Key Points
The fraction of invariant NKT cells demonstrating activation is increased during painful crises compared with steady state.
Regadenoson, an adenosine A2A receptor agonist, decreases the fraction of activated invariant NKT cells during painful crises.
Abstract
Adenosine A2A receptor (A2AR) agonists reduce invariant natural killer T (iNKT) cell activation and decrease inflammation in sickle cell disease (SCD) mice. We conducted a phase 1 trial of the A2AR agonist regadenoson in adults with SCD. The target dose was 1.44 μg/kg/h. iNKT cell activation was evaluated using antibodies targeting the p65 subunit of nuclear factor-κB (phospho-NF-κB p65), interferon-γ (IFN-γ), and A2AR. Regadenoson was administered to 27 adults with SCD. We examined 21 patients at steady state and 6 during painful vaso-occlusive crises (pVOC). iNKT cell activation was also measured in 14 African-American controls. During pVOC, the fraction of iNKT cells demonstrating increased phospho-NF-κB p65 and A2AR expression was significantly higher compared with controls (P < .01) and steady-state patients (P < .05). IFN-γ expression was also significantly higher compared with controls (P = .02). After a 24-hour infusion of regadenoson during pVOC, phospho-NF-κB p65 activation in iNKT cells decreased compared to baseline by a median of 48% (P = .03) to levels similar to controls and steady-state SCD. No toxicities were identified. Infusional regadenoson administered to adults with SCD at 1.44 μg/kg/h during pVOC decreases activation of iNKT cells without toxicity. This trial was registered at www.clinicaltrials.gov as #NCT01085201.
Introduction
Invariant natural killer T (iNKT) cells have been shown to be a significant contributor to the inflammation that promotes and sustains sickle cell vaso-occlusion, and blockade of iNKT cell activation or depletion of iNKT cells decreases pulmonary inflammation and injury in a murine model of sickle cell disease (SCD).1,2 In patients with SCD, iNKT cells are both increased in number and activation compared with healthy controls.1-3 These findings suggest that iNKT cells orchestrate an inflammatory cascade in SCD. Engagement of adenosine A2A receptors (A2AR) expressed on iNKT cells is a promising strategy for inhibiting their activation.2,4
Regadenoson is a selective A2AR agonist that is Food and Drug Administration approved for use during myocardial imaging.5 Administered as a bolus dose of 400 µg over 10 seconds for cardiac stress imaging, regadenoson binds to A2ARs on coronary artery smooth muscle cells to induce myocardial hyperemia for 2 to 4 minutes before coronary blood flow begins to return to normal.5 The vasodilation produced by regadenoson may cause a decrease in blood pressure and reflex tachycardia. Although such cardiovascular toxicities would be a concern in patients with SCD, the anti-inflammatory actions of A2ARs occur at an agonist concentration that is 10- to 100-fold lower than the cardiovascular actions, probably owing to a higher expression of A2ARs that are induced on activation of iNKT cells compared with vascular cells (G.L., J.J.F., M.M.O., E.M., J.K., O.O., A.R., F.C., D.N., D.G.N., and J.L., manuscript submitted March 2013).2 Increased expression of A2ARs on iNKT cells may also explain the potent anti-inflammatory effects of A2AR agonists in SCD mice (G.L., J.J.F., M.M.O., E.M., J.K., O.O., A.R., F.C., D.N., D.G.N., and J.L., manuscript submitted March 2013). Thus, a low-dose infusion of regadenoson has the potential to maintain a drug concentration that allows for a decrease in inflammation while avoiding cardiovascular side effects.
Based on the potent anti-inflammatory activity of A2AR agonists when given by infusion in murine models,2,4 we elected to administer regadenoson as a low-dose continuous infusion during painful vaso-occlusive crises (pVOC). Our goal was to reduce iNKT cell activation during pVOC without affecting cardiovascular function. Before administering regadenoson to patients with SCD during pVOC, we evaluated patients at steady state to identify a safe dose and duration of infusional regadenoson. We examined the activation status of circulating iNKT cells and CD3+ T cells before, during, and after infusion of the A2AR agonist regadenoson in 21 adults with SCD at steady state and in 6 who were admitted to the hospital for pVOC. To identify markers of activation in these cells that may be rapidly responsive to A2AR activation, we analyzed the p65 subunit of nuclear factor-κB (phospho-NF-κB p65) and interferon-γ (IFN-γ), along with A2AR expression.
Methods
Study conduct
A phase 1 dose-seeking and safety trial of regadenoson was conducted in adults with hemoglobin SS/hemoglobin S beta-thalassemia, and the effects of A2AR activation on iNKT cells during pVOC were examined (see supplemental Materials on the Blood website for details of the trial).3 Samples of peripheral blood iNKT cells were also obtained from 14 adult African-American controls (see supplemental Materials for control inclusion/exclusion criteria). The trial was performed at 5 collaborating hospitals, and blood samples were shipped to a central laboratory in La Jolla, CA, at 4°C to 6°C for the performance of flow cytometry. Institutional review boards approved the study protocol at participating sites, and informed consent was obtained from all participants in accordance with the Declaration of Helsinki.
Dose selection
To identify a safe and biologically effective dose of regadenoson, a 3-stage study was designed, with dose escalation confined to the 12-hour infusion in stage 1. The objective of the study was to examine the effects of A2AR activation on iNKT cells using the maximum dose of regadenoson that would be tolerated by ≥5 of 6 individuals for 24 hours during a pVOC (see supplemental Materials for definitions of steady state and pVOC). Dose-limiting toxicity (DLT) was defined as a sustained change in systolic blood pressure ≥30 mm Hg or a measurement ≤75 mm Hg, a sustained change in heart rate ≥30 beats per minute (bpm), or a measurement ≤40 bpm (see supplemental Materials for complete DLT criteria).
Stage 1. Stage 1 was a 3 + 3 dose escalation study to establish the dose of regadenoson that can be administered safely over a 12-hour infusion in adults with SCD who were at steady state. Three doses were tested: 0.24 (dose level 0), 0.6 (dose level 1), and 1.44 μg/kg/h (dose level 2) (see supplemental Materials for a discussion of the rationale for dose selection). Dose escalation was continued until either a maximum tolerated dose was identified or until 6 participants had been treated at the maximum planned dose (dose level 2).
Stage 2. Stage 2 examined the safety of regadenoson as a 24-hour infusion in 6 adults at steady state using the target dose that had been determined in stage 1.
Stage 3. Stage 3 examined the safety of regadenoson as a 24-hour infusion at the target dose in 6 adults during pVOC. Standard measures for pVOC therapy including, but not limited to, hydration, opioid therapy, and blood transfusions were administered at the discretion of the treating physician.
Laboratory methodology
iNKT cells were identified by flow cytometry as live, CD19− (SJ25-C1; Invitrogen), CD3+ (UCHT1; Invitrogen and BD), and Vα24-Jα18 T-cell receptor (TCR)+ (6B11; eBioscience) cells. Conventional T cells were identified as live, CD19−, CD3+, and Vα24-Jα18 TCR− cells. The active phosphorylated form of p65-NF-κB transcriptional factor was identified with anti–phospho-NF-κB p65 (Ser536, 93H1; Cell Signaling) antibody, and the human adenosine A2AR receptor was detected with a monoclonal anti-human adenosine A2AR antibody that detects the intracellular facing third intracellular loop.6 IFN-γ was detected with an anti-human IFN-γ (4S.B3; eBioscience3) antibody (see supplemental Materials for details of leukocyte immunostaining).
To identify iNKT cells that demonstrated increased activation on flow cytometry, we defined activation gates relative to the expression levels of phospho-NF-κB p65, A2AR, and IFN-γ in conventional CD3+ T cells. The lower boundaries of activation gates were set to exclude expression levels of 95% of conventional CD3+ T cells. The data were expressed as the percentage of iNKT cells in the activation gate and were reproducible when repeated on different days in steady-state patients.
Liquid chromatography-mass spectrometry/mass spectrometry was used to measure plasma levels of regadenoson (Advinus Therapeutics) (see supplemental Materials for pharmacokinetic methodology).
Statistical analysis
Laboratory results were analyzed with robust methods (median and range). Comparisons of iNKT cell activation parameters between controls and patients at steady state and during pVOC were performed. Direction of differences was hypothesized a priori, and 1-sided Wilcoxon rank sum (nonpaired) and Wilcoxon sign rank (paired) tests were used. Analysis of iNKT cell activation markers before and after regadenoson infusion were reported as percent change from the value at time 0 to the value at 24 hours from the start of the infusion ([% time 0 − % time 24 hours]/% time 0).
Results
Study participants
Twenty-seven adults with hemoglobin SS from the 5 participating centers were administered regadenoson (Table 1). To compare activation levels of iNKT cells and A2AR expression, 14 healthy African-American controls were also recruited. The mean age of the control group was similar to steady-state patients (31 vs 36 years); however, there was a lower percentage of men in the control group (36% vs 67%). Study enrollment began in June 2010 and ended in August 2012.
Subject demographics, SCD characteristics, and descriptors of pVOC characteristics in stage 3
| . | Cohort (n = 27) . | Cohort by stage . | ||
|---|---|---|---|---|
| Stage 1 (n = 15) . | Stage 2 (n = 6) . | Stage 3 (n = 6) . | ||
| Demographics | ||||
| Age at infusion (y), median (IQR) | 33.5 (12) | 32.2 (11) | 35.8 (11) | 33.3 (11) |
| Gender, % male | 56 | 53 | 67 | 50 |
| SCD characteristics | ||||
| Hemogloblin (g/dL), median (IQR) | 7.6 (2) | 7.7 (2) | 7.5 (1) | 6.1 (2) |
| WBC (k/μL), median (IQR) | 8.4 (4) | 8.1 (2) | 11.6 (5) | 9.8 (3) |
| Hydroxyurea, % | 59 | 40 | 83 | 83 |
| pVOC admission characteristics | ||||
| Drug start time (d), median (IQR)* | NA | NA | NA | 3.5 (2) |
| Duration of stay (d), median (IQR) | NA | NA | NA | 6 (2) |
| Postdrug admittance in 4 wk, median (range) | 0 (0-2) | 0 (0) | 0 (0-2) | 0 (0-2) |
| . | Cohort (n = 27) . | Cohort by stage . | ||
|---|---|---|---|---|
| Stage 1 (n = 15) . | Stage 2 (n = 6) . | Stage 3 (n = 6) . | ||
| Demographics | ||||
| Age at infusion (y), median (IQR) | 33.5 (12) | 32.2 (11) | 35.8 (11) | 33.3 (11) |
| Gender, % male | 56 | 53 | 67 | 50 |
| SCD characteristics | ||||
| Hemogloblin (g/dL), median (IQR) | 7.6 (2) | 7.7 (2) | 7.5 (1) | 6.1 (2) |
| WBC (k/μL), median (IQR) | 8.4 (4) | 8.1 (2) | 11.6 (5) | 9.8 (3) |
| Hydroxyurea, % | 59 | 40 | 83 | 83 |
| pVOC admission characteristics | ||||
| Drug start time (d), median (IQR)* | NA | NA | NA | 3.5 (2) |
| Duration of stay (d), median (IQR) | NA | NA | NA | 6 (2) |
| Postdrug admittance in 4 wk, median (range) | 0 (0-2) | 0 (0) | 0 (0-2) | 0 (0-2) |
Stage 1, 12-hour infusion at steady state, dose-seeking; stage 2, 24-hour infusion at steady state, target dose (1.44 μg/kg/h); stage 3, 24-hour infusion during pVOC, target dose (1.44 μg/kg/h). IQR, interquartile range; NA, not applicable.
Days since hospital admission (time starts on admittance order).
iNKT cells demonstrate activation during pVOC that is decreased by regadenoson
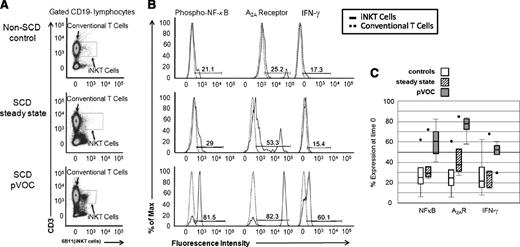
Analysis of circulating iNKT cells from patients with SCD during pVOC revealed an elevated percentage of iNKT cells that exhibited increased expression of phospho-NF-κB p65, indicative of enhanced iNKT cell activation compared with African-American controls (25% vs 61%, P ≤ .01) and steady-state patients (29% vs 61%, P = .02). IFN-γ expression during pVOC also differed significantly from controls (25% vs 52%, P = .02) and trended to higher levels compared with steady-state SCD (27% vs 52%, P = .06). A2AR expression on iNKT cells was also increased during pVOC compared with controls (26% vs 77%, P < .01) and steady-state patients (38% vs 77%, P = .03; Figure 1A-C).
Phospho-NF-κB p65, A2AR, and IFN-γ expression in iNKT cells of controls and patients in steady state and pVOC (stages 2 and 3, respectively) at time 0 (preinfusion). (A) Flow cytometry plot of iNKT cells from controls and patients in steady state and pVOC at time 0. (B) Phospho-NF-κB p65, A2AR, and IFN-γ expression in iNKT cells compared with CD3+ non-iNKT cells at time 0. Solid peaks represent expression in iNKT cells and dashed peaks represent expression in CD3+ non-iNKT cells. Activation gate is defined as increased expression relative to CD3+ non-iNKT cells. Percent of iNKT cells in the activation gate is reported. (C) iNKT cell inflammatory marker data at time 0 for controls (white boxes), patients in steady state (stage 2, hatched boxes), and pVOC (stage 3, dark gray boxes). Lower end of box denotes 25% of data, upper end of box denotes 75% of data, and median is the line within the box. Error bars are the lowest and highest data points within 1.5 times the interquartile range, and circles represent outliers.
Phospho-NF-κB p65, A2AR, and IFN-γ expression in iNKT cells of controls and patients in steady state and pVOC (stages 2 and 3, respectively) at time 0 (preinfusion). (A) Flow cytometry plot of iNKT cells from controls and patients in steady state and pVOC at time 0. (B) Phospho-NF-κB p65, A2AR, and IFN-γ expression in iNKT cells compared with CD3+ non-iNKT cells at time 0. Solid peaks represent expression in iNKT cells and dashed peaks represent expression in CD3+ non-iNKT cells. Activation gate is defined as increased expression relative to CD3+ non-iNKT cells. Percent of iNKT cells in the activation gate is reported. (C) iNKT cell inflammatory marker data at time 0 for controls (white boxes), patients in steady state (stage 2, hatched boxes), and pVOC (stage 3, dark gray boxes). Lower end of box denotes 25% of data, upper end of box denotes 75% of data, and median is the line within the box. Error bars are the lowest and highest data points within 1.5 times the interquartile range, and circles represent outliers.
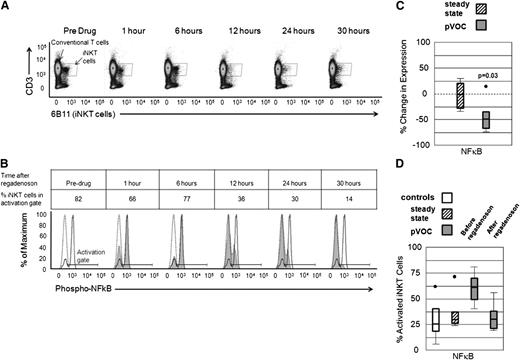
Within the group of patients examined during pVOC, the expression of phospho-NF-κB p65 in iNKT cells (as a percentage of the total iNKT cells in the activation gate) was higher compared with conventional CD3+ non-iNKT cells (Figure 2A-B). Following a 24-hour infusion of the A2AR agonist regadenoson to patients during pVOC (stage 3, 1.44 μg/kg/h), phospho-NF-κB p65 expression in iNKT cells decreased by a median of 48% when pre- and postdrug percentages of activated iNKT cells values were compared (P = .03; Figure 2C). Median absolute numbers of activated iNKT cells per milliliter before and after regadenoson were 3.4 × 103 and 4.5 × 102, respectively, for a median decrease of 1.8 × 103. The infusion of regadenoson during pVOC reduced the phospho-NF-κB p65 expression (measured as percentage of iNKT cells) in iNKT cells to similar levels observed in African-American controls and steady-state patients (Figure 2D). Expression of IFN-γ and A2AR in iNKT cells also decreased after administration of regadenoson by 25% and 23%, respectively; however, these changes did not achieve statistical significance.
Phospho-NF-κB p65 expression in iNKT cells in subjects who received a 1.44 μg/kg/h regadenoson infusion for 24 hours with a 6-hour observation period when no drug was infused. (A-B) Phospho-NF-κB p65 expression in iNKT cells in a participant examined during pVOC (stage 3). (A) Flow cytometry and iNKT cell gates in serial blood samples taken at the indicated times. Regadenoson was infused starting at time 0 and ending at 24 hours. (B) Phospho-NF-κB p65 expression in iNKT cells compared with CD3+ non-iNKT cells from a pVOC patient. Solid peak represents phospho-NF-κB p65 in iNKT cells before regadenoson. Dashed peak represents phospho-NFκB expression in CD3+ non-iNKT cells, and the shaded area is phospho-NF-κB p65 expression in iNKT at time points after regadenoson infusion. NF-κB activation gate is defined as increased expression of phospho-NF-κB p65 relative to CD3+ non-iNKT cells. Percent iNKT cells in the activation gate at time points after regadenoson infusion is reported. (C) Percent change in iNKT cell phospho-NF-κB p65 before and after a 24-hour infusion of regadenoson in patients in steady state (stage 2, hatched box) and pVOC (stage 3, dark gray box). (D) Phospho-NF-κB p65 expression in iNKT cells from African-American controls (white box), steady-state patients (stage 2, hatched box), and pVOC participants before and immediately after a 24-hour regadenoson infusion (stage 3, dark gray boxes).
Phospho-NF-κB p65 expression in iNKT cells in subjects who received a 1.44 μg/kg/h regadenoson infusion for 24 hours with a 6-hour observation period when no drug was infused. (A-B) Phospho-NF-κB p65 expression in iNKT cells in a participant examined during pVOC (stage 3). (A) Flow cytometry and iNKT cell gates in serial blood samples taken at the indicated times. Regadenoson was infused starting at time 0 and ending at 24 hours. (B) Phospho-NF-κB p65 expression in iNKT cells compared with CD3+ non-iNKT cells from a pVOC patient. Solid peak represents phospho-NF-κB p65 in iNKT cells before regadenoson. Dashed peak represents phospho-NFκB expression in CD3+ non-iNKT cells, and the shaded area is phospho-NF-κB p65 expression in iNKT at time points after regadenoson infusion. NF-κB activation gate is defined as increased expression of phospho-NF-κB p65 relative to CD3+ non-iNKT cells. Percent iNKT cells in the activation gate at time points after regadenoson infusion is reported. (C) Percent change in iNKT cell phospho-NF-κB p65 before and after a 24-hour infusion of regadenoson in patients in steady state (stage 2, hatched box) and pVOC (stage 3, dark gray box). (D) Phospho-NF-κB p65 expression in iNKT cells from African-American controls (white box), steady-state patients (stage 2, hatched box), and pVOC participants before and immediately after a 24-hour regadenoson infusion (stage 3, dark gray boxes).
Pharmacokinetics of infusional regadenoson
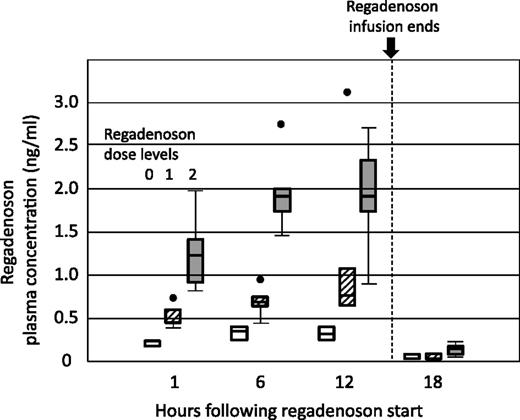
Increasing dose levels of regadenoson achieved commensurate higher plasma levels of regadenoson during the 12-hour infusion in stage 1 (Figure 3). Median peak plasma at 1.44 μg/kg/h was 1.9 ng/mL (range 0.77-2.74 ng/mL).
Pharmacokinetics of regadenoson infused for 12 hours during stage 1. Plasma concentrations of regadenoson were measured at 0, 1, 6, 12, and 18 hours. Dose level 0 was 0.24 μg/kg/h (white boxes), dose level 1 was 0.60 μg/kg/h (hatched boxes), and dose level 2 was 1.44 μg/kg/h (dark gray boxes).
Pharmacokinetics of regadenoson infused for 12 hours during stage 1. Plasma concentrations of regadenoson were measured at 0, 1, 6, 12, and 18 hours. Dose level 0 was 0.24 μg/kg/h (white boxes), dose level 1 was 0.60 μg/kg/h (hatched boxes), and dose level 2 was 1.44 μg/kg/h (dark gray boxes).
Infusional regadenoson at the target dose is safe in adults in SCD
Infusional regadenoson at 0.24, 0.60, and 1.44 μg/kg/h was safe in adults with SCD. One putative DLT was observed in a baseline participant at dose level 1. The study participant had a brief episode of bradycardia (heart rate = 49 bpm) without a decrease in systolic blood pressure after the infusion was completed and while sleeping. At that point in the study, the threshold for a DLT was <50 bpm. Per recommendations of the study cardiologist and data safety monitoring board, the threshold was thereafter changed to 40 bpm to avoid capturing normal physiologic variation. No other DLTs were observed in study participants at any dose level. In particular, heart rate and blood pressure were relatively stable throughout the regadenoson infusion, reflecting normal physiologic variation (see supplemental Figure).
Discussion
This study demonstrates that during pVOC, iNKT cells increase numerically in the blood, become more activated, and express higher levels of A2ARs compared with controls and steady-state adults with SCD. The increased iNKT cell activation during pVOC can be counteracted by a low-dose infusion of the A2AR agonist regadenoson, reducing iNKT cell activation to levels comparable to that observed in controls and steady-state patients.
To identify the activation state of iNKT cells and CD3+ T cells, we first determined the optimal indicator of changes in activation during 12- and 24-hour regadenoson infusions. In other studies, we discovered that phospho-NF-κB p65 expression in iNKT cells provides the most reliable index of rapid changes in iNKT cell activation in response to regadenoson (G.L., J.J.F., M.M.O., E.M., J.K., O.O., A.R., F.C., D.N., D.G.N., and J.L., manuscript submitted March 2013). Based on these results, we focused on phospho-NF-κB p65 expression in the current study to determine iNKT cell activation status in different disease states (steady state, pVOC) and before and after regadenoson infusion. We found that phospho-NF-κB p65 expression in iNKT cells increased in patients during pVOC vs steady state and that this increased phospho-NF-κB p65 expression in iNKT cells could be significantly reduced when regadenoson was infused at the target dose of 1.44 μg/kg/h. Notably, non-iNKT CD3+ T cells were not activated in patients at steady state or during pVOC. This decrease in iNKT cell phospho-NF-κB p65 expression occurred at nanomolar-range plasma levels of regadenoson and was attained without tachycardia or hypotension, consistent with the target dose (1.44 μg/kg/h) being within the therapeutic window. Our study extends the findings in SCD mouse models implicating iNKT cell activation in vaso-occlusion1,2 and demonstrates that infusion of regadenoson targets this critical cell type and is safe.
iNKT cells represent a small subset of T cells that influence both innate and adaptive immunity.7 Similar to the adaptive immune response of conventional T cells, iNKT cell activation requires antigen presentation to a TCR. However, TCRs expressed on iNKT cells are invariant (Vα24-Jα18-Vβ11 in humans) and recognize glycolipid antigens, not unlike the pattern recognition of innate immunity, that are presentation is restricted to antigen presenting cells expressing the major histocompatibility complex 1–like CD1d receptor.7,8 In patients with SCD, glycolipids generated from the tissue damage that occurs during the process of vaso-occlusion are likely activating iNKT cells, although certain host CD1d-restricted lipid antigens can be synthesized by macrophages.8 Following activation, iNKT cells rapidly secrete large amounts of cytokines and propagate a generalized leukocyte inflammatory response via IFN-γ–inducible chemokines and the activation and recruitment of T cells, natural killer cells, neutrophils, and macrophages.9,10 Owing to their rapid activation and stimulation of a broader inflammatory response, iNKT cells are thought to be important instigators of inflammation despite representing only 0.5% of peripheral blood T cells in healthy subjects.11 In a prior study of steady-state patients with SCD, the number of circulating iNKT cells was ∼5 times higher than healthy controls, and iNKT cells were more likely to be activated.1 Data from this study and another from our group demonstrate that during a pVOC, there is a further increase in the number and activation of iNKT cells (G.L., J.J.F., M.M.O., E.M., J.K., O.O., A.R., F.C., D.N., D.G.N., and J.L., manuscript submitted March 2013). Together with the SCD murine data implicating iNKT cells in the process of vaso-occlusion,1,2 these data provide further evidence for the role of iNKT cells in SCD morbidity. Decreasing iNKT cell activation in patients with SCD has the potential to dampen the severity of pVOC by interrupting the vicious cycle of vaso-occlusion, tissue injury/inflammation, and further vaso-occlusion that characterizes the pathogenesis of pVOC. Although infection is a concern when blocking iNKT cell activation, the approach used in this study minimizes the risk because of the short duration of treatment (24 hours) and half-life of regadenoson (∼5 minutes).
In the current study of patients with SCD, we discovered that phospho-NF-κB p65 is highly responsive to A2AR activation on iNKT cells, rapidly reflecting changes in iNKT cell activation status. NF-κB is a transcription factor that promotes inflammation in addition to having roles in cell survival and cell death.12 Signaling through the NF-κB pathway is crucial for iNKT cell proliferation and cytokine production.13 Pertinent to this study, NF-κB can be down-regulated following A2AR activation in a protein kinase A–dependent mechanism.14,15 Inhibition of NF-κB decreases the transcription of proinflammatory genes and thereby mediates the anti-inflammatory response to A2AR activation. In contrast to the transcriptional regulation of cytokines (IFN-γ), protein kinase A signaling occurs upstream, and its actions can be rapidly inhibited through phosphorylation and degradation. Potentially, this rapid signaling cascade allows phospho-NF-κB p65 to be a dynamic marker that is very responsive to the effects of regadenoson on iNKT cells. In our study, the greater decreases in iNKT cell phospho-NF-κB p65 expression following regadenoson compared with the more traditional lymphocyte activation marker IFN-γ may reflect this rapid down-regulation of NF-κB on A2AR activation. Infusion of regadenoson for a longer time might have induced a more robust response from IFN-γ in iNKT cells, because it is transcriptionally regulated.
We found an increased percentage of activated iNKT cells during pVOC compared with steady state, adding to the abundant evidence that already exists from studies in the NY1DD mouse model of SCD and patients with SCD implicating iNKT cells in the pathogenesis of vaso-occlusion. Prior studies in patients with SCD have demonstrated increased activation of neutrophils16,17 and monocytes,18,19 in addition to iNKT cells, and increased levels of numerous cytokines when pVOC and steady state are compared.20,21 These previous studies show that inflammation in vaso-occlusion is multicellular in origin and not confined to a single signaling pathway, presenting a challenge when identifying therapeutic interventions.22,23 iNKT cell activation, however, is an early event in immune response and may incite this cascade of multicellular activation and cytokine production that leads to the widespread inflammation in vaso-occlusion.
Adenosine signaling through the A2B receptor has been associated with priapism and red cell sickling in murine models of SCD.24-26 Although these findings have not been confirmed in patients with SCD, the data underscore the importance of the selectivity of regadenoson for the A2AR. In the current study, regadenoson produced A2AR-mediated anti-inflammatory effects in the absence of potential toxicities such as bradycardia and bronchospasm that could have been induced through A1 and A2B receptor signaling, respectively. These data support the notion that regadenoson is selective for A2AR at the plasma levels achieved in our study.
Limitations were present in this study. The key finding that regadenoson decreased activation of iNKT cells is based on sampling the circulating iNKT cell population. On activation, iNKT cells are recruited to sites of inflammation and marginate to the walls of blood vessels or emigrate out of the vasculature and into the surrounding tissues. Because activated iNKT cells migrate out of the vasculature, our results may underestimate the degree of activation and potentially the response of iNKT cells to regadenoson. The highest dose that we examined was not the maximally tolerated dose because no toxicities attributable to the drug occurred during the study. However, we anticipate that because of >100-fold upregulation of A2ARs on iNKT cells, a 2 ng/mL plasma concentration of regadenoson is sufficient for maximal effects of A2AR activation (G.L., J.J.F., M.M.O., E.M., J.K., O.O., A.R., F.C., D.N., D.G.N., and J.L., manuscript submitted March 2013). In fact, the target dose of 1.44 μg/kg/h decreased iNKT cell activation to levels similar to healthy controls. An alternative explanation for the effect of regadenoson on iNKT cell activation during pVOC is that the infusions were coincident with resolving crises and decreasing inflammation. However, in data presented separately, decreases in iNKT cell activation also occurred during dose-seeking stage 1 when regadenoson was administered to steady-state patients, suggesting that the observed reduction in iNKT cell activation during pVOC was caused by the actions of regadenoson as opposed to improving vaso-occlusion (G.L., J.J.F., M.M.O., E.M., J.K., O.O., A.R., F.C., D.N., D.G.N., and J.L., manuscript submitted March 2013). Finally, this study was not designed to determine whether the decrease in activation of iNKT cells associated with regadenoson infusion produces a clinical benefit. Design considerations that will be important to determine the clinical efficacy of regadenoson in a subsequent phase 2 trial, such as a requirement to initiate the drug early in the course of pVOC and a common approach among sites to the management of pVOC, were not incorporated into the phase 1 study because the primary outcome measure was safety. To definitely examine the effect of regadenoson on iNKT cell activation during pVOC and elucidate whether low-dose infusional regadenoson decreases the severity of pVOC or acute chest syndrome, a larger placebo-controlled study is needed.
In summary, low-dose infusional regadenoson administered at a rate of 1.44 μg/kg/h over 24 hours during pVOC decreased the activation of iNKT cells as measured by phospho-NF-κB p65 expression in adults with SCD without causing cardiovascular toxicities. The next step in the evaluation of low-dose infusional regadenoson for the treatment of pVOC and acute chest syndrome is a randomized-controlled phase 2 trial.
The online version of this article contains a data supplement.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 USC section 1734.
Acknowledgments
The authors thank Jillian Dargatz, Lisa Garrett, and Hillary Chu, research coordinators who contributed significantly to this study, and Annette Dalton, who was responsible for data management.
This study was supported by National Heart, Lung, and Blood Institute grants RC2HL101367 and R34HL108757 and a grant from Astellas Pharma (D.G.N.). The authors also acknowledge the support provided by Clinical and Translational Science Awards at all participating institutions.
Authorship
Contribution: J.J.F. designed and conducted the study, analyzed data, enrolled patients, and wrote the manuscript; G.L. performed laboratory measurements, analyzed data, and reviewed the manuscript; M.M.O. enrolled patients and reviewed the manuscript; E.M. and J.K. enrolled patients and reviewed the manuscript; O.O. enrolled patients and reviewed the manuscript; A.R. coordinated the study; F.C. analyzed data; D.N. designed and conducted the study, analyzed data, and reviewed the manuscript; J.L. designed and conducted the study, oversaw laboratory measurements, analyzed data, and reviewed the manuscript; and D.G.N. designed and conducted the study, analyzed data, and wrote the manuscript.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: Joshua J. Field, BloodCenter of Wisconsin, 8733 Watertown Plank Road, Milwaukee, WI 53226; e-mail: joshua.field@bcw.edu.
References
Author notes
J.J.F. and G.L. are co-first authors.
J.L. and D.G.N. are co-senior authors.