Abstract
Cell signaling mediated by the JAK2-STAT (Janus kinase 2-signal transducer and activator of transcription) pathway plays a critical role in hematopoiesis, and its aberrant activation is associated with the progression of hematological malignancies. For instance, somatic mutations in the JAK2 gene that lead to constitutive activation of STATs are involved in the pathogenesis of myeloproliferative neoplasms (MPN) and refractory anemia with ringed sideroblasts with thrombocytosis, one of the myelodysplastic/myeloproliferative neoplasms, unclassifiable (MDS/MPN, U). In addition, insufficient inactivation of the JAK-STAT pathway due to repression of SOCS-1 (suppressor of cytokine signaling 1) through hypermethylation of the SOCS-1 gene is involved in the disease progression of high-risk myelodysplastic syndromes (MDS) and acute myeloid leukemia (AML) (Brakensiek et al.; Br. J. Haematol. 2005;130:209). These observations prompted us to investigate the effect of the JAK2-selective inhibitor NS-018, which is in an early-phase clinical trial for MPN, on the colony formation by bone marrow hematopoietic progenitor cells from high-risk de novo MDS patients and the phosphorylation status of STAT3 in these cells.
Bone marrow mononuclear cells (BMMNCs) from six MDS patients and three healthy volunteers were collected with informed consent in accordance with the Declaration of Helsinki and with the approval of the Institutional Review Board. The MDS subtypes of the six patients included two RCMD, three RAEB-1 and one RAEB-2 according to the WHO classification. All MDS patients had complex cytogenetic abnormalities and their prognostic risks were defined to be high (N=1) or very high (N=5) in the IPSS-R. Commercially available normal human CD34+ BM cells were also examined for comparison. Cells were incubated in methylcellulose medium containing cytokines with or without NS-018. Burst forming unit-erythroid (BFU-E) and colony forming unit-granulocyte/macrophage (CFU-GM) were scored on day 14 of culture. Colony-forming cells were then collected and subjected to western blotting analysis.
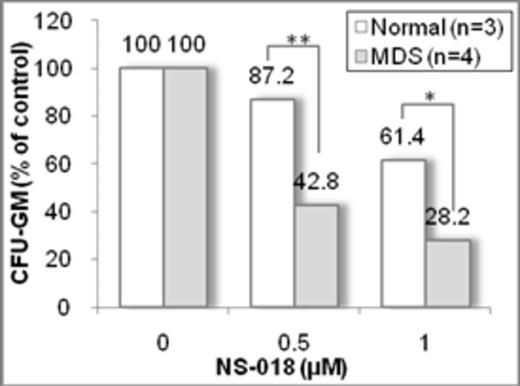
We first examined the colony-formation capacity of MDS-derived BMMNCs. CFU-GM colony formation was observed in four of six MDS samples, although the absolute CFU-GM numbers from MDS BMMNCs were less than those from normal BMMNCs and normal CD34+BM cells. MDS-derived BMMNCs produced larger numbers of CFU-GM colonies (78–99% of total colonies) than BFU-E, similar to the findings usually observed in AML, whereas normal BMMNCs and normal CD34+BM cells formed virtually the same numbers of CFU-GM colonies and BFU-E colonies. These results suggest that BMMNCs from four of the MDS patients enrolled in this study had characteristics similar to AML BMMNCs, although they showed less proliferation than AML BMMNCs. We next examined the effect of NS-018 on CFU-GM colony formation. NS-018 treatment decreased the numbers of CFU-GM colonies from MDS-derived BMMNCs in a dose-dependent manner, and this effect was significantly more potent against MDS-derived than against normal cells (57.2% inhibition in MDS cells vs. 12.8% inhibition in normal cells at 0.5 mM NS-018; see Figure). These results indicate that NS-018 is preferentially efficacious in inhibiting CFU-GM formation from BMMNCs from high risk MDS. In addition, the level of phospho-STAT3 in MDS-derived colony-forming cells was twice as high as in colony-forming cells from normal BMMNCs, and 1.0 mM NS-018 completely suppressed the phosphorylation of STAT3 in CFU-GM colony-forming cells from MDS.
Our results show for the first time that a JAK2 inhibitor, NS-018, potently suppresses the formation of MDS colonies. NS-018 could be a new therapeutic option for high-risk MDS patients.
Kodama:Nippon Shinyaku: Employment. Naito:Nippon Shinyaku: Employment. Nakaya:Nippon Shinyaku: Employment. Taniwaki:Novartis: Honoraria.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal