: 1 week on / 1 week off for the 1st month; 2 weeks on / 2 weeks off for the 2nd and 3rd month; 1 month on / 1 month off from the 4th month thereafter. In cases of loss of CCgR INTERIM was stopped and standard therapy (daily administration) was resumed. After 12 months, the patients who are in continuous CCgR are advised to continue the intermittent study schedule and to be followed indefinitely. The CgR status was evaluated at baseline (by conventional cytogenetics on bone marrow and FISH on peripheral-blood) and every 3 months during the study (only by FISH on peripheral-blood). If FISH (% of Ph+ cells) increased more than 1% in two consecutive examinations, evaluation of marrow cells metaphases was performed to confirm the loss of CCgR and to check for additional cytogenetic abnormalities. Quantitative molecular assessment of BCR-ABL transcript by RQ-PCR on peripheral blood was due at baseline and every 3 months during the study and mutational analysis of ABL was performed in case of loss of CCgR.
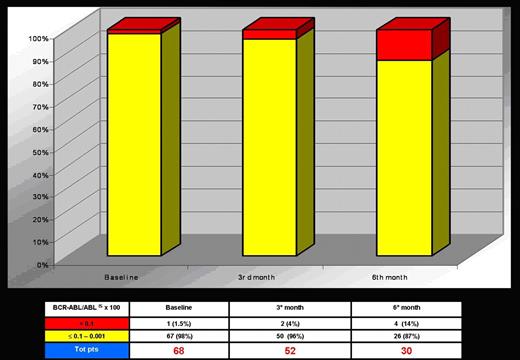
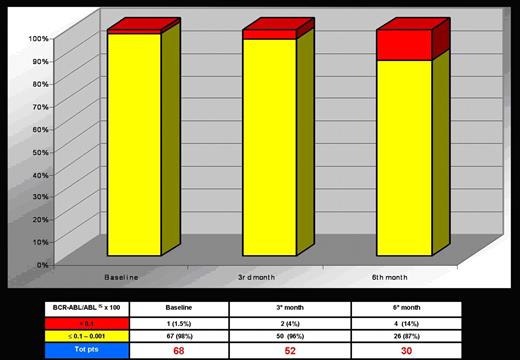
One-hundred and fourteen patients have been considered eligible, but 17 (15%) refused to enter into the protocol. Out of 97 enrolled patients, 87 started INTERIM, 5 patients (5%) went off the study for major protocol violation before the 3rd month and, at present, 82 patients are ongoing. Of these 82 patients, 52, 30 and 11 completed the 3rd, 6th and 9th month, respectively. The preliminary results of the first 6 months are here reported. The distribution of patients according to FISH results is shown in Fig. 1. Only 1/68 pts (at 6th month) showed an increased >1% in Ph+ cells by FISH but he maintained a CCgR when checked by conventional cytogenetic. As showed in Fig. 2, 96 to 87% of patients maintained a major molecular response MMR (≤0,1) according to International Scale (IS).
This study is trying to test the minimum effective dose of Imatinib to maintain the CCgR in elderly CML patients with stable CCgR. The preliminary results at 6 months do not show negative trends both for cytogenetic and molecular response. Therefore, the study is ongoing and all patients are expected to complete the trial time (12 months).
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal