Abstract
Before the contemporary development of rationally designed antineoplastic therapies, cladribine was identified as a lymphocyte-specific agent. Its profound impact on the natural history of hairy cell leukemia, with responses approaching 100% and a median duration of response of nearly a decade after only a single 7-day course, is well known and revolutionized the treatment of hairy cell leukemia. However, cladribine's impressive activity in other lymphoproliferative disorders has been generally underappreciated. Multiple single-arm phase 2 trials have demonstrated cladribine's potency across the full spectrum of lymphoid malignancies. In a limited number of phase 3 trials and cross-study analyses, cladribine compared favorably with fludarabine, another purine nucleoside analog that is more commonly used in the treatment of indolent lymphoid malignancies. Cladribine has been noted to have particular activity among lymphoid disorders with few effective therapies, specifically, chronic lymphocytic leukemia, lymphoplasmacytic lymphoma, marginal zone lymphoma, and mantle cell lymphoma. Recently approved novel agents may act in synergy with cladribine for these conditions and should be incorporated into future clinical studies.
Introduction
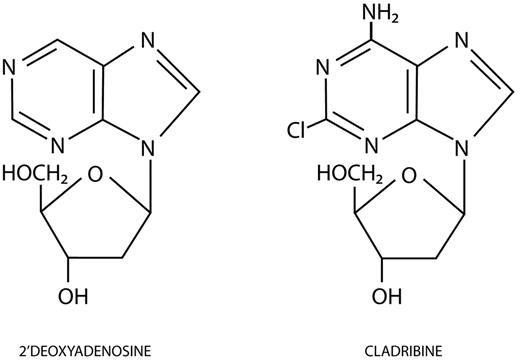
Forty years have passed since it was first observed that 2 girls born with severe combined immunodeficiency disease had the unusual findings of both profound lymphocytopenia and undetectable levels of adenosine deaminase (ADA) activity.1 In 1978, Cohen et al demonstrated that ADA deficiency leads to the toxic accumulation of deoxyadenosine triphosphate (dATP) and subsequent lymphocyte-specific cell death.2 Carson et al postulated that an ADA-resistant purine analog would be selectively toxic to lymphocytes3 ; they identified 2-chlorodeoxyadenosine, 2-CdA, or cladribine (Leustatin, Centocor Ortho Biotech) as the most potent ADA-resistant purine nucleoside analog from a field of candidate congeners.4 First synthesized in 1972 by Christensen et al, cladribine closely resembles deoxyadenosine, except for a chlorine atom substitution at the 2-position of the purine ring (Figure 1).5
Molecular structures of 2′-deoxyadenosine and cladribine. Cladribine has a chlorine atom substituted at the 2-position of the purine ring. (Illustration by Katya Kadyshevskaya.)
Molecular structures of 2′-deoxyadenosine and cladribine. Cladribine has a chlorine atom substituted at the 2-position of the purine ring. (Illustration by Katya Kadyshevskaya.)
Cladribine's remarkable activity in hairy cell leukemia (HCL), producing an overall response rate (ORR) of 98% with 91% complete responses (CRs), and extremely prolonged remissions after only a single 7-day course with a very favorable toxicity profile, is widely known.6 Less well promulgated is cladribine's role in the treatment of non-HCL indolent lymphoproliferative disorders. This review will focus on the considerable body of literature evaluating cladribine in these indolent lymphoid malignancies and propose a strategy for its further development in combination with novel agents.
Mechanism of action
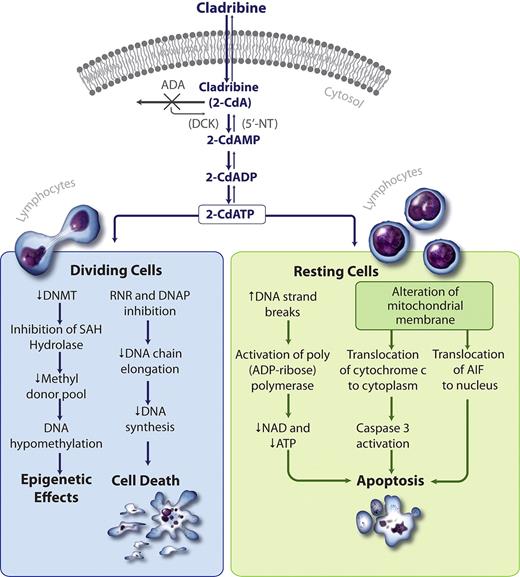
Like deoxyadenosine, cladribine enters lymphocytes through an efficient transport mechanism (Figure 2).7-9 Once in the cell, deoxyadenosine has 2 potential fates. Deoxyadenosine is subject to either irreversible deamination by ADA, leading ultimately to the uric acid excretion pathway, or serial phosphorylation by deoxycytidine kinase (DCK) to dATP. Cladribine, on the other hand, is resistant to deamination by ADA because of its chlorinated purine ring structure and instead becomes phosphorylated by DCK to its lymphocytotoxic form, 2-chlorodeoxyadenosine triphosphate.5,10 The intracellular concentrations of these phosphorylated purines are regulated by the competing kinetics of DCK, a phosphorylating enzyme, and 5′-nucleotidase (5′-NT), a dephosphorylating enzyme.11 Lymphocytes are unique for constitutively expressing high levels of DCK, such that the DCK to 5′-NT ratio favors phosphorylation, making them particularly susceptible to the actions of cladribine.12 Cladribine resistance has been correlated with DCK levels down-regulated to 5% or less, and 5′-NT levels 200% of normal, altering the DCK/5′-NT ratio, which then favors drug dephosphorylation.13 Decreased nucleoside transport, altered ribonucleotide reductase (RNR) regulation, and defective apoptotic pathways have also been implicated in cladribine resistance.14 Although non-HCL lymphocytes can have an elevated DCK/5′-NT ratio and are very sensitive to cladribine, it is unclear why cladribine's effect is most durable in HCL.11
Cladribine mechanism of action. 2-CdA enters the cell through an efficient transport system. Cladribine is resistant to deamination by ADA. The high DCK to 5′-NT ratio favors the formation of 2-chlorodeoxyadenosine monophosphate (2-CdAMP), 2-chlorodeoxyadenosine diphosphate (2-CdADP), and 2-chlorodeoxyadenosine triphosphate (2-CdATP). In dividing cells, the accumulation of 2-CdATP inhibits RNR and the DNA polymerases (DNAP), abrogating DNA synthesis and causing cell death. In resting cells, 2 unique pathways result in apoptosis. First, cladribine increases DNA strand breaks, activating poly(ADP-ribose)polymerase, PARP, which depletes NAD and ATP, resulting in apoptosis. Second, cladribine alters the mitochondrial membrane resulting in cytoplasmic translocation of cytochrome c and nuclear translocation of apoptosis-inducing factor (AIF), which leads to caspase-dependent and -independent apoptosis, respectively. Hypomethylation may occur through cladribine's inhibition of S-adenosylhomocysteine hydrolase (SAH), thereby diminishing the methyl donor pool.9 (Illustration by Katya Kadyshevskaya.)
Cladribine mechanism of action. 2-CdA enters the cell through an efficient transport system. Cladribine is resistant to deamination by ADA. The high DCK to 5′-NT ratio favors the formation of 2-chlorodeoxyadenosine monophosphate (2-CdAMP), 2-chlorodeoxyadenosine diphosphate (2-CdADP), and 2-chlorodeoxyadenosine triphosphate (2-CdATP). In dividing cells, the accumulation of 2-CdATP inhibits RNR and the DNA polymerases (DNAP), abrogating DNA synthesis and causing cell death. In resting cells, 2 unique pathways result in apoptosis. First, cladribine increases DNA strand breaks, activating poly(ADP-ribose)polymerase, PARP, which depletes NAD and ATP, resulting in apoptosis. Second, cladribine alters the mitochondrial membrane resulting in cytoplasmic translocation of cytochrome c and nuclear translocation of apoptosis-inducing factor (AIF), which leads to caspase-dependent and -independent apoptosis, respectively. Hypomethylation may occur through cladribine's inhibition of S-adenosylhomocysteine hydrolase (SAH), thereby diminishing the methyl donor pool.9 (Illustration by Katya Kadyshevskaya.)
Cladribine is cytotoxic to both dividing and nondividing lymphocytes from both healthy donors and patients with lymphoproliferative disorders.15 In resting cells, cladribine causes single-strand DNA breaks, inducing the DNA repair enzyme, poly(ADP-ribose) polymerase (PARP). PARP expression exhausts the intracellular pools of nicotinamide adenine dinucleotide (NAD) and adenosine triphosphate (ATP), precipitating apoptotic cell death.16,17 Recently, cladribine has also been shown to induce apoptosis by altering the mitochondrial transmembrane potential, enabling the translocation of cytochrome c and apoptosis-inducing factor into the cytosol, causing apoptosis in both a caspase-dependent and -independent process.18 In dividing cells, cladribine induces cytotoxicity by impairing DNA synthesis via 2 key mechanisms: (1) it potently inhibits RNR, and (2) it competes with dATP for incorporation into DNA by DNA polymerases α and β.19-21 Preclinical and clinical data suggest that cladribine may also have hypomethylating activity via its indirect inhibitory effects on DNA methyltransferase and depletion of the methyl donor pool.9
Pharmacokinetics
With plasma protein binding of only 20%, cladribine has a wide volume of distribution, specifically, 9 L/kg. Cladribine is excreted unchanged through the kidneys. Its elimination kinetics best fits a 2-compartment model with α and β half-lives of 35 minutes and 6.7 hours, respectively. Administering 0.14 mg/kg of cladribine as a 24-hour continuous intravenous infusion produces a mean steady-state concentration of 22.5nM. Given as a 2-hour bolus at 0.14 mg/kg, the mean peak plasma concentration reaches 198nM and falls to 22.5nM within 6 hours after completing the bolus. Two-hour bolus and infusional cladribine have comparable areas under the curve of 588 and 552nM/hour, respectively.22,23 The intracellular concentration of cladribine is known to be higher than the plasma concentration.15
Alternative routes of administration have been explored. Subcutaneous dosing (0.14 mg/kg) demonstrates equal bioavailability, volumes of distribution, and excretion profiles compared with an equivalent dose administered as a 2-hour intravenous bolus. Interestingly, rapid vascular uptake after subcutaneous dosing yielded a peak serum concentration greater than the 2-hour intravenous bolus, whose duration of infusion exceeded cladribine's brief biologic half-life, blunting its peak serum concentration. An oral dose of 0.28 mg/kg, double the typical intravenous dose, achieves similar area under the curve values as intravenous and subcutaneous dosing and has a bioavailability of 55%.24 Cerebral spinal fluid concentrations appear nonlinear relative to serum drug concentrations with markedly increased levels when cladribine is dosed more than 0.1 mg/kg per day. At the 0.15 mg/kg per day continuous infusion regimen, the mean cerebral spinal fluid concentration of cladribine reaches 20nM, twice the in vitro 50% inhibition for lymphoblast cell lines.25
Preclinical and phase 1 results
Cladribine selectively inhibits the growth of malignant human T-, B-, and null lymphoblastoid cell lines at nanomolar concentrations and is also lethal to resting lymphocytes. T-cell lines are as much as one log more sensitive to cladribine than B-cell lines.10 At equivalent concentrations, cladribine has almost no effect on solid tumor cell lines. The L1210 murine leukemia model demonstrates improved survival in cladribine-treated mice compared with mice receiving placebo or even fludarabine, a related purine nucleoside analog.4
The first phase 1 trial of cladribine began in 1981 at Scripps Clinic under the direction of Dr Ernest Beutler. Nine patients with refractory advanced hematologic malignancies received cladribine by continuous intravenous infusion at doses of 0.1 to 1 mg/kg per day for 5 to 14 days with dose escalations of 0.1 mg/kg per day for those without an initial therapeutic or toxic response. Cladribine lowered the blast count in all of the leukemic patients by a minimum of 50%. Dose-limiting toxicities included marked leukopenia and thrombocytopenia at doses more than 0.15 mg/kg per day. This study established the maximum tolerated dose of infusional cladribine to be 0.1 mg/kg per day for 7 days.15
Indolent NHL
Indolent non-Hodgkin lymphoma (NHL) constitutes a wide spectrum of B-cell histologies. Follicular lymphoma is the most common subtype in the United States and is frequently treated with rituximab in combination with cyclophosphamide, vincristine, and prednisone. Marginal zone and lymphoplasmacytic lymphomas, although less common, generally have excellent initial responses to fludarabine. Mantle cell lymphoma (MCL) has fewer, and generally unsatisfactory, available treatments. Standard curative therapies do not exist for any of the indolent lymphomas, and additional treatment options are needed. A large number of clinical studies indicate that cladribine has activity across the full spectrum of indolent lymphoma subtypes and can be a useful addition to the therapeutic armamentarium.
Single-agent therapy in previously treated indolent NHL
Eleven phase 2 studies have evaluated single-agent cladribine in the setting of relapsed or refractory indolent lymphomas (Table 1).26-36 Cladribine demonstrated impressive activity with overall and CR rates of up to 72% and 38%, respectively. Follicular and nonfollicular histologies appeared to be equally responsive. In the first of these studies, 40 relapsed and refractory low-grade lymphoma patients with a median of 3 prior therapies were treated with cladribine at 0.1 mg/kg per day by continuous infusion for 7 consecutive days. Two-thirds of these patients had follicular histology. The ORR was 43% (17 patients) with a CR rate of 20% (8 patients) and a 5-month median duration of response.26 A 2-hour bolus regimen produced similar results in a comparable patient population.27 These results were later reproduced in a trial where 80% of patients had a nonfollicular histology, mainly lymphoplasmacytic lymphoma (37%) or B-chronic lymphocytic leukemia (CLL)/small lymphocytic lymphoma (29%).28 Of 94 patients, 48 (51%) achieved a response and 12 (13%) had a CR. The median duration of response was 12 months for a CR and 6 months for a partial response. These nonrandomized trials clearly documented that cladribine can be highly active among pretreated patients and should be considered in this clinical setting. However, these patients were not exposed to the current therapeutic landscape, including rituximab, autologous stem cell transplantation, and emerging novel agents, such as bendamustine and bortezomib. Only future randomized studies incorporating cladribine will be able to determine an optimal approach.
Phase 2 studies of single-agent cladribine in previously treated indolent NHL
| Study . | Cladribine doses . | Histology (%) . | No. of prior therapies . | Response (%) . | Median duration, mo . | ||
|---|---|---|---|---|---|---|---|
| Follicular . | Other . | OR . | CR . | ||||
| Kay et al (1992)26 | A | 65 | 35 | 3 | 43 | 20 | 5 |
| Hickish et al (1993)29 | A | 82 | 18 | 4 | 71 | 29 | 6 |
| Hoffman et al (1994)30 | A | 25 | 75 | 2 | 45 | 15 | 9 |
| Morton et al (1996)27 | B | 67 | 33 | 2 | 63 | 11 | > 13 |
| Liliemark et al (1997)31 | C | 47 | 53 | 2 | 42 | 14 | 9 |
| Robak et al (1997)28 | C | 18 | 82 | 4 | 51 | 13 | > 6 |
| Kong et al (1998)32 | A | 55 | 45 | 3 | 45 | 9 | > 14 |
| Tulpule et al (1998)33 | B | 54 | 46 | 2 | 31 | 12 | > 12 |
| Tondini et al (2000)34 | B | 57 | 43 | 2 | 72 | 38 | Crossover |
| Ogura et al (2004)35 | D | 80 | 20 | 2 | 58 | 14 | 8 |
| Tobinai et al (2009)36 | C | 72 | 28 | 2 | 50 | 11 | > 12 |
| Study . | Cladribine doses . | Histology (%) . | No. of prior therapies . | Response (%) . | Median duration, mo . | ||
|---|---|---|---|---|---|---|---|
| Follicular . | Other . | OR . | CR . | ||||
| Kay et al (1992)26 | A | 65 | 35 | 3 | 43 | 20 | 5 |
| Hickish et al (1993)29 | A | 82 | 18 | 4 | 71 | 29 | 6 |
| Hoffman et al (1994)30 | A | 25 | 75 | 2 | 45 | 15 | 9 |
| Morton et al (1996)27 | B | 67 | 33 | 2 | 63 | 11 | > 13 |
| Liliemark et al (1997)31 | C | 47 | 53 | 2 | 42 | 14 | 9 |
| Robak et al (1997)28 | C | 18 | 82 | 4 | 51 | 13 | > 6 |
| Kong et al (1998)32 | A | 55 | 45 | 3 | 45 | 9 | > 14 |
| Tulpule et al (1998)33 | B | 54 | 46 | 2 | 31 | 12 | > 12 |
| Tondini et al (2000)34 | B | 57 | 43 | 2 | 72 | 38 | Crossover |
| Ogura et al (2004)35 | D | 80 | 20 | 2 | 58 | 14 | 8 |
| Tobinai et al (2009)36 | C | 72 | 28 | 2 | 50 | 11 | > 12 |
A indicates 0.1 mg/kg per day as continuous intravenous infusion for 7 days; B, 0.14 mg/kg per day as 2-hour intravenous infusion for 5 consecutive days; C, 0.12 mg/kg per day as 2-hour intravenous infusion for 5 consecutive days; and D, 0.09 mg/kg per day as continuous intravenous infusion for 7 days.
Combination therapy in previously treated indolent NHL
Attenuated doses of cladribine have been safely combined with other chemotherapeutic agents in patients with previously treated indolent lymphomas achieving promising results. Studies evaluating cladribine in combination with cyclophosphamide, mitoxantrone, or rituximab reported superior response rates compared with single-agent cladribine (Table 2).37-45 However, as is often the case in studies with indolent lymphomas, there were no statistical differences in overall survival (OS). Toxicities were typically hematologic and infectious, and were generally not severe. A single study cautioned against the routine use of infusional cladribine with cyclophosphamide as dose-limiting toxicities occurred in more than half the patients, with one-third of patients experiencing autoimmune phenomena, including autoimmune hemolytic anemia, immune thrombocytopenic purpura, and pure red cell aplasia (PRCA). However, one of the cohorts in the study represented a dose-finding arm, possibly explaining some of the observed toxicities.37
Phase 2 studies of cladribine combinations in indolent NHL
| Study . | Cladribine and other agent doses . | Other agents . | Histology, % . | Prior treatment, % . | OR, % . | CR, % . | Median duration, mo . | |
|---|---|---|---|---|---|---|---|---|
| Follicular . | Non . | |||||||
| Saven et al (1996)38 | A | Mitoxantrone | 57 | 43 | 100 | 70 | 22 | 5 (PR), 15 (CR) |
| Laurencet et al (1999)39 | B | Cyclophosphamide, prednisone | 0 | 100 | 42 | 88 | 22 | 12 (PR), 15 (CR) |
| Robak et al (1999)40 | C | Mitoxantrone, dexamethasone | 0 | 100 | 100 | 29 | 7 | > 6 |
| Rummel et al (2002)41 | D | Mitoxantrone | 51 | 49† | 32 | 90 | 44 | 24 |
| Van Den Neste et al (2004)37 | E | Cyclophosphamide | 26 | 74 | 100 | 51 | 14 | 11 |
| Robak et al (2004)42 | F | Rituximab | 12 | 88‡ | 100 | 69 | 15 | 7 |
| Robak et al (2006)43 | G | Rituximab, cyclophosphamide | 0 | 100§ | 100 | 71 | 11 | 11 |
| Laurencet et al (2007)44 | H | Cyclophosphamide, prednisone | 0 | 100 | 61 | 75 | 30 | 9 |
| Inwards et al (2008)45 (Study N0198) | I | Rituximab | 0 | 100‖ | 100 | 66 | 52 | 12 |
| Study . | Cladribine and other agent doses . | Other agents . | Histology, % . | Prior treatment, % . | OR, % . | CR, % . | Median duration, mo . | |
|---|---|---|---|---|---|---|---|---|
| Follicular . | Non . | |||||||
| Saven et al (1996)38 | A | Mitoxantrone | 57 | 43 | 100 | 70 | 22 | 5 (PR), 15 (CR) |
| Laurencet et al (1999)39 | B | Cyclophosphamide, prednisone | 0 | 100 | 42 | 88 | 22 | 12 (PR), 15 (CR) |
| Robak et al (1999)40 | C | Mitoxantrone, dexamethasone | 0 | 100 | 100 | 29 | 7 | > 6 |
| Rummel et al (2002)41 | D | Mitoxantrone | 51 | 49† | 32 | 90 | 44 | 24 |
| Van Den Neste et al (2004)37 | E | Cyclophosphamide | 26 | 74 | 100 | 51 | 14 | 11 |
| Robak et al (2004)42 | F | Rituximab | 12 | 88‡ | 100 | 69 | 15 | 7 |
| Robak et al (2006)43 | G | Rituximab, cyclophosphamide | 0 | 100§ | 100 | 71 | 11 | 11 |
| Laurencet et al (2007)44 | H | Cyclophosphamide, prednisone | 0 | 100 | 61 | 75 | 30 | 9 |
| Inwards et al (2008)45 (Study N0198) | I | Rituximab | 0 | 100‖ | 100 | 66 | 52 | 12 |
A indicates 0.1 mg/kg per day as continuous intravenous infusion for 7 days + mitoxantrone 5 mg/m2 intravenous day 1; B, 0.1 mg/kg per day subcutaneous bolus for 3 days; prednisone 40 mg/m2 PO for 5 days; cyclophosphamide 500 mg/m2 intravenous day 1; C, 0.12 mg/kg per day as 2-hour intravenous infusion for 5 consecutive days + mitoxantrone 10 mg/m2 + dexamethasone 20 mg/day intravenous/PO for 5 days; D, 5 mg/m2 2-hour intravenous infusion for 3 consecutive days + mitoxantrone 8 mg/m2 days 1 and 2, or 12 mg/m2 day 1 for first relapse; E, 5.6 mg/m2 per day for 3 days + cyclophosphamide 200 mg/m2 per day for 3 days; F, 0.12 mg/kg per day as 2-hour intravenous infusion for 5 consecutive days, days 2 to 6 + rituximab 375 mg/m2 as 6-hour intravenous infusion day 1; G, 0.12 mg/kg per day as 2-hour intravenous infusion for 3 consecutive days, days 2 to 4 + rituximab 375 mg/m2 as 6-hour intravenous infusion day 1 + cyclophosphamide 250 mg/m2 days 2 to 4; H, 0.1 mg/kg per day subcutaneous bolus injection for 5 days + prednisone 40 mg/m2 PO for 5 days + cyclophosphamide 500 mg/m2 intravenous day 1; and I, 5 mg/m2 2-hour intravenous infusion 5 consecutive days + rituximab 375 mg/m2 6-hour intravenous infusion day 1; and PR, partial response.
63% MCL.
65% CLL/small lymphocytic lymphoma.
17% MCL.
100% MCL.
Single-agent therapy in previously untreated indolent NHL
After demonstrating major activity in patients with heavily pretreated indolent lymphomas, single-agent cladribine was subsequently introduced in the front-line setting. As was the case for single-agent cladribine in previously treated patients, most patients had a follicular histology, with follicular and nonfollicular histologies generally demonstrating similar outcomes. Patients had OR and CR rates of up to 98% and 38%, respectively, comparing favorably to single-agent cladribine in the salvage setting. The median duration of response varied widely from 7 to 23 months (Table 3).46-52
Phase 2 studies of single-agent cladribine in previously untreated indolent NHL
| Study . | Cladribine doses . | Histology, % . | Response, % . | Median duration, mo . | ||
|---|---|---|---|---|---|---|
| Follicular . | Other . | OR . | CR . | |||
| Saven et al (1995)48 | A | 36 | 64 | 88 | 35 | 10 |
| Betticher et al (1996)49 | B | 100 | 0 | 85 | 24 | > 14 |
| Canfield et al (1997)50 | A | 92 | 8 | 64 | 8 | > 14 |
| Fridrik et al (1998)51 | C | 57 | 43 | 88 | 27 | 21 |
| Liliemark et al (1998)52 | C | 50 | 50 | 64 | 25 | 7 |
| Rummel et al (1999)47 | D | 39 | 61 | 76 | 38 | 23 |
| Blum et al (2006)46 | E | 74 | 26 | 98 | 32 | 23 |
| Study . | Cladribine doses . | Histology, % . | Response, % . | Median duration, mo . | ||
|---|---|---|---|---|---|---|
| Follicular . | Other . | OR . | CR . | |||
| Saven et al (1995)48 | A | 36 | 64 | 88 | 35 | 10 |
| Betticher et al (1996)49 | B | 100 | 0 | 85 | 24 | > 14 |
| Canfield et al (1997)50 | A | 92 | 8 | 64 | 8 | > 14 |
| Fridrik et al (1998)51 | C | 57 | 43 | 88 | 27 | 21 |
| Liliemark et al (1998)52 | C | 50 | 50 | 64 | 25 | 7 |
| Rummel et al (1999)47 | D | 39 | 61 | 76 | 38 | 23 |
| Blum et al (2006)46 | E | 74 | 26 | 98 | 32 | 23 |
A indicates 0.1 mg/kg per day as continuous intravenous infusion for 7 days; B, 0.1 mg/kg per day as continuous intravenous or subcutaneous infusion for 7 days; C, 0.12 mg/kg per day as 2-hour intravenous infusion for 5 consecutive days; D, 5 mg/m2 2-hour intravenous infusion for 5 consecutive days; and E, 0.14 mg/kg per day as 2-hour intravenous infusion for 5 consecutive days.
The Cancer and Leukemia Group B conducted the most recent single-arm phase 2 trial, enrolling 44 treatment naive indolent lymphoma patients, the majority (73%) of whom had a follicular subtype. Patients received bolus cladribine at 0.14 mg/kg per day over 2 hours for 5 consecutive days every 28 days for up to 6 cycles, achieving an ORR of 98% (43 patients) and 32% CRs (14 patients). The median response duration was 1.3 years for partial responses and 3.1 years for CRs, with a median OS of 7 years.46 Table 3 lists the other 6 reported cladribine studies in untreated indolent lymphoma patients, demonstrating its consistent activity across trials. Toxicity in the Cancer and Leukemia Group B study was considerable with 68% experiencing a grade 3 or 4 event, largely myelosuppression, and 8 patients had grade 3 infections. Four histologic transformations to high-grade lymphoma were documented, between 2 and 32 months after the last cycle of cladribine.46
Nonfollicular histologies appear equally sensitive to cladribine (Table 4).45,53-60 The largest single-arm phase 2 cladribine study in this setting enrolled 66 patients (80% untreated) with a variety of histologies, including follicular (40%), lymphoplasmacytic (30%), and MCL (20%). Even though half of the MCL patients were pretreated, that group achieved a 58% response rate with 25% CRs.47,53 Similarly, a study of 26 treatment-naive gastric marginal zone lymphoma patients reported an ORR of 100% with 84% CRs. Of 7 patients with an extragastric presentation, 3 patients achieved a CR. Of note, a fatal myocardial infarction and stroke were reported within days and 3 months of the first and third course of therapy, respectively.54 A small series of splenic marginal zone patients in relapse after chlorambucil demonstrated an ORR of 86%, but with a short duration of remission and significant infectious complications.55
Phase 2 studies of single-agent cladribine in nonfollicular type NHL
| Study . | Cladribine doses . | Histology . | Previously treated . | Treatment-naive . | Response, % . | Median duration, mo . | |
|---|---|---|---|---|---|---|---|
| OR . | CR . | ||||||
| Dimopoulos et al (1993)57 | A | WM | 20 | 9 | 59 | 3 | > 9 |
| Dimopoulos et al (1994)56 | A | WM | 0 | 26 | 85 | 12 | > 13 |
| Fridrik et al (1997)58 | B | WM | 10 | 0 | 90 | 10 | > 13 |
| Liu et al (1998)59 | B | WM | 13 | 7 | 55 | 5 | > 18 |
| Hellman (1999)60 | C | WM | 13 | 9 | 41 | 36 | 12 (mean) |
| Lefrère et al (2000)55 | A | Splenic MZL | 6 | 1 | 86 | 29 | 4 |
| Jäger et al (2002)54 | B | MZL | 0 | 26 | 100 | 84 | > 32 |
| Rummel et al (1999)53 | D | MCL | 5 | 7 | 58 | 25 | 19 |
| Inwards et al (2008)45 | D | MCL | 24 | 26 | 46 PT; 81 TN | 21 PT; 42 TN | PT: 5; TN: 14 |
| Study . | Cladribine doses . | Histology . | Previously treated . | Treatment-naive . | Response, % . | Median duration, mo . | |
|---|---|---|---|---|---|---|---|
| OR . | CR . | ||||||
| Dimopoulos et al (1993)57 | A | WM | 20 | 9 | 59 | 3 | > 9 |
| Dimopoulos et al (1994)56 | A | WM | 0 | 26 | 85 | 12 | > 13 |
| Fridrik et al (1997)58 | B | WM | 10 | 0 | 90 | 10 | > 13 |
| Liu et al (1998)59 | B | WM | 13 | 7 | 55 | 5 | > 18 |
| Hellman (1999)60 | C | WM | 13 | 9 | 41 | 36 | 12 (mean) |
| Lefrère et al (2000)55 | A | Splenic MZL | 6 | 1 | 86 | 29 | 4 |
| Jäger et al (2002)54 | B | MZL | 0 | 26 | 100 | 84 | > 32 |
| Rummel et al (1999)53 | D | MCL | 5 | 7 | 58 | 25 | 19 |
| Inwards et al (2008)45 | D | MCL | 24 | 26 | 46 PT; 81 TN | 21 PT; 42 TN | PT: 5; TN: 14 |
MZL indicates marginal zone lymphoma; PT, previously treated; and TN, treatment-naive.
A indicates 0.1 mg/kg per day as continuous intravenous infusion for 7 days; B, 0.12 mg/kg per day as 2-hour intravenous infusion for 5 consecutive days; C, 0.14 mg/kg per day as 2-hour intravenous infusion for 5 consecutive days; and D, 5 mg/m2 2-hour intravenous infusion for 5 consecutive days.
Despite cladribine's demonstrated activity in untreated indolent NHL, there has only been one published phase 3 study to date. The Polish Lymphoma Research Group randomly assigned 197 untreated indolent lymphoma patients to one of 3 monthly treatment arms: single-agent cladribine, cladribine plus cyclophosphamide, or cyclophosphamide, vincristine, and prednisone (CVP; Table 5).61 Histologic subtypes by WHO classification included CLL, 35%; follicular, 28%; marginal zone, 25%; and lymphoplasmacytic, 7%. The primary endpoint was progression-free survival (PFS). At 10 months, accrual to the CVP arm was prematurely discontinued because of statistically superior PFS (P < .001) and ORR (P < .001) in the cladribine-containing arms. At 3-year follow-up, PFS for combination cladribine and cyclophosphamide, single-agent cladribine, and CVP was 61%, 48%, and 22%, respectively. Statistical differences were observed between the CVP and the cladribine-containing arms, but not between the cladribine-containing arms. However, the combination cladribine and cyclophosphamide arm did report a statistically greater number of CRs than single-agent cladribine. No statistical differences were found in OS. In a multivariate analysis with International Prognostic Indices, cladribine therapy remained an independent prognostic factor for improved PFS. Endpoints for all histologic subgroups were reported as being similar. Infections occurred in 7% of patients in each arm, but grade 3 hematologic toxicity was most common in the combination cladribine and cyclophosphamide arm.61
Phase 3 study of cladribine with and without cyclophosphamide versus CVP
| Study/arm, doses . | Histology, % . | Response, % . | 3-y PFS, % . | ||
|---|---|---|---|---|---|
| Follicular . | Nonfollicular . | OR . | CR . | ||
| Kalinka-Warzocha et al (2008)61 | 28 | 72 | |||
| CdA | 24 | 76 | 64 (P = .002) | 22 (P = .003) | 48 |
| CCdA | 37 | 63 | 76 (P < .001) | 46 (P < .001) | 61 (P < .001) |
| CVP | 22 | 78 | 44 | 6.7 | 22 |
| Study/arm, doses . | Histology, % . | Response, % . | 3-y PFS, % . | ||
|---|---|---|---|---|---|
| Follicular . | Nonfollicular . | OR . | CR . | ||
| Kalinka-Warzocha et al (2008)61 | 28 | 72 | |||
| CdA | 24 | 76 | 64 (P = .002) | 22 (P = .003) | 48 |
| CCdA | 37 | 63 | 76 (P < .001) | 46 (P < .001) | 61 (P < .001) |
| CVP | 22 | 78 | 44 | 6.7 | 22 |
CdA indicates cladribine at 0.12 mg/kg per day as 2-hour intravenous infusion for 5 consecutive days; CCdA, same cladribine plus cyclophosphamide 800 mg/m2 bolus intravenous every 28 days; and CVP, cyclophosphamide 800 mg/m2 bolus intravenous day 1, vincristine 1.4 mg/m2 bolus intravenous day 1, prednisone 45 mg/m2 PO days 1 to 4 every 21 days.
Cladribine has consistently demonstrated impressive activity across a wide spectrum of indolent lymphomas in untreated patients. Except for its myelosuppressive qualities, it is well tolerated and can be considered as front-line single-agent therapy in appropriate indolent lymphoma patients with a good performance status. Indeed, cladribine was superior to CVP chemotherapy, another standard treatment, for response and survival endpoints in a study that mirrored a similar trial comparing single-agent fludarabine to CVP.61,62 However, with the superiority of rituximab-CVP compared with CVP alone and the promising results of rituximab-bendamustine, optimal front-line treatment of indolent lymphoma is in constant flux.63
Waldenström macroglobulinemia
Waldenström macroglobulinemia (WM) is a malignant B-cell lymphoplasmacytic lymphoma characterized by pathologic monoclonal IgM immunoglobulin overproduction. Several studies have evaluated cladribine in previously treated and untreated patients. In one study, 26 untreated symptomatic patients were given cladribine 0.1 mg/kg per day by 7-day continuous infusion and obtained an ORR of 85% with a median response duration of more than 13 months.56 Other reports corroborated the sensitivity of WM to cladribine, with response rates ranging from 41% to 90% and a median duration of response between 9 and 18 months (Table 4). These results are comparable with front-line fludarabine studies in similar patient populations, producing response rates from 38% to 100%.64 In its most recent update, the International Workshop on Waldenström Macroglobulinemia recommended cladribine as an acceptable first-line treatment for WM, either as a single agent or in combination with rituximab or cyclophosphamide. However, its use should probably be avoided in younger patients as recent reports have noted a potentially increased risk of Richter transformation, myelodysplasia, and acute myeloid leukemia (AML) in WM patients receiving a purine nucleoside analog.65
MCL
MCL can exhibit an indolent course, but most patients eventually require therapy for symptomatic progression. Even patients receiving the most aggressive combination chemotherapy or transplantation programs will relapse within a few years. Several single-arm studies with mixed populations of previously treated and untreated patients suggest that cladribine has considerable activity in MCL (Table 4). Twelve patients with MCL (7 previously treated) were administered cladribine 5 mg/m2 per day as a 2-hour bolus for 5 consecutive days, yielding an ORR of 58% (7 patients) with CRs in 25% (3 patients), and a median time to progression of 19 months.53 The North Central Cancer Treatment Group administered the same cladribine schedule to patients with MCL and documented an ORR of 81% (21 patients) with 42% CRs (11 patients) among 26 treatment naive patients and an ORR of 46% (11 patients) with 21% CRs (5 patients) for 24 previously treated patients.45
Combination approaches in previously treated and untreated MCL patients have also documented significant clinical activity. Among 29 previously untreated MCL patients administered combination cladribine and rituximab, there was an ORR of 66% (19 patients) with 52% CRs (15 patients). After 21.5 months of follow-up, 80% of patients in the combined treatment arm who had achieved a CR were still in remission.45 In another trial, of 18 MCL patients treated with combination cladribine and mitoxantrone, all responded (8 patients with a CR) and had a 24-month median duration of response.41 Among a different group of 9 MCL patients given cladribine in combination with cyclophosphamide and rituximab, 6 patients responded with 2 obtaining a CR.43
These results demonstrate that cladribine is highly active both as a single agent and as a component of combination therapy for MCL patients. Single-agent use of cladribine may be appropriate in selected patient populations, but the incorporation of cladribine into future multiagent approaches will probably be most effective.
CLL/small lymphocytic lymphoma
CLL and small lymphocytic lymphoma can be considered overlapping pathologic and clinical entities that are distinguished from each other by the presence of lymphadenopathy and lymphocytosis.66 Fludarabine has become the cornerstone of first-line CLL therapy, either as a single-agent or in combination with cyclophosphamide and/or rituximab.67,68 Cladribine's role in treating CLL has been largely overlooked, despite being a related purine nucleoside analog to fludarabine, with impressive clinical results. In preclinical studies, CLL B-lymphocytes undergo apoptosis when incubated with cladribine via mitochondrial membrane disruption, releasing proapoptotic cytochrome c and apoptosis-inducing factor.69,70 Multiple single-arm and randomized clinical trials have confirmed cladribine's major activity in CLL (Table 6).42,71-79
Phase 2 CLL cladribine studies: previously treated patients
| Study . | No. of patients . | Median no. of prior therapies . | Cladribine doses and other agents . | Response, % . | Median duration, mo . | |
|---|---|---|---|---|---|---|
| OR . | CR . | |||||
| Saven et al (1991)71 | 90 | 2 | A, B | 44 | 4 | 4 |
| Tallman et al (1995)75 | 26 | 2 | A | 31 | 0 | 16 |
| Robak et al (1996)72 | 92 | 2 | C | 36 | 5 | NA |
| Juliusson et al (1996)76 | 52 | 2 | C | 58 | 31 | 20 |
| Rondelli et al (1997)79 | 19 | > 2 | D | 68 | 10 | CR, 9+ |
| Robak et al (2000)74 | 104 | 2 | E | 51 | 15 | 11 |
| Robak et al (2000)74 | 80 | 2 | C | 45 | 9 | 9 |
| Robak et al (2001)73 | 19 | 2 | F, G | 37 | 5 | NA |
| Karlsson et al (2002)78 | 38 | 2 | H | 34 | 5 | 14 |
| Montillo et al (2003)77 | 20 | 2 | I | 35 | 15 | 12 |
| Robak et al (2004)42 | 15 | 2 | J | 73 | 7 | 6 |
| Study . | No. of patients . | Median no. of prior therapies . | Cladribine doses and other agents . | Response, % . | Median duration, mo . | |
|---|---|---|---|---|---|---|
| OR . | CR . | |||||
| Saven et al (1991)71 | 90 | 2 | A, B | 44 | 4 | 4 |
| Tallman et al (1995)75 | 26 | 2 | A | 31 | 0 | 16 |
| Robak et al (1996)72 | 92 | 2 | C | 36 | 5 | NA |
| Juliusson et al (1996)76 | 52 | 2 | C | 58 | 31 | 20 |
| Rondelli et al (1997)79 | 19 | > 2 | D | 68 | 10 | CR, 9+ |
| Robak et al (2000)74 | 104 | 2 | E | 51 | 15 | 11 |
| Robak et al (2000)74 | 80 | 2 | C | 45 | 9 | 9 |
| Robak et al (2001)73 | 19 | 2 | F, G | 37 | 5 | NA |
| Karlsson et al (2002)78 | 38 | 2 | H | 34 | 5 | 14 |
| Montillo et al (2003)77 | 20 | 2 | I | 35 | 15 | 12 |
| Robak et al (2004)42 | 15 | 2 | J | 73 | 7 | 6 |
A indicates 0.1 mg/kg per day as continuous intravenous infusion for 7 days; B, 0.028 to 0.14 mg/kg per day as 2-hour intravenous infusion for 5 consecutive days; C, 0.12 mg/kg per day days 1 to 5 2-hour intravenous bolus; D, 6 mg/m2 per day as 2-hour intravenous infusion for 5 consecutive days; E, 0.12 mg/kg per day 2-hour intravenous infusion for 5 consecutive days + prednisone 30 mg/m2 PO for 5 days; F, 0.12 mg/kg per day as 2-hour intravenous infusion for 5 consecutive days + cyclophosphamide 650 mg/m2 intravenous day 1 + mitoxantrone 10 mg/m2 intravenous day 1; G, 0.12 mg/kg per day as 2-hour intravenous infusion for 3 consecutive days + cyclophosphamide 650 mg/m2 intravenous on day 1 + mitoxantrone 10 mg/m2 intravenous on day 1; H, 10 mg/m2 per day PO for 3 consecutive days every 21 days; I, 4 mg/m2 per day as 2-hour intravenous infusion for 3 consecutive days + cyclophosphamide 350 mg/m2 per day intravenous for 3 days; and J, 0.12 mg/kg per day as 2-hour intravenous infusion for 5 consecutive days, days 2 to 6 + rituximab 375 mg/m2 as 6-hour intravenous infusion day 1; and NA, not available.
Single-agent therapy in previously treated CLL
Piro et al first evaluated cladribine in previously treated CLL patients, documenting an ORR of 50% in 18 patients.80 Continued enrollment to this trial ultimately accrued 90 refractory CLL patients, administering cladribine as either a 0.1-mg/kg per day 7-day continuous infusion or as a 0.028- to 0.14-mg/kg per day 2-hour bolus for 5 consecutive days, producing an ORR of 44% (40 patients) with 4% CRs (4 patients). Of those patients not meeting response criteria, more than half achieved a 50% or greater sustained reduction in the absolute lymphocyte count.71 A trial of bolus cladribine dosing in 92 previously treated patients reported remarkably similar results (Table 6).72 Patients in these 2 studies each had a median of 2 prior therapies, suggesting that single-agent cladribine was an effective salvage therapy in CLL. However, because these patients only received alkylating agents or steroids, and not modern combination therapies that incorporated fludarabine and rituximab, the role of single-agent cladribine in the current therapeutic arsenal of CLL salvage therapy is less clear. Small studies that have evaluated the activity of cladribine in fludarabine-refractory CLL patients, or the reverse, suggest that cross-resistance exists (“Cladribine versus fludarabine”) and that salvage cladribine in purine analog-refractory patients may actually be deleterious. In contrast, among patients who relapsed, but were not refractory, after prior purine analog therapy (either cladribine or fludarabine), cladribine has activity and may be considered in this scenario.34,81,82
Combination therapy in previously treated CLL
In the salvage setting, combination cladribine therapies have not been clearly shown to offer clinically significant advantages over single-agent cladribine use (Table 6). A group of heavily pretreated CLL patients (median prior chemotherapy courses, 7.5; range, 5-14) were given cladribine at a dose of 0.12 mg/kg per day by 2-hour bolus for 5 days in combination with cyclophosphamide and mitoxantrone. Responses were not superior to the historical reports for single-agent cladribine (37% ORR, 19 patients), but toxicities were considerable, including severe neutropenia and infections/fever of unknown origin in up to 50% and 83% of patients, respectively.73 A randomized study of cladribine in combination with prednisone versus cladribine alone also did not find a significant difference in response rates, although the combination arm had improved median OS (14 months vs 10 months, P = .001). The authors hypothesized that the survival advantage had resulted from a salutary effect of steroids on bone marrow function and CLL-induced autoimmunity but ultimately concluded that combination cladribine and prednisone therapy was not warranted except for pretreated patients with severe anemia or thrombocytopenia.74
Cladribine has been administered together with rituximab in 15 relapsed/refractory CLL patients producing an impressive ORR of 73% (11 patients) with 7% CRs (1 patient), providing the rationale for additional cladribine-rituximab combination approaches.42 However, with the current standard practice of incorporating rituximab into up-front CLL therapy, subsequent rituximab-cladribine combinations may prove less valuable.
Single-agent therapy in previously untreated CLL
Early studies evaluating front-line cladribine in untreated CLL patients produced an ORR ranging from 72% to 85% with a CR range of 25% to 37%.74,83,84 These results seem to be similar, or even more impressive, than outcomes from studies using single-agent fludarabine and are comparable with results reported for combination fludarabine and cyclophosphamide regimens.67 In the only prospective phase 3 trial evaluating cladribine in the front-line setting, 229 patients were randomized to cladribine, fludarabine, or high-dose intermittent chlorambucil. Cladribine was administered at 5 mg/m2 per day as a 2-hour bolus for 5 consecutive days. No statistical differences in ORR or OS were detected among the treatment arms; however, cladribine produced a significantly improved median time to progression of 25 months versus 10 months for fludarabine, and 9 months for chlorambucil (P < .001)85 (Table 7).85-88 For patients who are not candidates to receive combination chemotherapy regimens, administering single-agent cladribine is therefore a reasonable option.
Phase 3 CLL cladribine studies: untreated patients
| Study/regimen . | No. of patients . | Cladribine dose . | Response, % . | Median duration, mo . | OS, mo . | |
|---|---|---|---|---|---|---|
| OR . | CR . | |||||
| Robak et al (2000)86 | ||||||
| 2-CdA + P | 126 | A | 87 | 47 | 24 (P = .01): 46% | 24 (P = .6): 78% |
| Chl + P | 103 | 12 mg/m2/d PO + 30 mg/m2/d PO for 7 days | 57 | 12 | 24 (33%) | 24: 82% |
| Robak et al (2006)87 | ||||||
| 2-CdA | 166 | B | 77 | 21 | 24 (P = .47) | 51 (P = .73) |
| 2-CdA + Cy | 162 | C | 83 | 29 | 22 (P = .49) | Not yet reached |
| 2-CdA + Cy + M | 151 | D | 80 | 36 | 24 | Not yet reached |
| Karlsson et al (2007)85 | ||||||
| 2-CdA | 72 | E | 75 | NA | 25 (P < .001) | 82 (NS) |
| F | 73 | 25 mg/m2/d IV for 5 days | 70 | NA | 10 | 68 |
| Chl | 76 | 10 mg/m2/d PO for 10 days | 62 | NA | 9 | 62 |
| Robak et al (2010)88 | ||||||
| 2-CdA + Cy | 212 | F | 88 | 47 | 28 (P = .51) | 48 (P = .16): 62% |
| F + Cy | 211 | 25 mg/m2/d IV + 250 mg/m2/d IV for 3 days | 82 | 46 | 27 | 48: 61% |
| Study/regimen . | No. of patients . | Cladribine dose . | Response, % . | Median duration, mo . | OS, mo . | |
|---|---|---|---|---|---|---|
| OR . | CR . | |||||
| Robak et al (2000)86 | ||||||
| 2-CdA + P | 126 | A | 87 | 47 | 24 (P = .01): 46% | 24 (P = .6): 78% |
| Chl + P | 103 | 12 mg/m2/d PO + 30 mg/m2/d PO for 7 days | 57 | 12 | 24 (33%) | 24: 82% |
| Robak et al (2006)87 | ||||||
| 2-CdA | 166 | B | 77 | 21 | 24 (P = .47) | 51 (P = .73) |
| 2-CdA + Cy | 162 | C | 83 | 29 | 22 (P = .49) | Not yet reached |
| 2-CdA + Cy + M | 151 | D | 80 | 36 | 24 | Not yet reached |
| Karlsson et al (2007)85 | ||||||
| 2-CdA | 72 | E | 75 | NA | 25 (P < .001) | 82 (NS) |
| F | 73 | 25 mg/m2/d IV for 5 days | 70 | NA | 10 | 68 |
| Chl | 76 | 10 mg/m2/d PO for 10 days | 62 | NA | 9 | 62 |
| Robak et al (2010)88 | ||||||
| 2-CdA + Cy | 212 | F | 88 | 47 | 28 (P = .51) | 48 (P = .16): 62% |
| F + Cy | 211 | 25 mg/m2/d IV + 250 mg/m2/d IV for 3 days | 82 | 46 | 27 | 48: 61% |
2-CdA indicates cladribine; P, prednisone; Chl, chlorambucil; Cy, cyclophosphamide; M, mitoxantrone; F, fludarabine; IV, intravenously; PO, by mouth; and NS, not significant.
A indicates 0.12 mg/kg per day 2-hour intravenous infusion for 5 consecutive days + prednisone 30 mg/m2 PO for 5 days; B, 0.12 mg/kg per day as 2-hour intravenous infusion for 5 consecutive days; C, 0.12 mg/kg per day as 2-hour intravenous infusion for 3 consecutive days + cyclophosphamide 650 mg/m2 intravenous on day 1; D, 0.12 mg/kg per day as 2-hour intravenous infusion for 3 consecutive days + cyclophosphamide 650 mg/m2 intravenous on day 1 + mitoxantrone 10 mg/m2 intravenous on day 1; E, 5 mg/m2 2-hour intravenous infusion for 5 consecutive days; and F, 0.12 mg/kg per day as 2-hour intravenous infusion for 3 consecutive days + cyclophosphamide 250 mg/m2 per day intravenous for 3 consecutive days.
Combination therapy in previously untreated CLL
In a randomized trial, combination cladribine and prednisone produced improved response rates and PFS, but no difference in OS, compared with combination chlorambucil and prednisone or single-agent cladribine.86 A 3-drug combination of cladribine, mitoxantrone, and cyclophosphamide was compared with single-agent cladribine, with or without cyclophosphamide, producing increased CRs, but no differences in ORR, PFS, or OS. The 3-drug combination reported increased myelotoxicity and 40% of patients developed severe infections (Table 7).87 The nominal improvements in clinical endpoints derived from the cladribine, cyclophosphamide, and mitoxantrone combination treatment probably do not justify further investigational consideration of this regimen. Administering combination cladribine in sequence, instead of concurrently, offered a highly effective and potentially less toxic approach.89
Cladribine versus fludarabine
Cladribine's ability to induce apoptosis by altering the mitochondrial membrane potential is not shared by fludarabine and may serve as a nonoverlapping mechanism of action, potentially allowing cladribine's use to be exploited in certain clinical scenarios.70 Four fludarabine-refractory CLL patients were reported to have responded to subsequent cladribine therapy.90 However, far more clinical data exist to support cross-resistance between these 2 purine nucleoside analogs. Only 2 of 10 cladribine-refractory patients responded to second-line fludarabine.74 In another report, only 7% (2 patients) of fludarabine-refractory patients had a response to cladribine.91 In the sole randomized study, 60 patients with relapsed or refractory indolent lymphoma were assigned to either single-agent cladribine or fludarabine and then later crossed over to the alternative purine analog at progression. At crossover, 8 of 9 patients with an initial response responded again, but refractory patients did not, also suggesting cross-resistance.34
Despite the current trend favoring fludarabine-based therapies for CLL, it is not clear that front-line fludarabine is necessarily superior to cladribine. A randomized phase 3 study comparing single-agent fludarabine with cladribine in front-line CLL therapy noted a markedly superior median time to progression of 25 months for cladribine versus 10 months for fludarabine (P < .001).85 Final analysis of the PALG-CLL3 study that administered cyclophosphamide in combination with either cladribine or fludarabine to previously untreated CLL patients reported equivalent CR, ORR, and PFS with comparable toxicities.88 These 2 randomized reports indicate that cladribine is at least equivalent to fludarabine in the front-line therapy of CLL. Cross-study comparisons of either the fludarabine-cyclophosphamide arm or cladribine-cyclophosphamide arm from other front-line phase 3 CLL trials reinforce this conclusion (Table 8).67,87,88,92,93
Cladribine versus fludarabine in CLL
| Study . | Regimen . | No. of patients . | Response, 5 . | Median duration, mo . | |
|---|---|---|---|---|---|
| ORR . | CR . | ||||
| Robak et al (2006)87 | 2-CdA + Cy | 162 | 77 | 21 | 24 |
| Robak et al (2010)88 | 2-CdA + Cy | 211 | 88 | 47 | 28 |
| Robak et al (2010)88 | F + Cy | 212 | 82 | 46 | 27 |
| Eichhorst et al (2006)93 | F + Cy | 180 | 94 | 24 | 48 |
| Catovsky et al (2007)92 | F + Cy | 196 | 94 | 38 | 43 |
| Flinn et al (2007)67 | F + Cy | 141 | 74 | 23 | 32 |
| Study . | Regimen . | No. of patients . | Response, 5 . | Median duration, mo . | |
|---|---|---|---|---|---|
| ORR . | CR . | ||||
| Robak et al (2006)87 | 2-CdA + Cy | 162 | 77 | 21 | 24 |
| Robak et al (2010)88 | 2-CdA + Cy | 211 | 88 | 47 | 28 |
| Robak et al (2010)88 | F + Cy | 212 | 82 | 46 | 27 |
| Eichhorst et al (2006)93 | F + Cy | 180 | 94 | 24 | 48 |
| Catovsky et al (2007)92 | F + Cy | 196 | 94 | 38 | 43 |
| Flinn et al (2007)67 | F + Cy | 141 | 74 | 23 | 32 |
2-CdA indicates cladribine; Cy, cyclophosphamide; and F, fludarabine.
Fludarabine-based therapies in CLL have been unable to overcome the deleterious influence of the 17p13.1 deletion.92 Cladribine's ability to produce caspase-dependent apoptosis by altering the mitochondrial membrane potential, a p53-independent pathway, could potentially permit it to overcome the negative effects of the 17p13.1 deletion.88 A retrospective review of 20 patients with 17p13.1 deletions treated with front-line cladribine-cyclophosphamide noted an ORR of 80% (16 patients) with 50% CRs (10 patients) and a median PFS of 23 months.94 Final results of the PALG-CLL3 study reported a nonsignificant improvement in the CR rate for the cladribine arm of the 17p-deleted subgroup (40% vs 15%, P = .112). However, a random nonbalanced distribution of the 17p-deleted patients placed more advanced-stage patients in the fludarabine-based cohort, undermining this promising trend.88 Opportunity for improved therapies in this poor-prognosis setting remains an open area for future trials.
AML
Although the focus of this review is on the indolent lymphoproliferative disorders, it is important to mention recent cladribine data documenting its activity in AML, a disease in urgent need of new therapeutic options. Seventeen children with refractory AML were treated with cladribine at 8.9 mg/m2 per day for 5 days via continuous infusion, and 8 obtained a CR.95 When AML patients older than 40 years were randomized between induction daunorubicin, cytarabine, and cladribine (DAC) versus daunorubicin and cytarabine (DA) alone, patients treated with DAC had a 17% higher CR rate and statistically improved OS (26% DAC vs 15% with DA alone, P = .03).96 An abstract from the American Society of Hematology 2009 annual conference compared DAC with DA plus fludarabine (DAF) and standard DA in 673 adult untreated AML patients. The CR rate was significantly superior in the DAC arm (68% DAC vs 59% DAF and 56% DA, P = .013 and P = .08, respectively) as was the 3-year OS (46% DAC vs 30% DAF and 31% DA, P = .02).97 These encouraging results provide a foundation to design future studies using cladribine as a component in the induction phase of AML therapy.
Miscellaneous conditions
Cladribine has also been reported to have activity in a number of much less common hematologic neoplasms, including Langerhans cell histiocytosis,98 systemic mastocytosis,99 hypereosinophilic syndromes,100 myelofibrosis and myeloid metaplasia,101 cutanenous T-cell lymphoma,102 and B-cell prolymphocytic leukemia.77,103
Cladribine toxicities
Cytopenias
Myelosuppression is cladribine's principal toxicity (Table 9).42,85,86,88 Patients with CLL and indolent lymphoma experience similar rates and grades of neutropenia, thrombocytopenia, and anemia. The 2 most commonly administered cladribine regimens, 2-hour bolus and 7-day continuous infusion, also have comparable toxicities. In the front-line setting, grade 3 or 4 neutropenia and thrombocytopenia occurred in up to 60% of patients, with anemia only reported in up to 13%. Infection or fever of unknown origin developed in up to 45% of patients.72,74,83,85,87 Cross-study comparisons demonstrate that pretreated patients given cladribine had similar rates of myelotoxicity and infections as previously untreated patients.72,74 Repetitive administration of cladribine can result in cumulative myelosuppression, which limited many patient cohorts to only 3 or 4 cycles of the drug.26 Instead of analyzing bone marrow cellularity before the initiation of subsequent cycles, it is our practice to simply delay therapy until a patient's absolute neutrophil count has recovered to more than or equal to 1000/mm3 and the platelet count to more than or equal to 100 × 109/L.46,48
Cladribine and fludarabine toxicities
| Study/regimen . | Phase . | Disease . | Grade 3 or 4 . | ||
|---|---|---|---|---|---|
| Neutropenia, % . | Thrombocytopenia, % . | Infections, % . | |||
| Roback et al (2000)86 | |||||
| 2-CdA + P | 3 | CLL | 9 | 9 | 56 (all grades) |
| Robak et al (2004)42 | |||||
| 2-CdA + R | 2 | CLL and NHL (previously treated) | 12 | 13 | 27 |
| Karlsson et al (2007)85 | |||||
| 2-CdA | 3 | CLL | 57 | 43 | 36 |
| F | 3 | CLL | 34 | 24 | 24 |
| Robak et al (2010)88 | |||||
| 2-CdA + Cy | 3 | CLL | 20 | 12 | 28 |
| F + Cy | 3 | CLL | 21 | 11 | 27 |
| Study/regimen . | Phase . | Disease . | Grade 3 or 4 . | ||
|---|---|---|---|---|---|
| Neutropenia, % . | Thrombocytopenia, % . | Infections, % . | |||
| Roback et al (2000)86 | |||||
| 2-CdA + P | 3 | CLL | 9 | 9 | 56 (all grades) |
| Robak et al (2004)42 | |||||
| 2-CdA + R | 2 | CLL and NHL (previously treated) | 12 | 13 | 27 |
| Karlsson et al (2007)85 | |||||
| 2-CdA | 3 | CLL | 57 | 43 | 36 |
| F | 3 | CLL | 34 | 24 | 24 |
| Robak et al (2010)88 | |||||
| 2-CdA + Cy | 3 | CLL | 20 | 12 | 28 |
| F + Cy | 3 | CLL | 21 | 11 | 27 |
2-CdA indicates cladribine; P, prednisone; R, rituximab; F, fludarabine; and Cy, cyclophosphamide.
Cytopenias can persist for greater than 2 months, and have very rarely been associated with thrombocytopenia-induced retinal hemorrhage, pericardial tamponade, and the death of at least one patient from intracerebral hemorrhage.75,104 Fatal neutropenic sepsis has been reported in pretreated patients receiving cladribine.104
Opportunistic infections
Bacterial and opportunistic infections were the most frequent nonhematologic toxicities (Table 9). Marked and prolonged T-cell lymphocytopenia, in addition to monocytopenia and neutropenia, may explain this increased infectious risk. Cladribine has been found to suppress both CD4 and CD8 levels and reduce the CD4/CD8 ratio, lasting from 12 to 54 months.105 Identified opportunistic infections include cytomegalovirus, herpes simplex, dermatomal and disseminated herpes, Pneumocystis carinii, listeriosis, and disseminated fungi. Delayed infections, some occurring up to 31 months after cladribine treatment, occurred in previously treated and untreated patients and resulted in some patient deaths. Mycobacterium tuberculosis reactivation was reported in 2 patients.104 Deaths have also been attributed to interstitial pneumonia and generalized herpes simplex virus infection.35
By extrapolating from the recommendations for antiviral and Pneumocystis prophylaxis in patients treated with fludarabine, cyclophosphamide, and rituximab combinations, patients administered cladribine-based combination therapy should probably receive similar prophylactic antimicrobials, including standard prophylactic doses of bactrim and acyclovir.106 Caution should be exercised in using cladribine and prednisone combinations because of an increased risk for opportunistic and serious infections when purine analogs and steroids are used together.107 Because of the delayed nature of some of these infections, prophylaxis should continue for an extended period of time after treatment discontinuation.106
Secondary malignancy risk
Cladribine's immunosuppressive and DNA-damaging properties introduce the possibility of delayed treatment-induced secondary malignancies, especially when combined with other alkylating agents, such as cyclophosphamide. Several analyses have documented myelodysplasia in cladribine-treated patients, with one series reporting an incidence of 1.6%.45,108,109 Anecdotal experience at Scripps Clinic suggests that the risk of myelodysplasia may be even higher and that cladribine use can be a myelodysplasia risk factor. The exact mechanism and association remain speculative, although cladribine is incorporated into replicating DNA.108,109
A review of 1487 patients treated with either cladribine, an alkylating agent, or their combination did report a statistical increase in lung cancer incidence among cladribine-based therapy, but the authors made no definitive conclusions.110 A comparison of purine analog-treated patients to an age-adjusted population from the Surveillance, Epidemiology, and End Results database found a slight increase in observed-to-expected secondary malignancies. However, this finding was more consistent with the known increased incidence associated with lymphoproliferative disorders and not necessarily from purine analog exposure.111
Autoimmunity
Although autoimmune hemolytic anemia (AIHA) has been associated with cladribine, randomized studies have been unable to absolutely associate cladribine use with the development of AIHA.112-115 CLL patients randomly assigned to cladribine did not have a significant increase in AIHA compared with patients treated with chlorambucil.86 It may be that the underlying lymphoproliferative disorder itself represents the major risk factor for AIHA rather than the cladribine treatment. Nevertheless, the generally advocated approach is to avoid purine nucleoside analogs in patients with established AIHA or immune thrombocytopenia.
PRCA also only has a tenuous association with cladribine. A large retrospective review of 470 CLL patients identified only 8 patients with PRCA: 5 after cladribine therapy and 3 diagnosed before therapy. The incidence of PRCA in patients treated with single-agent cladribine was not greater than other CLL treatments described in the literature.116
Rare toxicities
Several other rare, but documented, toxicities include Stevens-Johnson syndrome/toxic epidermal necrolysis, stroke, tumor lysis syndrome, and transfusion-associated graft versus host disease.75,104 At approved doses, cladribine has been reported to produce a mild to moderate, but generally reversible, neuropathy in 10% to 15% of patients.117 This risk is not thought to be high because cladribine has been successfully applied to the treatment of multiple sclerosis.118 One patient with a preexisting paraneoplastic syndrome died of a rapidly progressive sensorimotor peripheral neuropathy after completing 2 courses of low-dose cladribine (0.1 mg/kg per day 2-hour infusion for 7 days).119 In addition, at very elevated doses (more than triple the approved dose), a delayed severe motor weakness developed.120
Future research proposals
Cladribine's most influential role in the treatment of indolent lymphoid malignancies will probably be as a component of front-line multiagent therapy. Previous attempts to combine cladribine with standard cytotoxic agents, such as cyclophosphamide and mitoxantrone, resulted in significant toxicities with only minor advantages over single-agent cladribine. Instead, regimens that incorporate cladribine with recently developed novel agents appear to be mechanistically the most promising research direction to take. These newer agents include lenalidomide and bortezomib, both offering broad activity across the spectrum of indolent lymphoma subtypes, nonoverlapping mechanisms of action compared with cladribine, and can be used in combination with rituximab. Another interesting hypothesis is to combine cladribine with one of the newly developed PARP inhibitors.121 Cladribine can trigger apoptosis by stimulating PARP expression, which depletes intracellular NAD and ATP (“Mechanism of action”), and PARP inhibitors may contribute to this process. However, a comprehensive preclinical analysis of this approach must be performed before its clinical implementation as there is also a potential for cell rescue if the PARP inhibitor prevents nucleotide depletion.
A further clinical trial proposal is the addition of bendamustine to cladribine together with rituximab. Bendamustine is a molecule that has a purine-like ring but acts as an atypical alkylating agent.122 As a purine nucleoside analog, cladribine may provide valuable structural synergies to bendamustine. Finally, rituximab has been safely and successfully used with both cladribine and bendamustine.123 Although this approach may cause significant myelosuppression, cladribine and bendamustine are among the most effective single-agent therapies for both CLL and MCL, possibly making this a highly effective regimen.
In conclusion, despite its spectacular success in treating HCL, cladribine has not been a widely adopted therapy for other lymphoproliferative disorders. We have summarized the literature demonstrating cladribine's remarkable activity in indolent lymphoid malignancies other than HCL, as well as in AML more recently. Cladribine induces clinically significant responses across a range of indolent lymphomas and CLL, among both untreated and previously treated patients. Lymphoplasmacytic lymphoma (WM) and marginal zone lymphoma appear particularly sensitive to cladribine's action. Patients with MCL, an aggressive lymphoproliferative disorder with limited therapeutic options, experienced durable responses after cladribine therapy. Cross-study comparisons and a phase 3 study in CLL suggested that cladribine may confer therapeutic advantages over fludarabine, supporting the continued clinical development of cladribine. Standard cladribine dosing is generally well tolerated, mainly causing cytopenias and fevers.
This review highlights cladribine's role in the management of patients with indolent lymphomas; however, the majority of these studies were small in size and included heterogeneous patient populations. In addition, the most appropriate dosing regimens and combination approaches have not been clearly defined. A mechanistic review suggests that cladribine may have synergy with some recently approved novel agents. Clearly, cladribine's promising role in indolent lymphomas, CLL, and AML has not been fully explored. It is incumbent on investigators in hematologic malignancies to be familiar with cladribine's unique activity and advantages in these disease states so that its future inclusion in clinical research protocols can be best exploited to advance patient care.
Authorship
Contribution: D.S.S. and A.S. conceived of the paper; D.S.S. composed the manuscript and compiled literature data; H.J.M. and E.D.S. compiled medical literature data, prepared tables and figures, and contributed to manuscript composition; and A.S. edited the manuscript.
Conflict-of-interest disclosure: A.S. is a consultant to EMD Serono for oral cladribine in multiple sclerosis. The remaining authors declare no competing financial interests.
Correspondence: Alan Saven, Division of Hematology/Oncology, Scripps Clinic Medical Group, 10666 North Torrey Pines Rd, MS217, La Jolla, CA 92037; e-mail: saven.alan@scrippshealth.org.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal