Abstract
Among FVIII-treated hemophilia A (HA) patients, 25-30% develop FVIII antibodies that inhibit its pro-coagulant function. However, the etiology of FVIII inhibitor formation remains poorly understood, particularly the role played by myeloid innate immune cells. Myeloid-derived suppressor cells (MDSC – CD11b/Gr1 co-expressing, immunosuppressive myeloid cells found in peripheral blood, lymphoid tissue and bone marrow) are expanded in cancer and inhibit adaptive immune responses against tumors. MDSC also mediate organ transplant tolerance. Therefore, we investigated MDSC dynamics during the course of FVIII exposure in HA mice and the potential to harness MDSC as a novel means of mediating FVIII tolerance.
6 to 12 week-old F8-knockout Balb/c hemophilia A mice (HA mice) were used in accordance with Queen's University Animal Care Committee protocols. Over 3 independent experiments, HA mice were either untreated (Week 0; n = 4-6) or tail vein infused weekly for 2 or 4 weeks with 2 IU rhFVIII (Kogenate-FS for 2 experiments or Advate for one; n = 6 and 7, at 2 and 4 weeks) or 200 μl HBSS vehicle alone (n = 4 and 5). Alternatively HA mice were infused weekly with 2 pdFVIII (Wilate) for 2 (n = 2) or 4 weeks (n = 2) or subjected to a 4-day G-CSF preconditioning regimen during Week minus-1 (10 μg/day, SC, in 200 μl HBSS; n = 4) or HBSS alone (n = 4), followed by 2 or 4 weekly rFVIII infusions. Red cell-lysed blood, spleen and bone marrow suspensions were subjected to MDSC flow cytometry using anti-CD11b(Mac1)-APC and anti-Gr1-PE (Miltenyi Biotec). Week 2 and 4 plasma was subjected to anti-FVIII antibody ELISA and Bethesda assays. Means were compared using student's t-test.
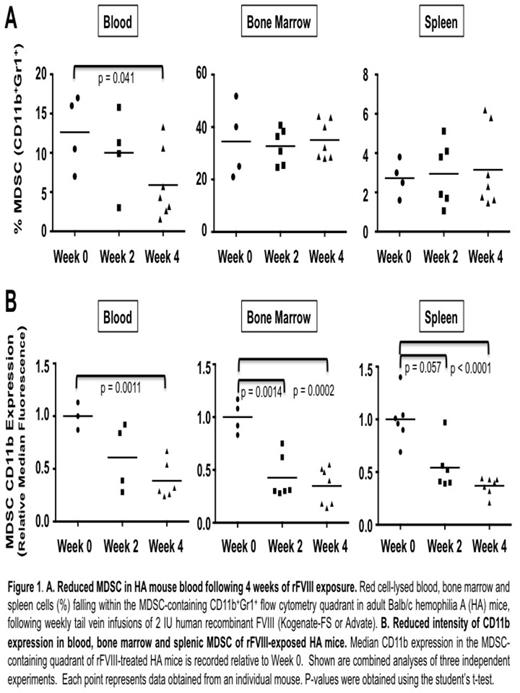
HA mice contained CD11b+Gr1+ MDSC in expected proportions in the blood, bone marrow and spleen. Peripheral blood MDSC proportions declined significantly from baseline following 4 weeks of rFVIII infusions (baseline mean 12.6%, versus 5.9% at 4 weeks, p = 0.041) (Figure 1A). More striking, however, was the observation of diminishing CD11b expression within all three MDSC compartments during the course of rFVIII exposure (magnitude of reduction: 46-65%; at 4 weeks in blood, p = 0.0011; at 2 and 4 weeks in bone marrow, p = 0.0014 and 0.0002; spleen, p = 0.057 and<0.0001) (Figure 1B). To our knowledge, diminished CD11b expression by MDSC has not been previously reported. Suggesting functional significance, 20-40% higher CD11b expression by week 4 MDSC in pdFVIII(Wilate)-treated HA mice (n = 2) was associated with 5 to 30-fold lower Bethesda unit inhibitors as compared to rFVIII. Further in vivo and in vitro studies are underway to confirm the functional impact of diminished MDSC CD11b expression. Next, in anticipation of future adoptive transfer experiments, we asked whether MDSC could be expanded in vitro from the BM of FVIII-na•ve HA mice. Using established 4-day MDSC culture conditions (GM-CSF + IL-6, Marigo et al., Immunity, 2010; or GM-CSF + G-CSF + day-3 IL-13, Highfill et al., Blood, 2010) we were able to achieve 7- to 10-fold expansion of MDSC over standard BM culture. Finally, in anticipation of future expansion of endogenous HA MDSC, we pre-treated a subset of mice with G-CSF (known to expand MDSC in vivo) (n = 4) versus HBSS vehicle (n = 4), followed by weekly rFVIII infusions. Encouragingly, we observed 2- to 4-fold reduced anti-FVIII antibody titres and functional inhibitors in G-CSF-pre-treated mice. G-CSF expansion of MDSC was modest and we are currently optimizing endogenous MDSC expansion regimens.
For the first time, to our knowledge, we assessed the dynamics of endogenous MDSC in FVIII-treated HA mice, revealing decreased circulating MDSC after 4 weeks of rFVIII exposure, and diminished CD11b expression in all examined MDSC compartments. We hypothesize this phenomenon leads to impaired MDSC function, necessary to mount an optimal adaptive immune response to FVIII, although this awaits further confirmation. Finally, our studies suggest it is feasible to expand MDSC ex vivo from na•ve HA bone marrow for future adoptive transfer experiments, and to expand endogenous HA MDSC using growth factors such as G-CSF, as novel investigative approaches to mediating FVIII tolerance. If warranted by murine studies, G-CSF may be an attractive novel immune tolerance strategy, given extensive clinical experience with human G-CSF administration.
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal