Abstract
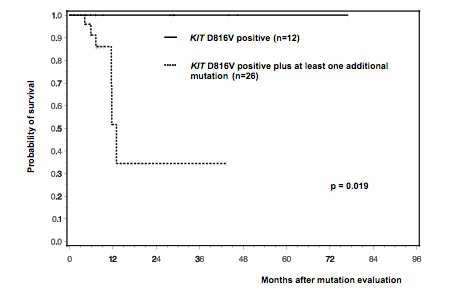
Systemic mastocytosis (SM) is a rare hematologic neoplasm characterized by abnormal accumulation of mast cells in various tissues, predominantly skin, bone marrow and visceral organs. The morphologic phenotype and extent of organ infiltration/dysfunction are basis for the subclassification of SM into indolent SM (ISM), smoldering SM (SSM), SM with associated hematologic non-mast cell disease (SM-AHNMD), aggressive SM (ASM) and mast cell leukemia (MCL). A somatic point mutation in the kinase domain of the receptor tyrosine kinase (TK) KIT at position 816 (KIT D816V) is present in >95% of patients and plays a central role in the pathogenesis and diagnosis of SM. To further explore mechanisms contributing to the clinical diversity of SM, we analyzed 39 KIT D816V mutated patients with different SM subtypes [ISM, n=10; SSM, n=2; ISM-AHNMD, n=5 (CMML, n=2; MDS/MPNu, n=3); ASM, n=1; ASM-AHNMD, n=14 (CMML, n= 5, MDS/MPNu, n=4, HES/CEL, n=2; AML, n=3); MCL, n=3; MCL-AHNMD n=4 (MDS/MPNu, n=2; HES/CEL, n=1; MDS, n=1)] for the presence of additional mutations. We applied next-generation sequencing to investigate ASXL1, CBL, IDH1/2, JAK2, KRAS, MLL-PTD, NPM1, NRAS, TP53, SRSF2, SF3B1, SETBP1, U2AF1 at mutational hotspot regions, and analyzed the complete coding regions of EZH2, ETV6, RUNX1, and TET2. Additional molecular aberrations were identified in 24/27 (89%) patients with advanced SM (SM-AHNMD, 5/5; ASM/MCL, 19/23) while only 3/12 (25%) ISM/SSM patients carried one additional mutation each (U2AF1, SETBP1, CBL) (p<0.001). In TET2, 9 missense, 4 nonsense, 13 frameshift mutations and one in-frame deletion as well as one splice site mutation were found in 15/39 (39%) patients. Ten of 15 (67%) patients carried more than one TET2 mutation. In SRSF2, missense mutations were identified in 14/39 (36%) patients which were clustered at codon 95 (P95H, n=9; P95L, n=2; P95R, n=2), with one additional mutation identified at codon 18 (V18L, n=1). In RUNX1, 10 mutations (9 missense mutations, 1 frameshift) were identified in 9/39 (23%) patients. Ten CBL mutations were identified in 8 patients (8 missense mutations, 1 duplication, 1 splice mutation) and 8 ASXL1 mutations were identified in 8 patients (7 frameshift and 1 nonsense mutation). In 13 patients, two (TET2, n=8; KRAS, n=1), three (CBL, n=2) or four mutations (TET2, n=2) were found in a single gene. Less frequently affected genes were KRAS (n=4), NRAS, JAK2, U2AF1 (n=2, each), EZH2, SETBP1, and ETV6 (n=1, each). No mutations were identified in MLL, IDH1, IDH2, NPM1, SF3B1 and TP53. In advanced SM, 21/27 patients (78%) carried ³3 mutations and 11/27 patients (41%) exhibited ³5 mutations. The median number of mutations was 4. The concurrent presence of KIT-TET2-SRSF2 (10/39, 26%), KIT-SRSF2-RUNX1 (7/39, 18%), KIT-TET2-CBL (5/39, 13%), KIT-SRSF2-ASXL1 (4/39, 10%) and KIT-TET2-ASXL1 (4/39, 10%) was strongly associated with (A)SM/MCL-AHNMD in 16/16 (100%) of patients (CMML, 7/16, 44%; MDS/MPNu, 6/16, 38%; HES/CEL, 3/16, 19%). Clinical follow-up was available for 38 patients. Six of 38 (16%) patients died, all of whom had at least one additional mutation with 5 patients positive for ³3 and 2 patients for ³5 mutations. The median survival of all 26 patients with at least one additional mutation was 12 months. In contrast, none of the 12 cases with KIT D816V mutation only died. This translated into a significant inferior survival (p=0.019) for patients with additional mutations (figure 1). We therefore conclude that, in addition to the multilineage involvement by KIT D816V, the presence of additional molecular aberrations is a new molecular feature that may contribute essentially to the abnormal phenotype and behaviour of neoplastic mast cells in advanced SM and thus to the clinical diversity and prognosis of advanced mast cell disorders.
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal