Abstract
Although it has been clearly demonstrated that venous thromboembolism is associated with an increased risk of subsequent overt cancer and arterial cardiovascular events in comparison with control populations, whether this association also applies to patients with isolated (ie, without concomitant involvement of the deep vein system) superficial vein thrombosis (SVT) in the legs is unknown. In 737 consecutive patients with isolated SVT not involving the sapheno-femoral junction, we conducted a retrospective investigation to assess the rate of cancer and that of arterial cardiovascular events occurring during follow-up. The event rates were compared with those occurring in 1438 controls having comparable characteristics. Both cases and controls were followed-up for an average period of 26 ± 8 months (range, 3-45). Malignancy was diagnosed in 26 cases (3.5%) and 56 controls (3.9%), leading to a hazard ratio of 0.86 (95% confidence interval, 0.55%-1.35%). Arterial cardiovascular events occurred in 32 cases (4.3%) and 63 controls (4.4%), leading to a hazard ratio of 0.97 (95% confidence interval, 0.63%-1.50%). We conclude that the occurrence of isolated SVT in the legs does not place patients at an increased risk of malignancies or arterial cardiovascular events. Whether this conclusion also applies to patients whose thrombosis involves the sapheno-femoral junction remains to be demonstrated.
Introduction
Although it has been clearly demonstrated that venous thromboembolism (VTE) is associated with an increased risk of subsequent overt cancer1 and arterial cardiovascular disorders2 in comparison with control populations, it is unknown whether this association also applies to patients presenting with isolated (ie, without concomitant involvement of the deep vein system) superficial vein thrombosis (SVT) in the legs. On the basis of anecdotal reports3 and findings from case-series,4,5 SVT is generally perceived as a marker of occult malignancy. However, a recent enquiry conducted in The Netherlands in 250 patients with spontaneous SVT and in matched control individuals identified through a search in an electronic database from a few primary health care centers failed to show an increase in the risk of cancer in patients with SVT.6
In the recently published Calisto study, ∼ 3000 patients with isolated SVT were randomized to receive either fondaparinux or placebo for 45 days.7 The study showed a remarkable advantage of fondaparinux over placebo for prevention of VTE events during this period. In 737 consecutive patients recruited in 32 centers that agreed to participate in this substudy, a retrospective enquiry was conducted to assess the rate of cancer and that of arterial thromboembolic events occurring after randomization. The event rates were compared with those occurring in 1438 controls having comparable characteristics recruited from the general population of northern Italy. Both cases and controls were followed-up for an average period of more than 2 years.
Methods
Patients
The Calisto study recruited 3002 patients 18 years of age or older between March 2007 and May 2009 who had acute symptomatic isolated SVT of the lower extremities at least 5 cm long, as confirmed by standardized compression ultrasonography.7 Consecutive patients recruited in each of 32 centers that agreed to participate in this substudy were eligible for the current investigation. Patients who had a history of cancer, symptomatic VTE, or symptomatic arterial cardiovascular events at any time before randomization into Calisto were excluded from this analysis. Other relevant exclusion criteria in the main Calisto study include SVT located within 3 cm of the sapheno-femoral junction; SVT associated with sclerotherapy or placement of an intravenous catheter; requirement for ligation of the sapheno-femoral junction or planned stripping of varicose veins; antithrombotic or nonsteroidal anti-inflammatory agents for > 48 or 72 hours, respectively, before the Calisto randomization; severe hepatic or renal impairment; thrombocytopenia; major surgery within the previous 3 months; and pregnant women or women of childbearing age not using a reliable contraceptive method.
Between March and September 2010, all consenting patients in participating centers were evaluated for the presence of risk factors for cancer or atherosclerosis and the development of either malignancies or arterial cardiovascular events in the follow-up after randomization into the Calisto study. For this purpose, they were contacted by telephone. Whenever information could not be obtained directly from patients, either a close relative or the family physician was interviewed. All suspected events had to be confirmed by objective tests, available for review by the study investigators. Date and cause of death were ascertained for patients who died during the follow-up period.
Data on the following variables were recorded using a case report form: smoking status (defined as regular smoking at least 10 cigarettes per day); high blood pressure (defined by the use of antihypertensive drugs); obesity (defined as a body-mass index > 25 kg/m2); diabetes mellitus (defined by the use of antidiabetic drugs); hyperlipidemia (defined by the use of lipid-lowering drugs); and alcohol abuse (defined as a pattern of drinking that results in harm to one's health, interpersonal relationships, or ability to work).
There was no standardized screening for occult cancer in the inception cohort. Thus, all cancer outcomes that presented after the diagnosis of SVT were detected based on clinical features that would prompt diagnostic imaging, histologic confirmation, or both. The diagnosis of nonfatal or fatal symptomatic cardiovascular disease, defined as ischemic stroke, ST-elevation or non–ST-elevation acute coronary syndrome, peripheral arterial disease, or sudden unexplained death was accepted on the basis of standardized criteria.
The study was conducted in accordance with the ethical principles of the Declaration of Helsinki. The Calisto study was approved by the Institutional Review Board in each of the participating centers. The Last Evidences Of Genetic Risk factor in the Aged (LEOGRA) study was approved by the Ethics Committee of the University of Padua and the Local Health Unit No. 4 of the Veneto Region.
Controls
The controls were recruited in a 2:1 ratio from the general population of LEOGRA study, ongoing in northern Italy.8-12 A group of 1438 subjects matched with cases for age (± 3 years) and sex, who had no history of cancer and had not experienced either venous (including SVT) or arterial thromboembolic disorders before the beginning of the observation, served as study controls. For 36 cases, only 1 matched control could be identified. Controls had comparable duration of follow-up of cases. Information on risk factors and the occurrence of subsequent events was collected after an identical pattern.
Statistical analysis
For the comparison of the baseline characteristics between cases and controls, ANOVA and the Bonferroni posthoc test were used. New cases of malignancies and cardiovascular events were expressed as cumulative incidence in the follow-up period. Frequencies were compared with the Pearson χ2 test.
Kaplan-Meier estimates and associated 95% confidence intervals (CIs) were calculated, and curves of cumulative incidence were plotted. Multivariate Cox proportional hazards modeling was used to compare the risk for new cancer and new arterial cardiovascular disorders in patients and matched controls, and to calculate hazard ratios (HRs) with 95% CIs.
All calculations were performed with SPSS Version 18.0 (SPSS).
Results
Of the 911 patients recruited from the Calisto study in the 32 participating centers, 63 (6.9%) could not be retrieved or did not agree to participate in this enquiry. Of the remaining 848, 111 (13.1%) were excluded because of history of cancer, previous VTE or previous arterial cardiovascular events. Thus, 737 were recruited for the current investigation, of whom 369 (50.1%) had been treated with fondaparinux. Mean age was 59.3 ± 13.8 years, and 251 (34.1%) patients were male. They were followed-up for an average period of 26 ± 8 months (range, 3-45).
Of the 1438 control individuals, mean age was 59.9 ± 16.7 years and 490 (34.1%) were male. They were followed-up for an average period of 26 ± 8 months (range, 3- 45).
The distribution of the main risk factors for cancer and atherosclerosis (smoking, diabetes, obesity, high blood pressure, alcohol abuse, and hyperlipidemia) did not differ between the 2 study groups (Table 1).
Main characteristics of the study patients and controls
| . | Patients (n = 737) . | Controls (n = 1438) . |
|---|---|---|
| Age, mean ± SD | 59.3 ± 13.8 | 59.9 ± 16.7 |
| Males | 251 (34.1) | 490 (34.1) |
| Risk factors | ||
| High blood pressure | 301 (40.8) | 546 (38.0) |
| Obesity | 226 (30.7) | 348 (24.2) |
| Smoking habit | 174 (23.6) | 345 (24.0) |
| Hyperlipidemia | 79 (10.9) | 158 (11.0) |
| Alcohol abuse | 54 (7.3) | 106 (7.4) |
| Diabetes mellitus | 48 (6.5) | 99 (6.9) |
| . | Patients (n = 737) . | Controls (n = 1438) . |
|---|---|---|
| Age, mean ± SD | 59.3 ± 13.8 | 59.9 ± 16.7 |
| Males | 251 (34.1) | 490 (34.1) |
| Risk factors | ||
| High blood pressure | 301 (40.8) | 546 (38.0) |
| Obesity | 226 (30.7) | 348 (24.2) |
| Smoking habit | 174 (23.6) | 345 (24.0) |
| Hyperlipidemia | 79 (10.9) | 158 (11.0) |
| Alcohol abuse | 54 (7.3) | 106 (7.4) |
| Diabetes mellitus | 48 (6.5) | 99 (6.9) |
Numbers in parentheses indicate percentages.
Three (0.4%) patients died and 6 (0.8%) were lost to follow-up. The corresponding figure for controls was 6 (0.4%) and 12 (0.8%), respectively.
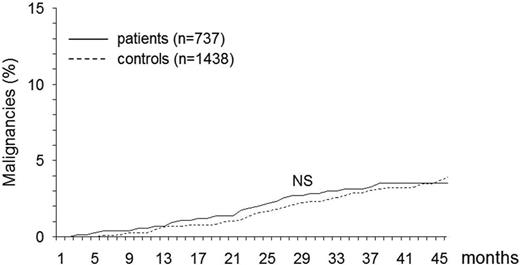
During the follow-up period, symptomatic malignancies were diagnosed in 26 patients (of whom 14 had been treated with fondaparinux) and 56 controls, resulting in a cumulative incidence of 3.5% (95% CI, 2.9%-4.8%) and 3.9% (95% CI, 2.9%-4.9%), respectively (Figure 1). Table 2 reports the main types of cancer that developed in the 2 study populations. The HR for developing symptomatic malignancies was 0.86 (95% CI, 0.55%-1.35%).
Cumulative incidence of new malignancies in patients with SVT and in control subjects. NS indicates not significant.
Cumulative incidence of new malignancies in patients with SVT and in control subjects. NS indicates not significant.
Malignancies occurring during the follow-up of the study patients and controls
| . | Patients (n = 737) . | Controls (n = 1438) . |
|---|---|---|
| Genito-urinary | 8 (1.0) | 16 (1.1) |
| Gastrointestinal and digestive | 5 (0.7) | 14 (0.9) |
| Skin* | 5 (0.7) | 4 (0.3) |
| Respiratory tract | 3 (0.4) | 8 (0.6) |
| Breast | 3 (0.4) | 7 (0.5) |
| Hematological malignancies | 2 (0.3) | 5 (0.3) |
| Brain | 2 (0.1) | |
| Total | 26 (3.5) | 56 (3.9) |
| . | Patients (n = 737) . | Controls (n = 1438) . |
|---|---|---|
| Genito-urinary | 8 (1.0) | 16 (1.1) |
| Gastrointestinal and digestive | 5 (0.7) | 14 (0.9) |
| Skin* | 5 (0.7) | 4 (0.3) |
| Respiratory tract | 3 (0.4) | 8 (0.6) |
| Breast | 3 (0.4) | 7 (0.5) |
| Hematological malignancies | 2 (0.3) | 5 (0.3) |
| Brain | 2 (0.1) | |
| Total | 26 (3.5) | 56 (3.9) |
Numbers in parentheses indicate percentages.
Including basal cell carcinoma.
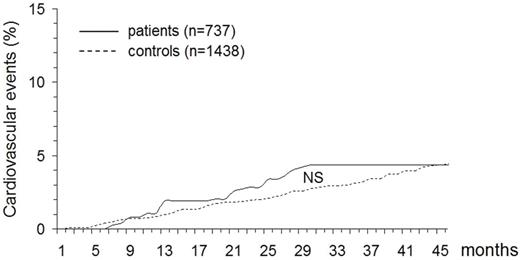
During the follow-up period, cardiovascular events occurred in 32 patients (of whom 15 had been treated with fondaparinux) and 63 controls, resulting in a cumulative incidence of 4.3% (95% CI, 2.9%-5.8%) and 4.4% (95% CI, 3.4%-5.5%), respectively (Figure 2). Table 3 reports the main types of events that developed in the 2 study cohorts. The HR for developing cardiovascular events was 0.97 (95% CI, 0.63%-1.50%).
Cumulative incidence of new arterial cardiovascular disorders in patients with SVT and in control subjects. NS indicates not significant
Cumulative incidence of new arterial cardiovascular disorders in patients with SVT and in control subjects. NS indicates not significant
Arterial cardiovascular events occurring during the follow-up of the study patients and controls
| . | Patients (n = 737) . | Controls (n = 1438) . |
|---|---|---|
| Acute coronary syndrome | 16 (2.2) | 33 (2.3) |
| Ischemic stroke | 12 (1.6) | 19 (1.3) |
| Peripheral arterial disease | 3 (0.4) | 9 (0.6) |
| Sudden unexplained death | 1 (0.1) | 2 (0.1) |
| Total | 32 (4.3) | 63 (4.4) |
| . | Patients (n = 737) . | Controls (n = 1438) . |
|---|---|---|
| Acute coronary syndrome | 16 (2.2) | 33 (2.3) |
| Ischemic stroke | 12 (1.6) | 19 (1.3) |
| Peripheral arterial disease | 3 (0.4) | 9 (0.6) |
| Sudden unexplained death | 1 (0.1) | 2 (0.1) |
| Total | 32 (4.3) | 63 (4.4) |
Numbers in parentheses indicate percentages.
Discussion
In recent years, many similarities have been shown between SVT and deep venous thrombosis (DVT). These conditions share common risk factors13 and are frequently concomitant.14,15 In addition, the natural history of isolated SVT in the legs, in analogy with that of DVT, is characterized by an unexpectedly high incidence of symptomatic VTE complications and extension or recurrence of the vascular disorder,7,15 so that proper anticoagulation with either low molecular weight heparin or fondaparinux is required for at least 4 weeks.7,16 Although it also would be interesting to know whether SVT places patients at an increased risk of subsequent malignancies, arterial cardiovascular events, or both, this information is unknown.
Our results, obtained by investigating the development of the above-mentioned events in the long-term follow-up (26 months on average) of a large cohort of consecutive patients with isolated SVT in the legs who had been enrolled in the Calisto study7 show that neither the incidence of subsequent cancer nor that of subsequent arterial cardiovascular events exceeds the rate expected in the general population (Figures 1–2). In addition, as shown in Tables 2 and 3, the type of events fully overlaps that observed in unselected control individuals. It is worth specifying that all cases and controls who had contemporary or previous VTE, a history of cancer or symptomatic complications of atherosclerosis were excluded from the analysis, that only symptomatic events qualified as study outcomes, and that the diagnosis of cancer and that of arterial cardiovascular events was accepted in the presence of stringent criteria. Not surprisingly, treatment with fondaparinux for the initial SVT did not impact the study results during this extended follow-up period.
Failure to record an increased risk of cancer is in contrast with anecdotal reports,3 with findings from case-series4,5 and with the general perception that thrombotic manifestations may be a sign of occult cancer irrespective of their localization and severity.1 It should be considered, however, that in most previous reports DVT was not excluded by systematic ultrasound examination at baseline. Because this may occur in up to 30% of patients with SVT,14,15 we cannot exclude that the reported risk of cancer was because of the coexistence of DVT. Our results are consistent with those of a recent enquiry conducted in The Netherlands,6 and suggest that in patients with isolated SVT in the legs—unlike those with DVT—neither the risk of subsequent manifest cancer nor that of subsequent arterial cardiovascular events exceeds that expected in the general population. Although the acute clinical course of isolated SVT in the legs is now regarded as less favorable than was believed in the past,7,15 the overall prognosis of this disease seems to be less malignant than that of DVT.
Among the potential limitations of our investigation is first, the failure to recruit all patients enrolled in the Calisto study. However, there was no selection bias, because invitation to participate in this substudy was offered to all centers. Participating centers enquired into the course of all accepting patients who had been recruited at their institution, and the rate of patients who could not be retrieved or were lost to follow-up was negligible. Second, based on the retrospective nature of the study design and the absence of central independent adjudication, the rate of events may have been underestimated. However, study investigators were instructed to proceed in a standardized way using a predefined questionnaire, and all clinically suspected events were objectively confirmed according to widely accepted stringent criteria. A central adjudication would have been unfeasible within the framework of this Calisto substudy. Of interest, even if the event rate had been as much as 25% higher than reported here, there would not have been a significant difference from that observed in matched control individuals. Third, control individuals could not be enrolled from the countries where the cases came from. However, cases and controls had similar baseline characteristics and risk factors for cancer and atherosclerosis and had comparable follow-up. Of interest, controls were recruited from a region where the cardiovascular risk would not be expected to be higher than that in controls from the other participating countries.17 Finally, our study results may only apply to patients with isolated SVT meeting the eligibility criteria required by the Calisto study.7
The potential implications of our study results are important. They suggest that in the absence of concomitant involvement of the deep vein system, patients with SVT can be reassured that the risk of subsequent cancer, myocardial infarction, or stroke does not exceed that expected in healthy individuals. Our results need confirmation from larger prospective cohort or population-based studies.
The online version of the article contains a data supplement.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 USC section 1734.
Acknowledgments
The authors are indebted to Dr Steven Quentzel for valuable advice and help in reviewing the manuscript.
Authorship
Contribution: P.P., A.L., and H.D. designed and performed the research in collaboration with a number of Calisto investigators; E.C. and V.T. contributed data from the control population; E.C. analyzed data; P.P. wrote the paper; and all authors agreed on the final version of the manuscript.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
A complete list of the members of the Calisto Study Group appears in the supplemental Appendix (available on the Blood Web site; see the Supplemental Materials link at the top of the online article).
Correspondence: Paolo Prandoni, Department of Cardiothoracic and Vascular Sciences, Clinica Medica 2, University of Padua, Via Giustiniani 2, 35128, Padua, Italy; e-mail: paoloprandoni@tin.it.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal