Abstract
Abstract 4719
Ephrin signaling has been shown to contribute to the pathogenesis of many solid tumors with respect to tumor growth, tumor cell survival, angiogenesis, and metastasizing capacity (Cytokine Growth Factor Rev. Dec;15(6):419-33, et al. Neuro Oncol. 2012). Recently, an aberrant DNA methylation status of ephrin receptors and ligands was described to be associated with outcome in acute lymphoblastic leukemia (Blood.2010 Mar 25;115(12):2412-9). In acute myeloid leukemia (AML), we found an intriguing heterogeneity in membrane receptor expression levels of EphB1. Therefore, we challenged to evaluate the role of EphB1 receptor forward signaling in AML.
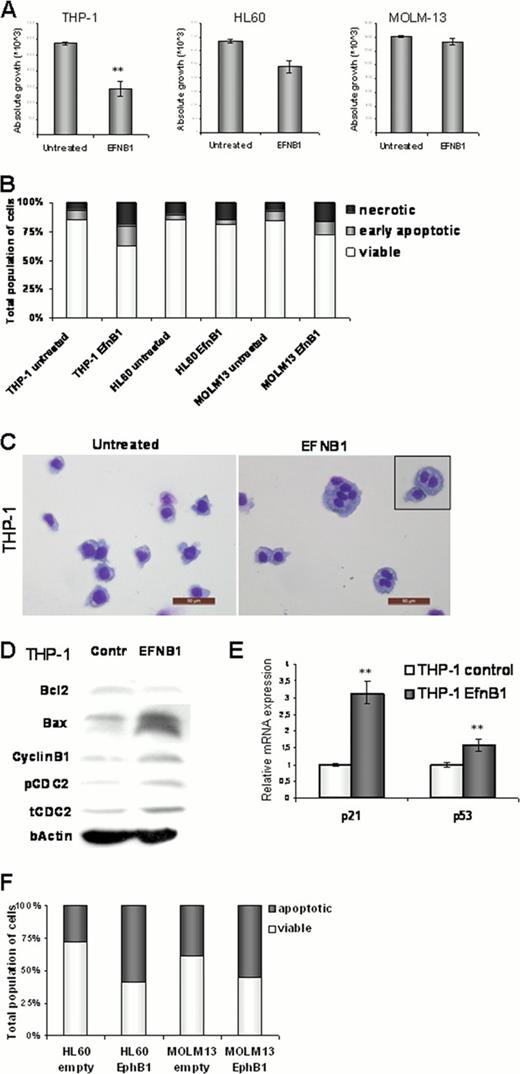
We investigated the influence of the EphB1 receptor forward signaling in THP-1 (EphB1high), HL60 (EphB1int), and MOLM13 (EphB1low) AML cell lines through exogenous stimulation with the EphB1 ligand; EfnB1. EfnB1 stimulation of the AML cell lines demonstrated to reduce AML growth solely in the EphB1high and EphB1int cell lines (Fig. 1A,P = 0.001 and P =.056). In addition, EfnB1 stimulation induced apoptosis most pronounced in the EphB1high cells (Fig. 1B). Interestingly, MGG stained cytospins of EfnB1 treated THP-1 showed multinucleation of AML cells (Fig. 1C). We hypothesized that these phenotypic effects could be assigned to cell cycle arrest in THP-1 cells. Additionally, cell cycle regulatory proteins CDC2 and CyclinB1 were evaluated by immunoblotting of EfnB1 stimulated THP-1 cells. Phosphorylation of the inactivating CDC2 Tyr15-site demonstrated to be up-regulated in EfnB1 stimulated THP-1 cells, which might be initiated by the increased total CDC2 protein levels that we found (Fig. 1D). CyclinB1 displays enhanced protein expression in EfnB1 treated THP-1 cells. Moreover, quantitative RT-PCR analysis showed that the expression of cell cycle inhibitor p21 is significantly induced by 3-fold in EfnB1 stimulated THP-1 cells, via increasing levels of p53 (Fig. 1E, both P = <0.001).
To verify whether the EfnB1 induced cell cycle arrest is EphB1 specific, we enforced EphB1 expression in HL60 EphB1int and MOLM13 EphB1low AML cells by introducing a GFP fused EphB1 overexpression construct. EphB1 overexpression increased EphB1 protein expression levels sufficiently in both AML cell lines, as confirmed by flowcytometric analysis and immunoblots. Exogenous EfnB1 stimulation further increased the apoptosis in EphB1 overexpressing cells in both AML cell lines (Fig. 1F). Again, we found increasing levels of phospho-CDC2Tyr15 and CyclinB1 by immunoblots.
From this study, we conclude that AML cells with high EphB1 expression can be forced into a cell cycle arrest upon ligand binding in vitro, while AML cells lacking EphB1 expression have a proliferative and anti-apoptotic survival advantage. The clinical significance and exploitation of EphB1 induced cell cycle arrest in AML will be analyzed in the near future.
EfnB1 induced activation of the EphB1 in AML cell lines promotes cell cycle arrest and apoptosis
EfnB1 induced activation of the EphB1 in AML cell lines promotes cell cycle arrest and apoptosis
(A) Absolute cell counts represent the growth inhibitory effects of EfnB1 ligand stimulation in AML cell lines THP-1 and HL60. (B) Flowcytometric Annexin V/PI apoptosis assay displayed the induction of apoptosis as a result of EfnB1 ligand stimulation in THP-1 and MOLM-13 AML cells. (C) MGG stained cytospins of EfnB1 treated THP-1 cells promotes the induction of multinucleated cells due to cell cycle arrest. (D) Immunoblots showed an enhanced apoptotic BAX/BCL2 ratio, in synergy with an upregulation of cell cycle inactivating checkpoint kinase CDC2Tyr15 upon EfnB1 stimulation in THP-1. (E) qRT-PCR confirms cell cycle inhibition by a 3-fold upregulation of p21 and a 1.5-fold induced expression of p53 in THP-1 EfnB1 stimulated cells. (F) The flowcytometric Annexin V/PI apoptosis assay showed that EfnB1 ligand induced apoptosis is even further induced in EphB1 overexpressing HL60 and MOLM-13 cells.
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal