Abstract
Homoharringtonine-based induction regimens have been widely used in China for patients with acute myeloid leukemia (AML), which have shown to improve the rate of complete remission (CR) and long-term survival. We aimed to further evaluate its efficacy and safety in treatment of de novo AML.
This phase 3 study was done in 17 institutions in China. Patients between the age of 14 and 59 with untreated AML were randomly assigned to receive HAA (homoharringtonine 2 mg/m2/day, days 1–7; cytarabine 100 mg/m2/day, days 1–7, aclarubicin 20 mg/day, days 1–7), HAD (homoharringtonine 2 mg/m2/day, days 1–7; cytarabine 100 mg/m2/day, days 1–7; daunorubicin 40 mg/m2/day, days 1–3) or DA (daunorubicin 40–45 mg/m2/day, days 1–3; cytarabine 100 mg/m2/day, days 1–7) regimen as induction therapy. Patients who achieved partial remission or had a decrease of blast ¡Ý60% could receive a same second induction course. All patients who had a complete remission were offered the same consolidation chemotherapy according to the cytogenetic-risk. The primary endpoints were CR and event-free survival (EFS). The trial is registered in Chinese Clinical Trial Register, number ChiCTR-TRC-06000054.
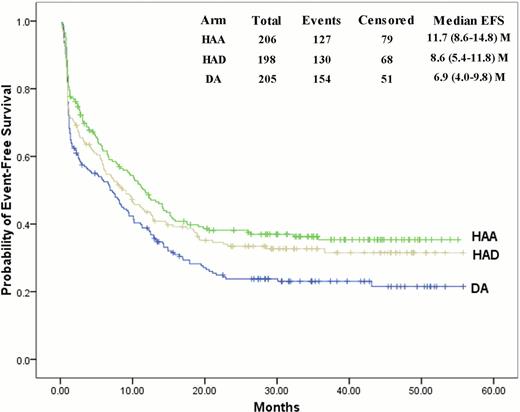
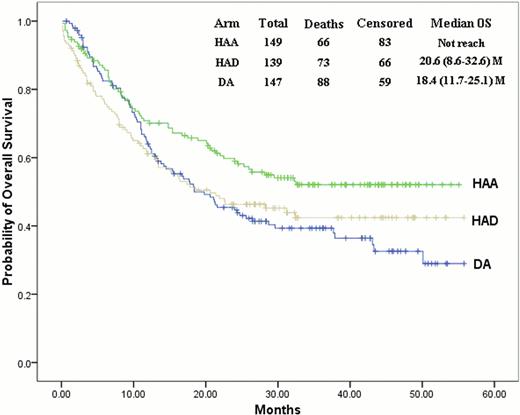
620 patients were randomly assigned to receive HAA (n=207), HAD (n=206) and DA (n=207) regimens. HAA or HAD regimen, as compared with DA regimen, resulted in a higher rate of CR in the first course of induction therapy (67.5% vs. 54.0%, P=0.005; 64.9% vs. 54.0%, P=0.026, respectively). The overall CR rate remained significantly higher in the HAA arm as compared with DA arm (75.0% vs. 61.9%, P=0.005). HAA or HAD regimen has similar rates of adverse events as compared with DA regimen, but was associated with significantly increased risk of induction death (5.8% vs. 1.0%, P=0.007; 6.6% vs. 1.0%, P=0.003, respectively). The EFS was greatly improved in the HAA arm (3-year EFS 35.4±3.5% vs.23.1±3.1%, P=0.002), while not significantly in the HAD arm (3-year EFS 32.7±3.5% vs.23.1±3.1%, P=0.078) as compared with the DA arm. Overall survival (OS) and relapse-free survival (RFS) did not differ significantly in the HAA or HAD arm as compared with DA arm, but an OS and RFS advantage of the HAA arm over the DA arm was observed in patients with favorable or intermediate cytogenetic profile (OS: P=0.014; RFS: P=0.022, respectively). Patients in the HAD arm with NPM1 but not FLT3ITD mutations, as compared with the patients in the DA arm, had an improved EFS (P=0.038). In intermediate cytogenetic profile, patients with mutant CEBPA had prolonged RFS in the HAA arm as compared with the DA arm (P=0.045).
Homoharringtonine-based induction regimens are associated with a higher rate of CR and improved survival as compared with DA regimen in AML. The toxicity is mild with the exception of a higher rate of induction death.
Kaplan-Meier estimates for the event-free survival in all patients.
Kaplan-Meier estimates for overall survival in patients with favorable or intermediate cytogenetic profile.
Kaplan-Meier estimates for overall survival in patients with favorable or intermediate cytogenetic profile.
Kaplan-Meier estimates for relapse-free survival in patients with favorable or intermediate cytogenetic profile
Kaplan-Meier estimates for relapse-free survival in patients with favorable or intermediate cytogenetic profile
No relevant conflicts of interest to declare.

This icon denotes a clinically relevant abstract
Author notes
Asterisk with author names denotes non-ASH members.


This feature is available to Subscribers Only
Sign In or Create an Account Close Modal