Abstract
Abstract 3033
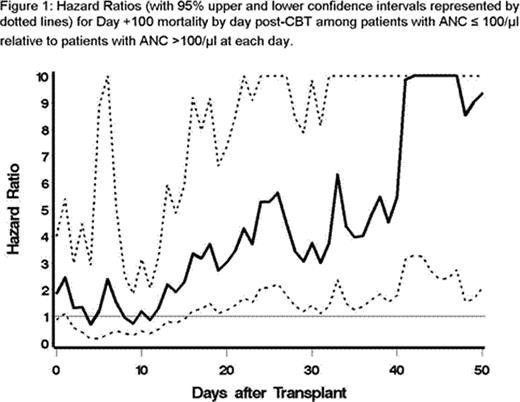
Background: Cord blood transplant (CBT) recipients have higher infection-related morbidity and mortality than recipients of other stem cells sources following allogeneic transplant due to delayed hematopoietic recovery and immune reconstitution. Even in recipients of myeloablative (MA) double cord blood transplant (dCBT), time to engraftment (defined as the first of two consecutive days with an absolute neutrophil count (ANC) ≥ 500/μl) is delayed more than three weeks resulting in higher rates of infection in the first 100 days post-transplant. However, a better understanding of the relationship between duration of neutropenia and risk of early transplant related mortality is needed to assess the clinical impact of methodologies aimed at reducing neutropenia post transplant. Previously, the relationship between severe neutropenia (ANC ≤ 100/μl) and risk of death was evaluated for allogeneic bone marrow transplant recipients using proportional hazards models and demonstrated a significantly increased risk for those with severe neutropenia at day 15 or beyond following transplantation (Offner et al, Blood, 1996; 88(10): 4058–62). Here we use a similar model to determine how duration of severe neutropenia relates to risk of death following CBT. Methods: All patients (n=137) who received a CBT on a research protocol at a single institution from 2006–2010 were eligible. On each day from day 0 to day +100, surviving patients were divided into those with ANC ≤ 100/μl and those with ANC >100/μl and the number of patients who died by day +100 determined for each group. Hazard ratios (HR) with 95% confidence intervals for day +100 mortality were then calculated for day post-CBT with the HR representing the risk of day +100 death among those with ANC ≤ 100/μl relative to those with ANC >100/μl for each day. Results: Of the 137 patients who received a CBT on a research protocol, 99 patients (72%) received MA conditioning regimens and 38 patients (28%) received reduced-intensity conditioning regimens (RIC). Twenty-two patients (16%) received a single cord blood unit while the remainder received dCBT. As the overall results and trends observed for patients receiving MA or RIC regimens were similar, only the combined results are presented. Thirty-one patients (23%) died before day +100. Causes of death were primary graft failure (17), infection (7), disease relapse (5), multi-organ failure (1), and leukoencephalopathy (1). The median time to engraftment was the same (20 days) for those with death before day+100 (9–39 days) as those alive at day+100 (6–69 days). The hazard ratio for day 100 mortality by day post-CBT was significantly higher for patients with ANC ≤ 100/μl beginning on day +16 and remained so through day +50, at which point the number of patients with ANC ≤ 100/μl was small and thus the calculations were no longer significant. The HRs for each day post-CBT from day 0 to day +50 are plotted in Figure 1 along with their 95% upper and lower confidence intervals. From the graphical plot of these HRs, one can identify date ranges (days 0–12, days 13–23, days 24–40, days 41–100) where the HRs are roughly equivalent with a clear increase in HR in the next date range. Modeling ANC ≤ 100/μl as a time-dependent covariate, we calculated HRs for each of these date ranges and found a significantly increased risk of day 100 mortality for days 12–23 (HR=2.96, p=0.01), days 24–40 (HR=5.53, p=0.0004), and days 41–100 (HR=14.59, p<0.0001) for patients with ANC ≤ 100/μl. The HR was 1.16 (0.49–2.76, p=0.73) for days 0–12; however, some of these patients had initial autologous recovery following RIC regimens and poor outcomes. Conclusions: Our study demonstrates that severe neutropenia, defined as ANC ≤ 100, poses a significant increased risk of day 100 mortality for recipients of CBT as early as 12–23 days post-CBT. Importantly, this patient cohort was transplanted in the modern era of aggressive supportive care, including use of newer antimicrobials. However, despite this, severe neutropenia remains a significant risk factor for day 100 mortality in recipients of CBT. Interestingly, median time to engraftment (ANC ≥ 500/μl) was the same for those with death before day+100 as those alive at day+100 suggesting that time to ANC of 100 may be a better predictor of early death following CBT. Thus, strategies that result in more rapid myeloid recovery (to an ANC of 100) remain essential for recipients of CBT.
Disclosures:
No relevant conflicts of interest to declare.
Author notes
*
Asterisk with author names denotes non-ASH members.
© 2011 by The American Society of Hematology
2011

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal