Abstract
The highly evolutionarily conserved Hippo pathway (HP) regulates proliferation, apoptosis polarity and stem cell maintenance. In Humans, TAZ (transcriptional co-activator with PDZ-binding motif) and YAP (Yes associated protein) are the functional effectors, regulating gene expression by co-activating several transcription factors (RUNX, TEADS, SMADS). Phosphorylation inactivates both, causing expulsion from the nucleus and either proteosomal degradation or sequestration by 14-3-3 or membrane protein complexes. A 4 protein complex of LATS 1/2 kinase, MST1/2, MOB and SAV1, phosphorylates YAP and TAZ, and is regulated by many pathways including Neurofibromin 2 (NF2) , and WNT, and by cell-cell contact. YAP and TAZ share many binding and interaction partners, sharing overlapping functions, but they also have exclusive interactions leading to some exclusive functions, notably in SMAD family and TGFβ regulation. Deregulation of HP is associated with numerous solid tumors (breast, lung, colorectal, ovary, liver), with upregulation conferring an adverse prognosis, but mutations of HP components is rare, suggesting other mechanism for deregulation. The role of the HP pathway in AML is undefined and similar to most solid tumors, mutations in AML were not observed by TCGA.
To define the role of the HP in AML we used reverse phase protein array (RPPA) technology to print protein from leukemia enriched cells from 511 newly diagnosed AML patients. The RPPA was probed with 231 antibodies, including antibodies against both total and phosphorylated HP components NF2, YAP (p-Serine 127) and TAZ (p-Serine 89). We could not validate antibodies for LATS 1 or 2.
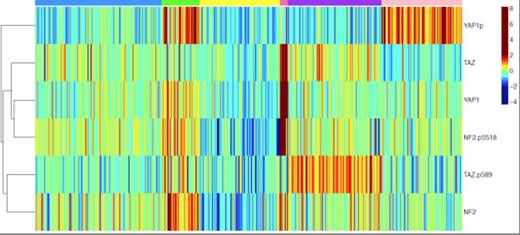
Levels of total NP2, TAZ YAP were often above that of normal CD34+ cells in 30%, 15% and 17% of cases. Levels of phosphorylated NF2 (p=0.07, 23%) and YAP (p=0.009, 10%) were often above, and those of pTAZ (p= 0.008, 29%) below that of normal CD34+ cells. Higher levels of pTAZ were associated with FAB MO and M1 subtypes, higher CD7 and CD34 expression and higher % marrow blasts. Higher levels of pYAP were associated with lower % marrow and peripheral blood blasts and with cases transformed from RAEBT and CMML. The optimum number of principal components for these 6 antibodies was two, depending on the phosphorylation state of YAP and TAZ. Another statistical method determined the optimal number of patient groups to be 6 (figure 1) with expression 1) Pan Off (19% n=79, Yellow in Fig.) 2) High Total (2%, n =8, Dark Pink.) with very high levels of NF2, TAZ and YAP and high pNF2, 3) Active HP (30% n=123, Blue), with moderate total levels but scant phosphorylation, 4) YAP inactive (19%, n=78, Light Pink) and 5) TAZ inactive (22.5%, n=93, Purple) where only one is phosphorylated and 6) Both inactivated (9% n=37, Green) with both heavily phosphorylated. The TAZ inactive motif was prognostically adverse for overall survival (OS) (Median 30 weeks p=0.009) compared to active, pYAP, both inactivated (median 48-60 weeks), while the High Total pattern had longer OS (median 224 wks). Notably, high pTAZ alone or with pYAP was associated with inferior response and OS in patients treated with demethylating and histone deacetylating drugs. Favorable cytogenetics patients with active HP, High pTAZ or both inactive, treated with fludarabine-HDAC had shorter OS and significantly shorter remission duration (52-70 weeks vs. not reached). High pTAZ was associated with higher SMAD family levels and with unfavorable cytogenetics as well as with higher levels of phosphorylated (inactivated) β-Catenin, likely due to cytoplasmic localization.
Expression of HP members. Colors across top refer to groups defined above. Color scale shows expression on a Log-2 scale.
Expression of HP members. Colors across top refer to groups defined above. Color scale shows expression on a Log-2 scale.
HP proteins are heterogeneously expressed in AML and key effectors YAP and TAZ are differentially phosphorylated. Differential phosphorylation of YAP and TAZ has not been previously reported. This suggests that active YAP in the presence of inactive TAZ is inducing expression of genes that provide proliferative or survival advantages to leukemic blasts. The observation that the Highly active HP group did very well may be identifying a high proliferative group that is more susceptible to cycle specific chemotherapy, similar to Burkitt's leukemia. In summary, this data suggest that the HP pathway is active in AML and has a previously undescribed pattern of separate phosphorylation of YAP and TAZ.
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal