Abstract
Abstract 3469
Stem cell transplantation is important in the management of multiple myeloma. In the United States, the standard of care is administration of growth factors to accelerate neutrophil recovery after stem cell transplant. The need for growth factors after transplant has not been investigated recently.
We analyzed a cohort of 166 patients at our institution who underwent autologous transplant for multiple myeloma without receiving growth factors after transplant and compared them with 498 patients who received standard filgrastim beginning on posttransplant day 5. TABLE
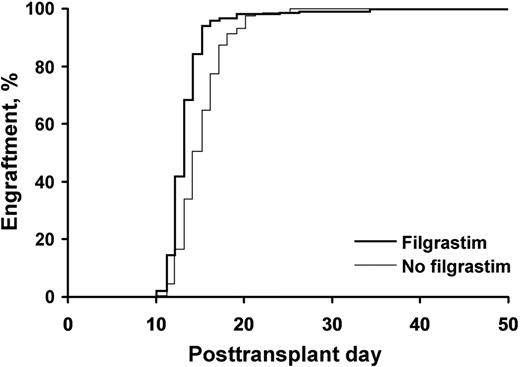
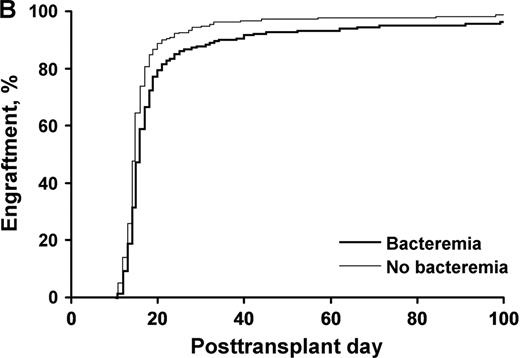
A neutrophil count of 500/μL was achieved in a median of 12.5 days in patients receiving growth factor, compared with 13.5 days in those not receiving growth factor (P<.001) Fig 1. Platelet engraftment was identical (median, 14.5 days; P=.12) in both groups, despite a lower median number of CD34+ cells infused in patients who did not receive growth factors. Incidence of nonstaphylococcal bacteremia was identical in both groups. The median hospital stay was 3.5 days shorter in the group not receiving growth factor. Bacteremia impacted platelet but not neutrophil engraftment. Figure 2
It is feasible and reasonable to perform autologous stem cell transplant for multiple myeloma without administering growth factors. Estimated savings would be $4,600 for each transplanted patient. Growth factor administration is not required for a safe outcome and decreases the number and severity of some of the fluid-related complications after transplant such as acute respiratory distress syndrome, allergic reactions, alveolar hemorrhage, and rarely splenic rupture.
Patient Characteristics
| . | Patient Group . | . | |
|---|---|---|---|
| Characteristic . | Filgrastim (n=498) . | No Filgrastim (n=166) . | P Value . |
| Men | 290 (58) | 102 (61) | .52 |
| Age, y | 59.8 (53.0–65.4) | 61 (53.9–66.8) | .25 |
| Serum M spike, g/dL | 0.8 (0.1–1.8) | 0.6 (0.1–1.8) | .22 |
| Urine total protein, g/24 h | 0.10 (0.06–0.33) | 0.09 (0.05–0.28) | .67 |
| b2M, mcg/mL | 2.56 (1.97–3.71) | 2.71 (2.10–3.66) | .13 |
| LDH, units/L | 187 (158–229) | 184 (152–214) | .35 |
| Myeloma bone disease | 422 (85) | 146 (88) | .37 |
| Marrow PCs, % | 8 (2–21) | 8 (2–23) | .60 |
| Radiotherapy | 137 (28) | 48 (29) | .76 |
| Abnormal metaphase cytogenetics | 98 (20) (n=494) | 27 (16) (n=165) | .36 |
| Albumin, g/dL | 3.7 (3.3–4.0) | 3.3 (3.1–3.4) | <.001 |
| Creatinine, mg/dL | 1.0 (0.9–1.2) | 0.9 (0.8–1.1) | .002 |
| ISS | |||
| Stage I | 222 (46) (n=481) | 21 (13) (n=165) | <.001 |
| Stage II | 200 (42) | 125 (76) | <.001 |
| Stage III | 59 (12) | 19 (12) | NA |
| CD34+ cells infused, ×106 cells/kg | 4.33 (3.78–5.51) | 4.19 (3.50–5.03) | .01 |
| All-cause mortality before day +100 | 10 (2) | 3 (2) | NA |
| . | Patient Group . | . | |
|---|---|---|---|
| Characteristic . | Filgrastim (n=498) . | No Filgrastim (n=166) . | P Value . |
| Men | 290 (58) | 102 (61) | .52 |
| Age, y | 59.8 (53.0–65.4) | 61 (53.9–66.8) | .25 |
| Serum M spike, g/dL | 0.8 (0.1–1.8) | 0.6 (0.1–1.8) | .22 |
| Urine total protein, g/24 h | 0.10 (0.06–0.33) | 0.09 (0.05–0.28) | .67 |
| b2M, mcg/mL | 2.56 (1.97–3.71) | 2.71 (2.10–3.66) | .13 |
| LDH, units/L | 187 (158–229) | 184 (152–214) | .35 |
| Myeloma bone disease | 422 (85) | 146 (88) | .37 |
| Marrow PCs, % | 8 (2–21) | 8 (2–23) | .60 |
| Radiotherapy | 137 (28) | 48 (29) | .76 |
| Abnormal metaphase cytogenetics | 98 (20) (n=494) | 27 (16) (n=165) | .36 |
| Albumin, g/dL | 3.7 (3.3–4.0) | 3.3 (3.1–3.4) | <.001 |
| Creatinine, mg/dL | 1.0 (0.9–1.2) | 0.9 (0.8–1.1) | .002 |
| ISS | |||
| Stage I | 222 (46) (n=481) | 21 (13) (n=165) | <.001 |
| Stage II | 200 (42) | 125 (76) | <.001 |
| Stage III | 59 (12) | 19 (12) | NA |
| CD34+ cells infused, ×106 cells/kg | 4.33 (3.78–5.51) | 4.19 (3.50–5.03) | .01 |
| All-cause mortality before day +100 | 10 (2) | 3 (2) | NA |
Patients who received filgrastim achieved neutrophil engraftment faster than those who did not P<.001.
Patients who received filgrastim achieved neutrophil engraftment faster than those who did not P<.001.
Presence of bacteremia had no effect on neutrophil engraftment (p=.28) but slowed the rate of platelet engraftment platlets P<.001.
Presence of bacteremia had no effect on neutrophil engraftment (p=.28) but slowed the rate of platelet engraftment platlets P<.001.
Gertz:Celgene: Honoraria; Millenium: Honoraria, Membership on an entity's Board of Directors or advisory committees. Lacy:Celgene: Research Funding. Dispenzieri:Celgene: Honoraria, Research Funding; Binding Site: Honoraria. Kumar:Celgene: Consultancy, Research Funding; Millennium: Research Funding; Merck: Consultancy, Research Funding; Novartis: Research Funding; Genzyme: Consultancy, Research Funding; Cephalon: Research Funding.
Asterisk with author names denotes non-ASH members.

This icon denotes an abstract that is clinically relevant.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal