Abstract
We investigated functional relationships between microRNA 221/222 (miR-221/222) cluster and p27, a key regulator of cell cycle, in chronic lymphocytic leukemia (CLL). The enforced expression of miR-221/222 in the CLL cell line MEC1 induced a significant down-regulation of p27 protein and conferred a proliferative advantage to the transduced cells that exhibited faster progression into the S phase of the cell cycle. Accordingly, expression of miR-221/miR-222 and p27 was found to be inversely related in leukemic cells obtained from peripheral blood (PB) of 38 patients with CLL. Interestingly, when miR-221/222 and p27 protein were evaluated in different anatomic compartments (lymph nodes or bone marrow) of the same patients, increased expression of the 2 miRNAs became apparent compared with PB. This finding was paralleled by a low expression of p27. In addition, when CLL cells were induced in vitro to enter cell cycle (eg, with cytosine phosphate guanine oligodeoxynucleotide), a significant increase of miR-221/222 expression and a marked down-regulation of p27 protein were evident. These data indicate that the miR-221/222 cluster modulates the expression of p27 protein in CLL cells and lead to suggest that miR-221/222 and p27 may represent a regulatory loop that helps maintaining CLL cells in a resting condition.
Introduction
Chronic lymphocytic leukemia (CLL), the most frequent leukemia among adults in the western world, is characterized by the progressive accumulation of mature monoclonal CD5+ malignant B lymphocytes in peripheral blood (PB), bone marrow (BM), and lymphoid organs.1 It is becoming increasingly evident that the ultimate phenotype of CLL results from a fine tuning of proliferation, survival, and reduced cell death that is provided both by a number of dysregulated genes and by microenvironmental stimuli.1-3
Circulating leukemic CLL cells are in the G0/early G1 phase of the cell cycle. Focal aggregates (proliferation centers [PCs]) of proliferating prolymphocytes and paraimmunoblasts can be identified in lymph nodes (LNs) and BM4 and represent the CLL proliferating reservoir that replenishes the downstream accumulation compartment. Proliferating cells differ from circulating resting leukemic lymphocytes in terms of expression of several molecules, including apoptosis-regulators such as Survivin,4 Noxa, and Mcl-15,6 ; chemokines such as CCL-17 and CCL-227 ; and proliferation-related genes such as Ki67.4 Another remarkable difference between cells circulating in the PB and those proliferating in the PCs is the expression of p27Kip1 (p27), a key regulator of cell cycle, which is very high in PB cells while it is virtually absent in PCs.8
p27 is encoded by the CDKN1B gene, a member of the Cip/Kip family of cyclin-dependent kinase inhibitors, that negatively controls cell-cycle progression (reviewed by Koff9 ). p27 binds to cyclin-dependent kinase 2 and cyclin E complexes to prevent cell-cycle progression from G1 to S phase.10 It is therefore highly expressed in cells arrested in G0/G1, and its expression declines as cells progress toward the S phase. Studies in several tumor types (reviewed in Chu et al11 ) indicate that p27 and its levels of expression have prognostic and potentially also therapeutic implications. As for CLL, previous studies12,13 have shown an increase of p27 in circulating CLL cells. More recently14 high expression of p27 has been found to predict rapid progression of the disease. Still the biologic significance of p27 in CLL is not entirely clear.
The mechanisms of regulation of p27 expression are numerous and range from its subcellular distribution9 to phosphorylation and subsequent ubiquitination and degradation events.15-18 A further level of control is represented by microRNA (miRNA) binding.19-23 miRNAs are an abundant class of small noncoding RNAs that modulate the expression of target mRNAs at the posttranscriptional level.24-27 Many miRNAs have been shown to play a role in cancer, as either oncogenes or tumor suppressor genes,28,29 and are preferentially located in genomic loci frequently altered in cancer.30 This is the case of the most frequent genetic aberration in CLL, the 13q14 deletion31 that involves the miRNAs miR-15a and miR-16-1.32 Interestingly, p27 expression has been shown to be regulated by miR-221/222 in different cancers, including glioblastoma, papillary thyroid carcinoma, prostate cancer, and liver cancer.20-22,33,34
All these data indicate that p27 and its regulation are relevant to CLL biology and provide the rationale for the present work, where we have investigated the functional relationships between p27 and the miR-221/222 cluster in CLL cells. We here show that the miR-221/222 cluster is able to modulate the expression of p27 protein and propose that miR-221/222 and p27 represent a regulatory loop that helps keeping CLL cells in a resting condition.
Methods
Cell culture and stimuli
MEC1 cell line was obtained from Deutsche Sammlung von Mikroorganismen und Zellkulturen GmbH and cultured in RPMI 1640 medium supplemented with 10% fetal calf serum (FCS), 2mM l-glutamine, and 15 μg/mL gentamicin (complete RPMI; Invitrogen).
Leukemic cells were cultured in RPMI 1640 medium supplemented with 10% FCS, 2mM l-glutamine, and 15 μg/mL gentamicin (complete RPMI; Invitrogen).
Purified leukemic B cells were cultured at a concentration of 3 × 106 cells/mL in the presence or absence of goat F(ab1)2 anti–human immunoglobulin M antibodies (10 μg/mL; Caltag Laboratories), and/or recombinant soluble CD40L according to the manufacturer's instructions (100 ng/mL CD40L + 1 μg/mL enhancer; Alexis Corporation), cytosine phosphate guanine oligodeoxynucleotide (2.5 μg/mL; stimulatory CpG-ODN type B, human specific; InvivoGen), recombinant human interleukin-2 (IL-2; 100 U/mL; PeproTech) for 24 hours. Cells were then either lysed and stored at −80°C or resuspended in TRIzol for RNA extraction or stained for immunofluorescence.
Tissue samples and cell purification
Leukemic lymphocytes were obtained from PB, BM, or LNs of patients with CLL diagnosed according to National Cancer Institute Working Group guidelines.35 All patients were either untreated or off therapy for at least 6 months before the beginning of the study. All tissue samples were obtained after informed consent, following the guidelines of the institutional review board of San Raffaele Scientific Institute.
Leukemic CD19+CD5+ cells were purified from PB and BM by negative selection with the use of either a B lymphocyte–enrichment kit (RosetteSep; StemCell Technologies) following the manufacturer's instructions, or purified with the use of magnetically activated cell sorting (MACS) MicroBeads (Miltenyi Biotec). LN specimens were dissected and homogenized on a metal sieve and purified with the use of MACS MicroBeads by negative selection. The coexpression of CD19 and CD5 on cell surfaces was checked by flow cytometry (FC500; Beckman Coulter); the purity of all preparations was always above 98%.
CD19+ B lymphocytes were purified from PB of healthy donors by negative selection with the use of a B lymphocyte–enrichment kit (RosetteSep; StemCell Technologies) following the manufacturer's instructions and further purified with the use of MACS MicroBeads (Miltenyi Biotec).
CD19+ B cells from discarded inflamed tonsils were purified with the use of MACS MicroBeads for negative selection. Purity of CD19+ lymphocytes was greater than 98%.
All tissue samples were obtained with the approval of the institutional Ethics Committee of San Raffaele Scientific Institute. Informed consent was provided according to the Declaration of Helsinki.
miR-expressing lentiviral vector production
MEC1 cell line cells were transduced with the self-inactivating pRRLsin-18.pptPGK-GFPwpre lentiviral (LV) vector containing a U3-deleted HIV-1 long terminal repeat and a phosphoglycerate kinase (PGK)–driven green fluorescent protein (GFP) cassette. To generate the miR-expressing LV vectors a fragment of approximately 700 base pairs (bp) corresponding to the desired miRNAs and the surrounding sequences was amplified from normal genomic DNA. We included a large portion of miRNA surrounding sequence in the attempt to allow correct processing of the miRNAs to their mature form and to induce overexpression while preserving a physiologic mechanism of miRNAs production. We cloned the amplified fragment into the SalI site in the expression vector, putting miRNA's sequences under the control of the strong PGK promoter. The vector also contained the enhanced GFP sequence, useful to evaluate transduction efficiency and to allow cell sorting. We used the same strategy to prepare different vectors, encoding for miR-221, miR-222, and mir-223, used as control.
Vesicular stomatitis virus G protein–pseudotyped LV particles were prepared by transient cotransfection of 293T cells, collected, and concentrated by standard procedures and titrated on 293T cells. Transduction efficiency was evaluated by flow cytometry (FC500; Beckman Coulter).
Western blot analysis
Cells were lysed with ice-cold lysis buffer (NaCl 0.15M; 1% NP40; 1mM EDTA, pH = 8; 50mM Tris-HCl, pH = 7) with fresh protease and phosphatase inhibitors (Pepstatin, leupeptin, phenylmethanesulfonylfluoride, sodium orthovanadate, and NaF). Whole proteins from cells were resolved by sodium dodecyl sulfate–polyacrylamide gel electrophoresis, and proteins from gel were electron-transferred onto nitrocellulose membranes and incubated with either anti-p27 (BD Biosciences) or anti–β-actin and horseradish peroxidase–conjugated (Sigma-Aldrich). Immunoreactivity was shown by incubation with goat anti–mouse immunoglobulin (Upstate Biotechnology) conjugated with horseradish peroxidase, followed by enhanced chemiluminescence reaction (Pierce Chemical) and film exposures. Protein expression was quantified by densitometric analysis performed with ImageQuant software (GE Healthcare) on images acquired with Personal Densitometer (NonLinear Dynamics).
Nucleofection of cells
With the use of the Nucleofector system (Amaxa Biosystems), specific anti-miRNAs were introduced into purified CLL B cells (1.2 × 107 cells) after resuspension in 100 μL of Cell Line Nucleofector Solution V. Then cells were mixed with 1 μg of the specific miRIDIAN miRNA-221 Inhibitor or miRIDIAN miRNA-222 Inhibitor (Dharmacon RNA Technologies). The same amount of miRIDIAN miRNA Inhibitor Negative Control was used as a negative control. Nucleofections were done with the use of the Amaxa Nucleofector device with the U-13 program. Transfection efficiency was assessed with the Block-iT Alexa Fluor Red Fluorescent Oligo (Invitrogen). CLL cells were collected and processed for immunoblotting analysis at 24 and 48 hours after nucleofection.
RNA extraction and real-time polymerase chain reaction
Total RNA was extracted from cells with the use of TRIzol (Invitrogen) according to the manufacturer's protocol.
Quantification of mature miRNAs was achieved by real-time polymerase chain reaction (PCR) with the TaqMan miRNA assay kit (Applied Biosystems) according to manufacturer's instruction. Briefly, 10 ng of total RNA was reverse-transcribed (RT) with specific primers, subsequently 1.5 μL of RT product was used as template for real-time PCR. All real-time experiments were performed in triplicate. Data were analyzed with the SDS2 software (Applied Biosystems) following the manufacturer-suggested comparative method. Data were normalized on the expression of small nuclear RNA (snRNA) U6 and expressed either as relative expression (2−ΔCt) or as fold change relative to control (2−ΔΔCt).
Cell synchronization
MEC1 cells were synchronized by the addition of 2mM thymidine (Sigma-Aldrich) for 18 hours, washed, and resuspended in complete RPMI, cultured for an additional 9 hours, and treated a second time with 2mM thymidine for an additional 12 hours. After the second incubation in thymidine, cells were washed twice in PBS to release from thymidine block and resuspended in complete RPMI. Cells were analyzed for cell-cycle distribution at the time of release and 4, 6 and 24 hours after the release.
Cell-cycle analysis
Transduced MEC1 cells were washed in PBS, resuspended in DNA staining buffer (PBS, 1% NP-40 with 300 Kuntz unit of RNAse and 50 μg/mL propidium iodide), incubated for 10 minutes at room temperature and analyzed for DNA content on a FC500 cytometer (Beckman Coulter). Data were analyzed with CXP analysis software (Beckman Coulter). Cell doublets and debris were excluded from further analysis by gating individual cells in peak area versus peak width plot.
Immunofluorescence and confocal microscopy
Purified cells from patient specimens (PB and LN/BM) were disseminated on a poly-L-ornithine (Sigma-Aldrich) precoated slides, fixed with PAF 4% (Sigma-Aldrich), and permeabilized with Triton X-100 0.3%. The cells were incubated overnight with primary antibodies (used at a 1:100 dilution in PBS containing 1 mg/mL bovine serum albumin and 10% FCS), and then stained with secondary antibodies (used at a 1:250 dilution). Primary antibodies were mouse anti-p27 monoclonal antibody (BD Biosciences) and rabbit anti–human Ki67 polyclonal antibody (Abcam).
Secondary antibodies were goat anti–mouse-conjugated with Alexa Fluor 488 for p27, goat anti–rabbit-conjugated with Alexa Fluor 633 for Ki67. We used propidium iodide (Sigma-Aldrich) for the nuclear staining. Cells were acquired with an inverted 63×/1.25 NA oil objective on a 3-laser (excitation wavelengths: 488, 546, and 633 nm) laser scanning confocal microscope (Radiance 2100; Bio-Rad). Fluorescent signals from single optical sections were sequentially acquired and analyzed by Paint Shop Pro 7.02 (Jasc Software).
Statistical analysis
Statistical analyses were performed with GraphPad Prism Version 4.00 (GraphPad Software). Spearman correlation analysis was performed between miR-221/222 and p27 levels. Differences in cell-cycle phase distribution were assessed with 2-way analysis of variance and Bonferroni test. Differences in the expression of p27 and miR-221/222 between groups were evaluated with the Wilcoxon test. An effect was considered to be statistically significant at P values less than .05.
Results
Inducing overexpression of miR-221 and miR-222 in CLL cells
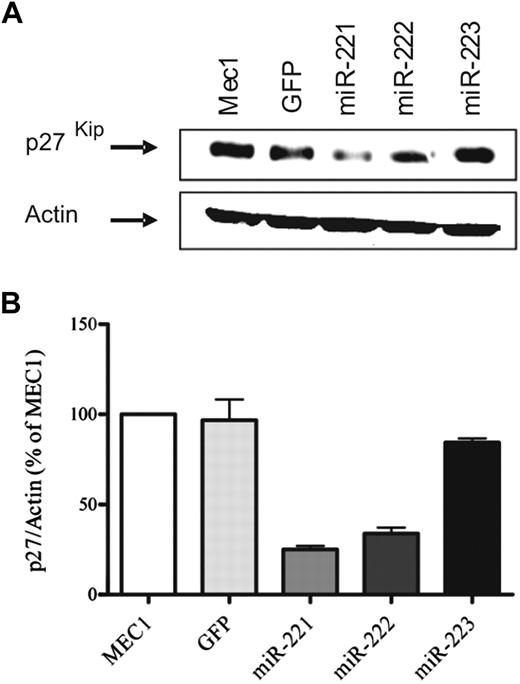
We used the CLL cell line MEC1 that constitutively expresses low levels of miR-221 and miR-222 (Figure 1A). To overexpress these 2 miRNAs and miR-223, used as control, we selected a LV vector and cloned the miRNA-containing region in a PGK-driven enhanced GFP cassette. Transduction efficiency ranged from 50% to 80%; after sorting GFP-positive cells we obtained a homogeneous population stably expressing the corresponding transduced miRNA, as assessed by TaqMan miRNA Assay. The level of miR-221 and miR-222 expression was 3 and 12 times higher, respectively, compared with GFP-only–transduced cells (Figure 1A). Cells transduced with miR-223 vector significantly overexpressed it (data not shown).
Characterization of miR-221/222–overexpressing MEC1 cell lines. MEC1 cells were infected with miR-expressing LV vectors. Infected cells were sorted, and overexpression of miR-221/222 was assessed by real-time PCR (A). Control and miR-transduced cells were plated at the concentration of 5 × 105/mL and counted by Trypan-blue exclusion at the indicated time points (B left). The experiment was repeated after synchronization by double thymidine block and plating (B right). Synchronized cells were stained with propidium iodide at the indicated time points after block release, and cell-cycle distribution was analyzed by flow cytometry. Percentage of cells in G1, S, and G2/M phase of the cell cycle is shown (C). Graph represents the average of 3 different experiments (mean + SD). Differences were analyzed by 2-way analysis of variance and Bonferroni test.
Characterization of miR-221/222–overexpressing MEC1 cell lines. MEC1 cells were infected with miR-expressing LV vectors. Infected cells were sorted, and overexpression of miR-221/222 was assessed by real-time PCR (A). Control and miR-transduced cells were plated at the concentration of 5 × 105/mL and counted by Trypan-blue exclusion at the indicated time points (B left). The experiment was repeated after synchronization by double thymidine block and plating (B right). Synchronized cells were stained with propidium iodide at the indicated time points after block release, and cell-cycle distribution was analyzed by flow cytometry. Percentage of cells in G1, S, and G2/M phase of the cell cycle is shown (C). Graph represents the average of 3 different experiments (mean + SD). Differences were analyzed by 2-way analysis of variance and Bonferroni test.
miR-221 and miR-222 regulate cell-cycle progression and p27 expression in MEC1 cell lines
To evaluate the functional changes after miR-221 and miR-222 transduction and overexpression, we first analyzed the growth capacity of the different transduced cell lines. After 24, 48, 72, and 96 hours of in vitro culture, we observed an increased cell number in the case of miR-221/222–transduced cell lines compared with untransduced or GFP-only–transduced cells (Figure 1B left), based on cell count by Trypan Blue dye exclusion. We observed an increase of 24% and 21%, respectively, in the number of miR-221– and miR-222–transduced cells recovered after 4 days. The effect of miR-221 and miR-222 on cell proliferation became even more evident on cell synchronization by thymidine block before plating and counting (Figure 1B right). In this case, after 4 days we recovered 50% more miR-222 cells and 28% more miR-221 cells compared with control (P < .005 for miR-221; P < .05 for miR-222).
To exclude that these differences could be due to anomalies in the apoptotic behavior, we analyzed the percentage of apoptotic cells in the population by annexin V–7-amino actinomycin D staining. No appreciable differences between the percentage of apoptotic cells in the GFP-only–transduced population and the miR-221/222–transduced cell population were detected (data not shown). The same results were obtained in the condition of serum deprivation (data not shown).
To better clarify the mechanism for the increase in cell number, we again synchronized cells in the G1 phase and analyzed by flow cytometry the cell-cycle distribution at 4, 6, and 24 hours after cell-cycle block release. At every time point analyzed, miR-221– and miR-222–transduced cells had a lower percentage of cells in the G1 phase and a higher percentage of cells in the S phase and in the G2/M phase (Figure 1C). In details, after 4 hours from the block release miR-221– and miR-222–transduced cells had, respectively, 19.7% (± 1.3%) and 19.9% (± 0.3%) of cells in G0/G1 phase of the cell cycle, whereas the percentage of control-transduced cells in G0/G1 phase was 30.9% (± 1.5%) (P < .05; Figure 1D). Six hours after block release the percentages of cells in G0/G1 phase were 37.3% (± 3.6%) (control cells), 23.2% (± 4.0%) (miR-221) and 26.1% (± 3.4%) (miR-222; P < .05 for miR-221; P < .01 for miR-222). Similar differences were maintained also after 24 hours; the percentage of cells in G0/G1 being 58.7% (± 3.0%) (control), 46.4% (± 0.4%) (miR-221), and 47.9% (± 3.7%) (miR-222; P < .05; Figure 1D). These data document that miR-221– and miR-222–transduced cells progressed more quickly through the cell-cycle phases.
Because miR-221/222 modulates the expression of the cell-cycle inhibitor p27 in different cell types,20,22 we evaluated this possibility in our experimental model. The immunoblot analysis of whole-cell lysate from MEC1 cells and MEC1-transduced cells clearly showed a marked reduction of p27 protein expression in miR-221– and miR-222–transduced cells (Figure 2A). Densitometric analysis showed that the levels were 80% lower than controls (Figure 2B).
miR-221/222 transduction decreases p27 expression in MEC1 cells. Total cell lysate from parental MEC1 cells or MEC1 transduced with vectors encoding miR-221/222 and 223, as control, were subjected to Western blot analysis. Blots were stained with anti-p27 antibody and then stripped and reprobed with β-actin as loading control. A representative blot is shown (A). Densitometric analysis shows the entity of reduction in p27 expression. The average of 3 independent experiments is shown in the graph (mean + SD; B).
miR-221/222 transduction decreases p27 expression in MEC1 cells. Total cell lysate from parental MEC1 cells or MEC1 transduced with vectors encoding miR-221/222 and 223, as control, were subjected to Western blot analysis. Blots were stained with anti-p27 antibody and then stripped and reprobed with β-actin as loading control. A representative blot is shown (A). Densitometric analysis shows the entity of reduction in p27 expression. The average of 3 independent experiments is shown in the graph (mean + SD; B).
miR221/222 and p27 expression are inversely related in primary CLL cells
On the basis of the results obtained in the MEC1 cell line, we analyzed primary CLL leukemic lymphocytes from PB. We first evaluated the expression of p27 protein on purified B lymphocytes from 38 patients with CLL and confirmed by Western blot that the protein was readily detectable in all patients analyzed. The levels of expression were rather variable, although readily detectable in all cases, as quantified by densitometric analysis (Figure 3A).
Expression of p27 and miR-221/222 inversely correlate in CLL cells from patients. Western blot experiment with the use of leukemic cells of 5 representative CLL cases shows variable, although readily detectable, levels of expression of p27 protein (A top). β-Actin is used as loading control. Quantification by densitometric analysis expression is reported as p27/Actin ratio between OD (A bottom). Quantitative PCR analysis for miR-221 and miR-222 (TaqMan miRNA assay) on the same patients are reported (B (top and bottom, respectively). (C) The negative correlation trendline between miR-221 (left) or miR-222 (right) and p27 in 38 patients with CLL.
Expression of p27 and miR-221/222 inversely correlate in CLL cells from patients. Western blot experiment with the use of leukemic cells of 5 representative CLL cases shows variable, although readily detectable, levels of expression of p27 protein (A top). β-Actin is used as loading control. Quantification by densitometric analysis expression is reported as p27/Actin ratio between OD (A bottom). Quantitative PCR analysis for miR-221 and miR-222 (TaqMan miRNA assay) on the same patients are reported (B (top and bottom, respectively). (C) The negative correlation trendline between miR-221 (left) or miR-222 (right) and p27 in 38 patients with CLL.
Next, we analyzed on the same series of patients the expression levels of the 2 miRNAs by quantitative real-time PCR. In addition, miR-221 and miR-222 showed different levels of expression in different patients (Figure 3B), but, interestingly, their pattern of expression was reciprocal with the pattern of p27 protein (Figure 3A-B). When the expression values of all 38 patients were plotted on a graph, the inverse correlation between miR-221/222 and p27 expression was confirmed and statistically significant (Figure 3C; Spearman r = −0.34, P < .05 for miR-221; r = −0.33, P < .05 for miR-222).
When clinical and biologic features were analyzed (Table 1), we found that the expression of miR-222 correlated with the IGHV gene mutational status, because the presence of unmutated IGHV genes was associated with higher miR-222 expression (P < .05).
Clinical and biologic features of the patients with CLL analyzed
| Patient . | Age at diagnosis, y . | Sex . | Stage* . | IGVH† . | CD38, % . | ZAP70‡ . | Disease progression . | miR-221§ . | miR-222§ . | FISH‖ . |
|---|---|---|---|---|---|---|---|---|---|---|
| Pt 1 | 69 | M | 0/A | 93.7 | 49.7 | Pos | S | 0.070 | 1.480 | 13q deletion (biallelic) |
| Pt 2 | 69 | M | 2/B | 97.9 | 71.4 | Neg | P | 0.083 | 1.344 | 13 q deletion |
| Pt 3 | 67 | M | 1/A | 95.8 | 0.8 | Pos | P | 0.197 | 3.637 | Normal |
| Pt 5 | 44 | F | 0/A | 100.0 | 3.2 | Neg | P | 0.031 | 1.836 | Normal |
| Pt 9 | 65 | M | 0/A | 95.8 | 4.3 | Neg | S | 0.253 | 3.083 | 13 q deletion |
| Pt 10 | 67 | F | 1/B | 98.3 | 3.7 | Neg | P | 0.172 | 2.692 | 11q deletion |
| Pt 11 | 80 | M | 1/A | 95.8 | 1.0 | Pos | P | 0.037 | 0.562 | 17p deletion |
| Pt 14 | 69 | M | 0/A | 100.0 | 1.8 | Pos | S | 0.041 | 0.380 | 13 q deletion |
| Pt 15 | 56 | M | 0/A | 96.5 | 0.2 | Neg | S | 0.663 | 3.060 | 17 p deletion |
| Pt 16 | 49 | M | 1/A | 98.3 | 2.3 | Pos | S | 0.284 | 3.242 | 17p deletion |
| Pt 17 | 76 | F | 3/C | 100.0 | 13.3 | Pos | P | 0.029 | 1.520 | 17p deletion |
| Pt 19 | 70 | F | 0/A | 88.5 | 0.1 | Neg | S | 0.067 | 1.983 | 13 q deletion (biallelic) |
| Pt 20 | 68 | M | 2/A | 100.0 | 3.7 | Pos | P | 0.204 | 2.933 | 17p deletion |
| Pt 22 | 69 | F | 1/A | 100.0 | 0.2 | Pos | S | 0.122 | 1.596 | 13 q deletion |
| Pt 24 | 69 | M | 2/B | 90.6 | 0.0 | Neg | P | 0.039 | 0.354 | 13 q deletion |
| Pt 27 | 66 | M | NA | 91.5 | 2.0 | Neg | P | 0.054 | 1.762 | 13 q deletion |
| Pt 29 | 60 | F | 0/A | 91.7 | 0.7 | Neg | P | 0.264 | 2.747 | Not done |
| Pt 30 | 69 | M | 0/A | 100.0 | 59.5 | Pos | P | 0.161 | 5.129 | 13 q deletion |
| Pt 33 | 59 | F | NA | 92.0 | 12.3 | Neg | P | 0.040 | 0.696 | Not done |
| Pt 34 | 59 | F | 0/A | 100.0 | 10.0 | Neg | S | 0.229 | 3.219 | 11q deletion |
| Pt 35 | 58 | F | 1/A | 97.2 | 7.0 | Neg | P | 0.179 | 2.490 | 13 q deletion |
| Pt 40 | 53 | M | 2/B | 100.0 | 59.4 | Pos | P | 0.853 | 2.824 | 17p deletion |
| Pt 44 | 76 | M | 2/A | 100.0 | 92.0 | Pos | S | 1.003 | 2.035 | 12q trisomy |
| Pt 45 | 57 | F | 0/A | 96.9 | 72.3 | Neg | S | 0.951 | 1.785 | 17p deletion |
| Pt 48 | 49 | M | 4/C | 100.0 | 92.4 | Pos | P | 1.084 | 3.528 | 11q deletion |
| Pt 49 | 53 | F | 1/B | 100.0 | 6.7 | Neg | P | 0.791 | 5.300 | 13 q deletion |
| Pt 50 | 61 | M | 0/A | 100.0 | 11.0 | Neg | S | 0.663 | 4.980 | 11q deletion |
| Pt 51 | 39 | M | 1/A | 97.2 | 2.1 | Neg | P | 0.458 | 3.520 | Not done |
| Pt 56 | 57 | F | 1/B | 100.0 | 62.7 | Pos | P | 0.006 | 0.470 | 17p deletion |
| GR30 | 47 | M | 0/A | 95.9 | 0.7 | Neg | S | 0.549 | 2.059 | Not done |
| GR31 | 56 | F | 0/A | 90.3 | 1.6 | Neg | S | 0.135 | 0.806 | Normal |
| GR32 | 63 | M | 0/A | 93.4 | 3.3 | Neg | S | 0.056 | 1.542 | 13q deletion |
| GR33 | 63 | F | 2/A | 88.7 | 66.4 | Pos | S | 0.100 | 0.946 | 12 trisomy |
| GR35 | 38 | F | 0/A | 94.1 | 1.0 | Neg | S | 0.840 | 3.611 | 13 q deletion (biallelic) |
| GR36 | 60 | M | 1/A | 89.2 | 0.9 | Neg | S | 0.135 | 1.477 | Not done |
| GR37 | 52 | F | 1/A | 95.6 | 1.8 | Neg | S | 0.466 | 2.316 | Not done |
| GR39 | 53 | M | 0/A | 92.9 | 5.3 | Not done | S | 0.183 | 1.176 | 13 q deletion (biallelic) |
| GR40 | 47 | M | 0/A | 94.8 | 2 | Neg | S | 0.512 | 3.014 | 13q deletion |
| Patient . | Age at diagnosis, y . | Sex . | Stage* . | IGVH† . | CD38, % . | ZAP70‡ . | Disease progression . | miR-221§ . | miR-222§ . | FISH‖ . |
|---|---|---|---|---|---|---|---|---|---|---|
| Pt 1 | 69 | M | 0/A | 93.7 | 49.7 | Pos | S | 0.070 | 1.480 | 13q deletion (biallelic) |
| Pt 2 | 69 | M | 2/B | 97.9 | 71.4 | Neg | P | 0.083 | 1.344 | 13 q deletion |
| Pt 3 | 67 | M | 1/A | 95.8 | 0.8 | Pos | P | 0.197 | 3.637 | Normal |
| Pt 5 | 44 | F | 0/A | 100.0 | 3.2 | Neg | P | 0.031 | 1.836 | Normal |
| Pt 9 | 65 | M | 0/A | 95.8 | 4.3 | Neg | S | 0.253 | 3.083 | 13 q deletion |
| Pt 10 | 67 | F | 1/B | 98.3 | 3.7 | Neg | P | 0.172 | 2.692 | 11q deletion |
| Pt 11 | 80 | M | 1/A | 95.8 | 1.0 | Pos | P | 0.037 | 0.562 | 17p deletion |
| Pt 14 | 69 | M | 0/A | 100.0 | 1.8 | Pos | S | 0.041 | 0.380 | 13 q deletion |
| Pt 15 | 56 | M | 0/A | 96.5 | 0.2 | Neg | S | 0.663 | 3.060 | 17 p deletion |
| Pt 16 | 49 | M | 1/A | 98.3 | 2.3 | Pos | S | 0.284 | 3.242 | 17p deletion |
| Pt 17 | 76 | F | 3/C | 100.0 | 13.3 | Pos | P | 0.029 | 1.520 | 17p deletion |
| Pt 19 | 70 | F | 0/A | 88.5 | 0.1 | Neg | S | 0.067 | 1.983 | 13 q deletion (biallelic) |
| Pt 20 | 68 | M | 2/A | 100.0 | 3.7 | Pos | P | 0.204 | 2.933 | 17p deletion |
| Pt 22 | 69 | F | 1/A | 100.0 | 0.2 | Pos | S | 0.122 | 1.596 | 13 q deletion |
| Pt 24 | 69 | M | 2/B | 90.6 | 0.0 | Neg | P | 0.039 | 0.354 | 13 q deletion |
| Pt 27 | 66 | M | NA | 91.5 | 2.0 | Neg | P | 0.054 | 1.762 | 13 q deletion |
| Pt 29 | 60 | F | 0/A | 91.7 | 0.7 | Neg | P | 0.264 | 2.747 | Not done |
| Pt 30 | 69 | M | 0/A | 100.0 | 59.5 | Pos | P | 0.161 | 5.129 | 13 q deletion |
| Pt 33 | 59 | F | NA | 92.0 | 12.3 | Neg | P | 0.040 | 0.696 | Not done |
| Pt 34 | 59 | F | 0/A | 100.0 | 10.0 | Neg | S | 0.229 | 3.219 | 11q deletion |
| Pt 35 | 58 | F | 1/A | 97.2 | 7.0 | Neg | P | 0.179 | 2.490 | 13 q deletion |
| Pt 40 | 53 | M | 2/B | 100.0 | 59.4 | Pos | P | 0.853 | 2.824 | 17p deletion |
| Pt 44 | 76 | M | 2/A | 100.0 | 92.0 | Pos | S | 1.003 | 2.035 | 12q trisomy |
| Pt 45 | 57 | F | 0/A | 96.9 | 72.3 | Neg | S | 0.951 | 1.785 | 17p deletion |
| Pt 48 | 49 | M | 4/C | 100.0 | 92.4 | Pos | P | 1.084 | 3.528 | 11q deletion |
| Pt 49 | 53 | F | 1/B | 100.0 | 6.7 | Neg | P | 0.791 | 5.300 | 13 q deletion |
| Pt 50 | 61 | M | 0/A | 100.0 | 11.0 | Neg | S | 0.663 | 4.980 | 11q deletion |
| Pt 51 | 39 | M | 1/A | 97.2 | 2.1 | Neg | P | 0.458 | 3.520 | Not done |
| Pt 56 | 57 | F | 1/B | 100.0 | 62.7 | Pos | P | 0.006 | 0.470 | 17p deletion |
| GR30 | 47 | M | 0/A | 95.9 | 0.7 | Neg | S | 0.549 | 2.059 | Not done |
| GR31 | 56 | F | 0/A | 90.3 | 1.6 | Neg | S | 0.135 | 0.806 | Normal |
| GR32 | 63 | M | 0/A | 93.4 | 3.3 | Neg | S | 0.056 | 1.542 | 13q deletion |
| GR33 | 63 | F | 2/A | 88.7 | 66.4 | Pos | S | 0.100 | 0.946 | 12 trisomy |
| GR35 | 38 | F | 0/A | 94.1 | 1.0 | Neg | S | 0.840 | 3.611 | 13 q deletion (biallelic) |
| GR36 | 60 | M | 1/A | 89.2 | 0.9 | Neg | S | 0.135 | 1.477 | Not done |
| GR37 | 52 | F | 1/A | 95.6 | 1.8 | Neg | S | 0.466 | 2.316 | Not done |
| GR39 | 53 | M | 0/A | 92.9 | 5.3 | Not done | S | 0.183 | 1.176 | 13 q deletion (biallelic) |
| GR40 | 47 | M | 0/A | 94.8 | 2 | Neg | S | 0.512 | 3.014 | 13q deletion |
CLL indicates chronic lymphocytic leukemia; IGVH, immunoglobulin heavy-chain variable region; FISH, fluorescent in situ hybridization; Pt, patient; Pos, positive; Neg, negative; P, progressive; and S, stable.
Shown as percentage of similarity to the closest germline gene.
Based on the cutoff of 20%.38
miR-221 and mi-222 values as detected by TaqMIRNA Assay from PB samples.
According to the hierarchical model of Döhner et al.31
miR-221/222 are differentially expressed in different anatomical compartments
We then evaluated p27 levels and miR-221/222 expression, by Western blot and RT-PCR, respectively, in purified B lymphocytes obtained from both PB and LN/BM from the same patients with CLL.
We collected a set of 7 paired samples (PB and LN/BM) and found that the expression of p27 was reduced in the LN/BM (Figure 4A; average expression 1.69 ± 0.43 vs 3.74 ± 1.16 in LN/BM vs PB; P < .05). This reduction was paralleled by the increased expression of the 2 miRNAs (Figure 4B; average expression for miR-221: 1.16 ± 0.49 vs 0.35 ± 0.14; average expression for miR-222: 5.73 ± 1.31 vs 2.13 ± 0.41; P < .05). The levels of both miRNAs in normal PB and tonsillar B lymphocytes were low, similarly to those of PB CLL cells; those of LN/BM CLL samples were the highest found (data not shown).
Differential expression of p27 and miR-221/222 in different CLL tissue compartments. Western blot analysis with the use of whole-cell lysates of purified CLL B cells from paired LN/BM and PB obtained from 7 patients. The blots were probed with anti-p27 antibodies; β-actin was used as control for equal protein loading. A representative case is shown (A top). The differences in p27 expression are quantified by densitometric analysis (A bottom). Quantitative PCR analysis (B) for miR-221 and miR-222 (TaqMan miRNA assay) quantifies the differences in expression between purified leukemic cells from paired LN/BM and PB from the same 7 patients depicted in panel A. Statistical significant differences were evaluated by the Wilcoxon test. Immunofluorescent staining with p27 (green), propidium iodide (PI; red), and Ki67 (blue) of CLL cells purified from LNs (first and third columns, 63× and 240× magnification, respectively) and PB cells (second and fourth columns, 63× and 240× magnification, respectively) of a representative patient with CLL (C; 63×/1.25 NA oil objective). LN cells exhibit lower levels of p27 in comparison to PB cells. Ki67 staining could detect positive cells only in LN cells, whereas PB cells are negative.
Differential expression of p27 and miR-221/222 in different CLL tissue compartments. Western blot analysis with the use of whole-cell lysates of purified CLL B cells from paired LN/BM and PB obtained from 7 patients. The blots were probed with anti-p27 antibodies; β-actin was used as control for equal protein loading. A representative case is shown (A top). The differences in p27 expression are quantified by densitometric analysis (A bottom). Quantitative PCR analysis (B) for miR-221 and miR-222 (TaqMan miRNA assay) quantifies the differences in expression between purified leukemic cells from paired LN/BM and PB from the same 7 patients depicted in panel A. Statistical significant differences were evaluated by the Wilcoxon test. Immunofluorescent staining with p27 (green), propidium iodide (PI; red), and Ki67 (blue) of CLL cells purified from LNs (first and third columns, 63× and 240× magnification, respectively) and PB cells (second and fourth columns, 63× and 240× magnification, respectively) of a representative patient with CLL (C; 63×/1.25 NA oil objective). LN cells exhibit lower levels of p27 in comparison to PB cells. Ki67 staining could detect positive cells only in LN cells, whereas PB cells are negative.
The expression of p27 was also analyzed by immunofluorescence in B lymphocytes purified from the PB and LN/BM obtained from the same patients with CLL. PB leukemic B lymphocytes were found to homogeneously express high levels of p27 that appeared to be mainly if not exclusively localized in the nucleus (Figure 4C). In contrast, leukemic B cells from LN/BM were found to express much lower levels of p27. Conversely, although all PB cells were Ki67-negative, LN/BM cells were heterogeneous in terms of proliferation potential as indicated by Ki67 staining (Figure 4C). Interestingly, LN Ki67-positive cells expressed lower levels of p27.
Because we initially demonstrated that overexpression of miR221/222 was associated with a reduction in p27 levels (Figure 2), we next asked how blocking miR-221/222 action would affect p27 expression in the cells with higher expression of the 2 miRNAs, namely cells from the LN/BM compartment. The forced expression of anti–miR-221 and anti–miR-222 in BM CLL cells increased the levels of p27 compared with cells transfected with negative control sequences (supplemental Figure 1, available on the Blood Web site; see the Supplemental Materials link at the top of the online article).
miR-221/222 and p27 expression after proliferative signals in CLL cells
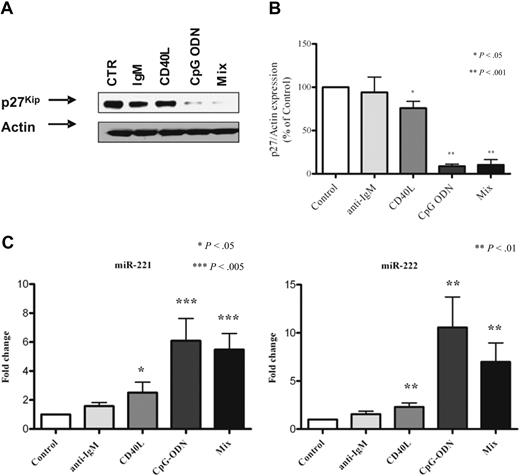
We investigated the possibility that miR-221/222 and p27 might be modulated after B-cell activation induced by ligands such as anti-immunoglobulin M and CD40L, either alone or in combination with CpG-ODN. Purified B cells from PB of 10 patients with CLL were analyzed, and, among the stimuli provided, CpG (with or without additional stimuli) resulted in the most marked down-regulation of p27 protein (average reduction, 93% for CpG alone; P < .001). CD40L costimulation produced a significant although lower effect (average reduction 25%; P < .05), whereas B-cell receptor engagement produced virtually no effect (Figure 5A-B). The combination of the 3 stimuli did not significantly modify the response (90%; P < .001).
Exogenous stimuli down-regulate p27 and up-regulate miR221/222 in CLL cells. Protein lysates from purified CLL cells of 1 representative case stimulated with anti-immunoglobulin M (IgM) antibodies, CD40L, CpG-ODN, and the mix of the 3 reagents were subjected to Western blot. Blots were probed with anti-p27 antibody and then stripped and reprobed with β-actin as loading control (A). Mean values of densitometric analysis from 10 different patients are reported (B). Bars represent SE. Asterisks mark significant reduction for CD40L, CpG-ODN, and mix. TaqMan miRNA assay was used to detect miR-221 (C left) and miR-222 (C right) levels in control and stimulated cells. The mean value in 10 patients is reported. The average miR-221 induction was 1.4 ± 0.2 (anti-IgM), 2.5 ± 0.7 (CD40L), 6.1 ± 1.5 (CpG-ODN); and 5.5 ± 1.1 (mix of the 3 stimuli), and the average miR-222 induction was 1.6 ± 0.3 (anti-IgM), 2.3 ± 0.4 (CD40L), 10.6 ± 3.2 (CpG-ODN), and 6.9 ± 1.9 (mix of the 3 stimuli). Bars represent SE. Significance is indicated by asterisks. Statistics were calculated with the Wilcoxon test.
Exogenous stimuli down-regulate p27 and up-regulate miR221/222 in CLL cells. Protein lysates from purified CLL cells of 1 representative case stimulated with anti-immunoglobulin M (IgM) antibodies, CD40L, CpG-ODN, and the mix of the 3 reagents were subjected to Western blot. Blots were probed with anti-p27 antibody and then stripped and reprobed with β-actin as loading control (A). Mean values of densitometric analysis from 10 different patients are reported (B). Bars represent SE. Asterisks mark significant reduction for CD40L, CpG-ODN, and mix. TaqMan miRNA assay was used to detect miR-221 (C left) and miR-222 (C right) levels in control and stimulated cells. The mean value in 10 patients is reported. The average miR-221 induction was 1.4 ± 0.2 (anti-IgM), 2.5 ± 0.7 (CD40L), 6.1 ± 1.5 (CpG-ODN); and 5.5 ± 1.1 (mix of the 3 stimuli), and the average miR-222 induction was 1.6 ± 0.3 (anti-IgM), 2.3 ± 0.4 (CD40L), 10.6 ± 3.2 (CpG-ODN), and 6.9 ± 1.9 (mix of the 3 stimuli). Bars represent SE. Significance is indicated by asterisks. Statistics were calculated with the Wilcoxon test.
In parallel, we checked miR-221/222 levels and observed an up-regulation of miR-221/222 (Figure 5C) in all conditions, which was statistically significant in the case of CpG (P < .002 for miR-221; P < .006 for miR-222) and CD40L (P < .02 for miR-221; P < .006 for miR-222). Again, the combination of the 3 stimuli did not improve the effect, compared with CpG alone.
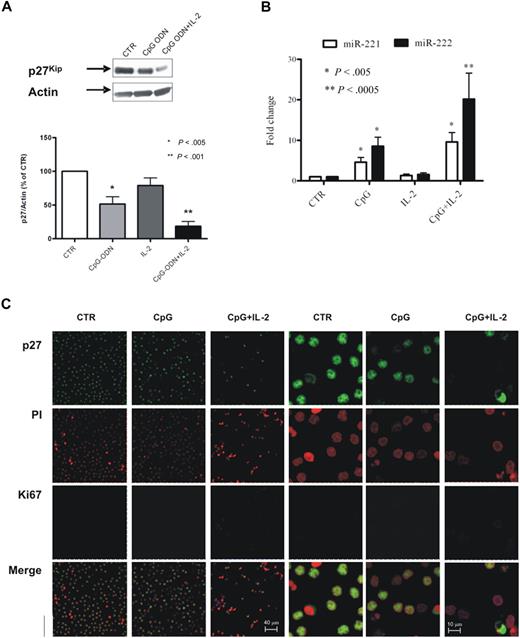
On the basis of these results and on the previous notion that the addition of IL-2 increases the stimulatory effect of CpG, we stimulated B cells from 16 additional patients with CLL with CpG-ODN alone (10 cases) or in combination with IL-2 (6 cases) and analyzed p27 and miR-221/222 expression. The average decrease of p27 expression was close to 50% for CpG alone and almost 80% for CpG plus IL-2 (relative p27/actin expression being 51.4 ± 10.9 for CpG alone and 18.2 ± 7.3 for CpG + IL-2, compared with 100% of control), as quantified by densitometric analysis on Western blot analysis (Figure 6A). In parallel, we also evaluated by quantitative RT-PCR the expression levels of miR-221/222 and detected a significant increase of miR-221/222 expression in treated cells especially in the presence of IL-2 (Figure 6B). The average miR-221 levels were 4.6 (± 1.1) and 9.2 (± 2.3) times higher in CpG and CpG plus IL-2–stimulated cells, respectively (P < .005); for miR-222 the average level of induction was 8.5 (± 2.2) for CpG alone (P < .005) and 20.2 (± 6.4) in combination with IL-2 (P < .001).
CpG-ODN–stimulated CLL cells from PB exhibit a pattern of expression similar to leukemic cells from LNs. Purified PB leukemic B cells were stimulated with CpG-ODN alone or in combination with IL-2, and p27 expression levels were evaluated by Western blot. A representative blot is shown (A top). The average result from analysis of stimulation on 16 patients is shown as p27/Actin ratio calculated by densitometric analysis (A bottom). miRNA expression in the cells from the same experiments was evaluated by TaqMan miRNA assay, and the results were expressed as fold change (2−ΔΔCt; B). Statistical significance was evaluated by the Wilcoxon test. Purified CLL PB cells stimulated with CpG-ODN alone or in combination with IL-2 were stained with anti-p27 antibody (green), propidium iodide (PI; red) to identify nuclei, and Ki67 (blue) antibody (C; 63×/1.25 NA oil objective). Confocal analysis of stained cells shows, compared with unstimulated cells (first and fourth columns, 63× and 240× magnification, respectively), a marked reduction of p27 expression and a parallel increase in Ki67 expression in cells stimulated with CpG-ODN (second and fifth columns, 63× and 240×, respectively) and in combination with IL-2 (third and sixth columns, 63× and 240×, respectively). CTR indicates control.
CpG-ODN–stimulated CLL cells from PB exhibit a pattern of expression similar to leukemic cells from LNs. Purified PB leukemic B cells were stimulated with CpG-ODN alone or in combination with IL-2, and p27 expression levels were evaluated by Western blot. A representative blot is shown (A top). The average result from analysis of stimulation on 16 patients is shown as p27/Actin ratio calculated by densitometric analysis (A bottom). miRNA expression in the cells from the same experiments was evaluated by TaqMan miRNA assay, and the results were expressed as fold change (2−ΔΔCt; B). Statistical significance was evaluated by the Wilcoxon test. Purified CLL PB cells stimulated with CpG-ODN alone or in combination with IL-2 were stained with anti-p27 antibody (green), propidium iodide (PI; red) to identify nuclei, and Ki67 (blue) antibody (C; 63×/1.25 NA oil objective). Confocal analysis of stained cells shows, compared with unstimulated cells (first and fourth columns, 63× and 240× magnification, respectively), a marked reduction of p27 expression and a parallel increase in Ki67 expression in cells stimulated with CpG-ODN (second and fifth columns, 63× and 240×, respectively) and in combination with IL-2 (third and sixth columns, 63× and 240×, respectively). CTR indicates control.
We finally analyzed by immunofluorescence the cellular distribution of p27 and Ki67 in stimulated cells. CpG in association with IL-2 induced a marked down-regulation of p27 with a concomitant increase of Ki67 expression (Figure 6C), resembling the pattern observed in leukemic CLL cells purified from infiltrated tissues (LNs or BM).
Discussion
In this study we have investigated the expression of miR-221/miR-222 and their functional relationship with p27 in CLL.
p27 has a central role in cancer11 with profound implications in biologic, prognostic, and therapeutic terms. The miR-221/miR-222 cluster has been shown to regulate p27 protein expression in different types of cancer, such as glioblastoma, papillary thyroid carcinoma, prostate cancer, and liver cancer.20-22,34 We used the MEC1 cell line and primary leukemic cells from patients with CLL and confirmed that regulation of p27 protein by miR-221 and miR-222 occurs in CLL B lymphocytes as well.
An extensive study of miRNA expression in patients with CLL showed39-41 a general down-regulation of miRNA expression, including miR-222.40-42 Further, miR-221 and miR-222 have been related to clinical aspects such as time to first treatment,41 although no functional explanation has been provided.
Indeed, the functional behavior of miR-221/222–transduced MEC1 cells points to a deregulation of cell-cycle machinery as indicated by the improvement in the G1/S transition and the faster growing rate of miR-221– and miR-222–expressing cells, and both events are consistent with a down-modulation of p27 activity.
This inverse correlation between these molecules has been also observed in primary leukemic cells from the PB of patients with CLL, whereby the pattern of expression of miR-221, miR-222, and p27 is reciprocal, with the former expressed at low levels whereas the latter markedly expressed. In clear contrast, the comparative analysis of the expression levels of miR-221/222 and p27 protein in other anatomic compartments of the disease than PB (ie, LN/BM) showed an increased expression of the 2 miRNAs in the tissues that was paralleled by a low expression of p27. Accordingly, in preliminary experiments, the forced expression of both anti-miRs in CLL cells from BM was able to up-regulate p27 protein (supplemental Figure 1).
The pattern of expression of p27 in the different anatomical compartments of CLL is compartment specific and appears to be related to the proliferative potential of the involved leukemic cells. CLL B lymphocytes from the PB are resting cells that mostly are in the G0 phase of the cell cycle and, accordingly, express high levels of p27 protein. In contrast, within the infiltrated lymphoid tissues, large proliferating leukemic B cells cluster to form the so-called PCs whose cells are Ki67+ and have a greatly reduced expression of p27.8 Our analysis in different compartments of the disease shows a clear increase of miR-221/222 (as well as of Ki67) along with the down-modulation of p27 in tissue leukemic B cells. These evidences strongly suggested that the regulation of the 2 microRNAs might be related to proliferative activity of the leukemic cells. This was indeed confirmed by reproducing the pattern of tissue expression with the in vitro stimulation of leukemic PB cells with CpG and to a lesser extent with CD40L, both known to be survival/mitogenic stimuli for CLL cells.4,6,43,44 Because microenvironmental cellular and soluble stimuli are present within the proliferation areas, it is intriguing to speculate that such stimuli may induce the transcription and may maintain high levels of miR-221/222 molecules in cells proliferating within LN/BM. Because cells exit the proliferation compartment, the stimuli sustaining miRNA transcription are expected to be lost, and the levels of miR-221/222 expression drastically fall, leading the cells into a quiescent state. Along the same line, p27 down-regulation in the tissues may represent the first step of activation of quiescent cells, promoting passage through the G1 phase and allowing entrance in the S phase. That notwithstanding, the reduction in p27 levels alone is not sufficient to allow the completion of cell cycle and to induce mitosis. Accordingly, in our in vitro model we were able to detect up-regulation of miRNAs and concomitant down-regulation of p27 but only a minimal increase in Ki67 expression. More signals are probably required to induce completion of cell cycle and active proliferation that are probably provided by yet unidentified soluble and cellular components of the PC microenvironment.
Some indications come from the experiments showing that the most effective stimulation that leads to up-regulation of miRNAs and down-regulation of p27 is the engagement of Toll-like receptor 9 (TLR9) by CpG-ODN. CLL cells express a vast repertoire of TLRs, including TLR9,44,45 and these specific TLRs are functional, suggesting a potential role of costimulatory signals in modulating CLL cell response. Because CLL leukemic cells appear to be able to recognize self-antigens and microbes through their surface immunoglobulins, it is plausible to suggest that CLL cells are stimulated by endogenous DNA, maybe in the context of PCs, by interacting with TLR9.
Significant up-regulation of miR-221/222 expression is also evident after CD40L engagement that in vivo is provided by CD40L-positive CD4+ T cells present in the tissue PCs.
In conclusion, we present evidence that miR-221/222 modulate the expression of p27 in CLL cells as in other types of cancers. This modulation might induce leukemic cells to enter the cell cycle after microenvironmental interactions and stimulation within the tissues and would maintain CLL lymphocytes in resting conditions in the bloodstream. The miR-221/222–p27 system may then represent a regulatory loop that is involved in the delicate balance of survival/proliferation in CLL.
The online version of this article contains a data supplement.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 USC section 1734.
Acknowledgments
We thank Rossano Cesari for helpful suggestions in the design of the research; we also thank Benedetta Apollonio for collaborative support in the laboratory and Kostas Stamatopoulos for providing CLL samples and clinical data.
This work was supported by the Associazione Italiana per la Ricerca sul Cancro (AIRC; P.G. and F.C.-C.), the CLL Global Research Foundation (F.C.-C.), Fondo per gli Investimenti della Ricerca di Base (F.C.-C.), and Progetto Integrato Oncologia (F.C.-C.). C.S. is recipient of a fellowship from Fondazione Ferrero.
Authorship
Contribution: M.F. designed and performed research, analyzed data, and wrote the paper; M.M. contributed to scientific design and revised the paper; C.S. and C.F. performed experiments; L.S. collected clinical information and analyzed data, C.R. produced vital reagents; G.F. revised the paper, and P.G. and F.C.-C. designed and supervised research and wrote the paper.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: Paolo Ghia, Università Vita-Salute San Raffaele, Via Olgettina 60, 20132, Milano, Italy; e-mail: ghia.paolo@hsr.it.




This feature is available to Subscribers Only
Sign In or Create an Account Close Modal