Key Points
Following endothelial damage, platelet cross-linking during closure of the vessel lumen is mediated by GpIbα-VWF interactions.
Disruption of GpIbα-VWF interactions restores vessel patency by specifically disaggregating the external layer of occlusive thrombi.
Abstract
Interactions between platelet glycoprotein (Gp) IIb/IIIa and plasma proteins mediate platelet cross-linking in arterial thrombi. However, GpIIb/IIIa inhibitors fail to disperse platelet aggregates after myocardial infarction or ischemic stroke. These results suggest that stability of occlusive thrombi involves additional and as-yet-unidentified mechanisms. In the present study, we investigated the mechanisms driving platelet cross-linking during occlusive thrombus formation. Using computational fluid dynamic simulations and in vivo thrombosis models, we demonstrated that the inner structure of occlusive thrombi is heterogeneous and primarily determined by the rheological conditions that prevailed during thrombus growth. Unlike the first steps of thrombus formation, which are GpIIb/IIIa-dependent, our findings reveal that closure of the arterial lumen is mediated by GpIbα-von Willebrand Factor (VWF) interactions. Accordingly, disruption of platelet cross-linking using GpIbα-VWF inhibitors restored vessel patency and improved outcome in a mouse model of ischemic stroke, although the thrombi were resistant to fibrinolysis or traditional antithrombotic agents. Overall, our study demonstrates that disruption of GpIbα-VWF interactions restores vessel patency after occlusive thrombosis by specifically disaggregating the external layer of occlusive thrombi, which is constituted of platelet aggregates formed under very high shear rates.
Introduction
Thrombus development in the circulation is the most common cause of mortality and morbidity worldwide.1 Available thrombolytic treatments target only one constituent of the thrombus, fibrin, and are therefore poorly efficient to dissolve arterial and platelet-rich thrombi.2,3 Recent clinical studies demonstrated that inhibitors of platelet glycoprotein (Gp)IIb/IIIa do not significantly impact the patency of preoccluded arteries,4 although they efficiently prevent initial thrombus formation and dissolve fresh platelet aggregates.5-7 These clinical data suggest that occlusive thrombosis involves additional and as-yet-unidentified mechanisms of platelet cross-linking.
Numerous studies demonstrated that local shear rates determine platelet cross-linking mechanisms.1 Thus, in the range of physiological arterial shear rates (1000-10 000 s−1), large aggregate formation requires platelet activation and occurs in a GpIIb/IIIa-dependent manner.8 Under very high shear rates (>10 000 s−1), platelet aggregation can occur independently of platelet activation.9 In addition, stability of the developing thrombus on the vessel wall relies on a fibrin network.1 However, during occlusive thrombus formation, the respective contributions of these shear-specific mechanisms remain unknown.
In a recent study, Momi et al.10 demonstrated that blockade of the interaction between GpIbα and von Willebrand Factor (VWF) leads to reperfusion after photothrombosis-induced stroke in guinea pigs, whereas GpIIb/IIIa inhibitors are ineffective. Although the beneficial effect of GpIbα-VWF targeting through inhibition of the postreperfusion thrombo-inflammatory reaction is well established in transient mechanical stroke models,11-14 the reason why GpIbα-VWF blockade dissolves already-formed occlusive thrombi remains unclear.15
In the present study, using computational fluid dynamics (CFD) simulations and in vivo thrombotic stroke models, we dissected the mechanisms involved in platelet cross-linking during occlusive thrombus formation and propose an integrated view of the thrombotic process, from vessel wall damage to lumen closure. We demonstrate, in particular, that platelet cross-linking during closure of the vessel lumen is dependent on GpIbα-VWF interactions. Accordingly, disruption of GpIbα-VWF interactions restores vessel patency by specifically disaggregating the external layer of occlusive thrombi.
Methods
Detailed methods can be found in the supplemental Data available on the Blood Web site.
CFD simulation
Fluid dynamics were simulated using Ansys Multiphysics package including the Fluent module (v14.5; Montigny-le-Bretonneux, France). We assumed the validity of the continuum hypothesis and no-slip boundary condition. We considered the flow to be 2-dimensional, steady, laminar, and incompressible. The sizes of the modeled middle cerebral artery (MCA) and common carotid artery (CCA) were 200 and 750 µm, respectively. The thrombus was modeled as a growing disk inside the arterial lumen. The viscosity of the fluid was set to 0.00345 kg/m ⋅ s−1 and the density to 998.2 kg/m3.16 The inlet velocity was set at 9.2 cm ⋅ s−1 for MCA simulation and at 30.0 cm ⋅ s−1 for CCA simulation.17
Animals
Experiments were performed on adult, male Swiss mice (35 ± 2 g; CURB, Caen, France), VWF-deficient mice18 (VWF−/−), and C57BL/6 wild-type (WT) mice (15-18 weeks old). All experiments were conducted in accordance with directives from the European Communities Council (November 24, 1986, 86/609/EEC) and French Legislation (act no. 87-848, Ministère de l'Agriculture et de la Forêt) on animal experimentation. All the experiments were validated by the local ethical committee of Normandy (CENOMEXA) registered under the reference CENOMEXA-0113-03 and received the agreement number N/03-01-13/03/01-18.
Surgical procedures
Mice were deeply anesthetized with 5% isoflurane (Baxter, Paris, France, in a mixture of N2O/O2), placed in a stereotaxic device, and maintained under anesthesia for up to 2 hours with 2% isoflurane19 in a 70%:30% mixture of N2O/O2. Through a skin incision between the right eye and ear, the masseter muscle was excised, and a small craniotomy (diameter, 1 mm) was performed on the parietal bone to expose the right MCA.
Thrombus induction
A piece of Whatman filter paper strip soaked in freshly prepared FeCl3 (5% to 20%, Sigma-Aldrich, Lisle d'Abeau, France) was placed on the intact dura mater on top of the MCA for 4 minutes.20,21 Cerebral blood flow (CBF) in the MCA territory was determined by laser Doppler flowmetry (Oxford Optronix). CBF was continuously measured before MCA occlusion (MCAo, 100% baseline) and throughout the duration of ischemia and thrombolysis. Occlusion time was defined as the time between FeCl3 application and CBF diminution <20% of baseline. No intracranial bleeding was detected in the animals included in this study.
Blockade of platelet receptors
Mice were intravenously injected with either aurintricarboxylic acid (ATA; 20 mg/kg; Sigma-Aldrich), a polycarboxylated compound that binds to the A1 domain of VWF, thus preventing binding of VWF to platelet GpIbα,22 or GR144053 trihydrochloride (GR; 10 mg/kg; R&D Systems, Lille, France), a nonpeptidic GpIIb/IIIa inhibitor. ATA and GR solutions were passed through 0.22-µm filters before administration. When mentioned, ATA or GR was injected as a rapid bolus (200 µL) during thrombus formation, which was considered ongoing until CBF dropped to <50% of the baseline value after FeCl3 application (assessed by laser Doppler flowmetry). In another set of experiments, the VWF binding domain of GpIbα was blocked by injection of 2.5 mg/kg of Fab fragments of the monoclonal antibody Xia.B223,24 (Emfret Analytics, Würzburg, Germany). The activated GpIIb/IIIa was blocked by injection of 2.5 mg/kg of Fab fragments (achieving 100% receptor blockade23 ) of the monoclonal antibody Leo.H425 (Emfret Analytics). Isotype control IgG rats were purchased from Jackson Immunoresearch. Fab fragments were prepared using Pierce Fab preparation kit (Fischer Scientific, Illkirch, France) according to the manufacturer’s instructions.
Magnetic resonance imaging analysis
Experiments were carried out on a Pharmascan 7 T/12 cm system using surface coils (Bruker Biospin, Wissembourg, France). T2-weighted images were acquired using a Multi-Slice Multi-Echo sequence: TE/TR 51 ms/2500 ms. Lesion sizes were quantified on T2 weighted images using ImageJ software (v1.45r, National Institutes of Health). Magnetic resonance angiographies were performed using a 2D-TOF sequence.26
Statistical analyses
Results are expressed as mean ± SEM. Statistical analyses were performed using Kruskal-Wallis test followed by Mann-Whitney test (unless otherwise indicated). Values were considered statistically significant if P < .05.
Results
Rheological conditions define 3 distinct regions in developing thrombi
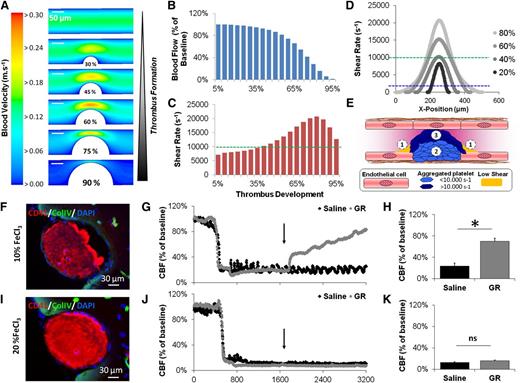
We first performed CFD simulations of the blood flow in the mouse MCA at different stages of thrombus development (Figure 1A; supplemental Figure 1). The aim of this simulation was to determine the spatial and temporal variations in shear rate during the whole thrombotic process. The simulation revealed that during the first part of the thrombotic process, the peak shear rate at the top of the thrombus slowly increases from ∼7000 s−1 to 10 000 s−1 until 40% of the thrombus is constituted (green dotted line). Then, the shear rate rapidly increases up to a maximum of ∼20 000 s−1 when 80% of the artery is occluded (Figure 1B-D). Notably, during all the process of thrombus formation, the shear rate at the base of the thrombus remains <1.500 s−1 (blue dotted line). Thus, according to the shear rate prevailing during thrombus formation, 3 regions in the resultant occlusive thrombus can be determined (Figure 1E): the first one is the base of the thrombus that is exposed to shear rates <1500 s−1 during all the thrombotic process (yellow areas). The second one is the “core” of the thrombus, corresponding to the part of the thrombus constituted during the first steps of development (0% to 40%) at shear rates comprised between 1500 and 10 000 s−1 (light blue platelets). The third one is formed during the last step of thrombus formation (40% to 100%) at very high shear rates (>10 000 s−1) and is responsible for vessel lumen closure (dark blue platelets). Because platelet cross-linking mechanisms may vary according to the shear rate,8 these results raise the intriguing possibility that the molecular interactions mediating thrombus stability are different in these 3 regions.
CFD simulations and in vivo occlusive thrombosis. (A) Blood velocity map inside the MCA is represented at different stages of occlusive thrombus formation. (B) Computed blood flow in the MCA from 5% to 95% of thrombus formation. (C) Peak shear rate at the thrombus surface during thrombosis. (D) Profile of the shear rate along the thrombus surface at different stages of thrombus formation. (E) Schematic drawing of the 3 different regions in occlusive thrombi. (F) Representative immunohistological images of the MCA 20 minutes after topical application of 10% FeCl3 (representative of 5 mice). (G) Representative laser Doppler profile of the CBF inside the MCA territory following 10% FeCl3 application and treatment by either saline or GR (10 mg/kg, a GpIIb/IIIa inhibitor) 20 minutes after thrombosis. (H) Corresponding quantification of the CBF at the end of the monitoring period (40 minutes posttreatment, n = 5). (I-K) Same as in F-H but using a solution of 20% FeCl3. ns, not significant.
CFD simulations and in vivo occlusive thrombosis. (A) Blood velocity map inside the MCA is represented at different stages of occlusive thrombus formation. (B) Computed blood flow in the MCA from 5% to 95% of thrombus formation. (C) Peak shear rate at the thrombus surface during thrombosis. (D) Profile of the shear rate along the thrombus surface at different stages of thrombus formation. (E) Schematic drawing of the 3 different regions in occlusive thrombi. (F) Representative immunohistological images of the MCA 20 minutes after topical application of 10% FeCl3 (representative of 5 mice). (G) Representative laser Doppler profile of the CBF inside the MCA territory following 10% FeCl3 application and treatment by either saline or GR (10 mg/kg, a GpIIb/IIIa inhibitor) 20 minutes after thrombosis. (H) Corresponding quantification of the CBF at the end of the monitoring period (40 minutes posttreatment, n = 5). (I-K) Same as in F-H but using a solution of 20% FeCl3. ns, not significant.
To test this hypothesis, we first developed in vivo models of MCA thrombosis in mice by topical application of FeCl3. Different concentrations of FeCl3 (5% to 20%) were tested to achieve either partial or complete occlusion of the MCA. As assessed by immunohistofluorescence analyses and Doppler flowmetry, use of 5% FeCl3 failed to induce thrombosis (supplemental Fig 2); use of 10% FeCl3 induced partial thrombosis, whereas use of 20% FeCl3 triggered a stable and fully occlusive thrombus (Figure 1F-K). The residual laser Doppler signal in mice with occlusive thrombi results from blood movements in the probed tissue outside the MCA. Both 10% and 20% FeCl3 led to ischemic brain damage in the MCA territory (supplemental Fig 3), as assessed by magnetic resonance imaging. According to CFD simulations, thrombi resulting from topical application of 10% FeCl3 are primarily composed of the thrombus core, which constituted at shear rates <10 000 s−1, whereas occlusive thrombi resulting from topical application of 20% FeCl3 constituted mainly at shear rates >10 000 s−1.
Because current knowledge indicates that platelet cross-linking is primarily mediated by GpIIb/IIIa-dependent interactions,27 we investigated whether intravenous injection of a GpIIb/IIIa inhibitor (GR, 10 mg/kg) 20 minutes after thrombus formation can disaggregate occlusive and nonocclusive thrombi.5,7 Interestingly, whereas the nonocclusive thrombi (induced by 10% FeCl3) were efficiently disaggregated by GpIIb/IIIa inhibition, the fully occlusive thrombi (induced by 20% FeCl3) were resistant (Figure 1F-K). Accordingly, administration of GR reduced ischemic brain damage exclusively when the thrombi were nonocclusive (supplemental Fig 3). Therefore, this thrombotic stroke model reproduces the findings of clinical studies involving patients with occlusive (resistant to GpIIb/IIIa inhibitors) and nonocclusive thrombi (responsive to GpIIb/IIIa inhibitors). Overall, these data suggest that the core of the thrombus is primarily constituted of platelets cross-linked by GpIIb/IIIa-dependent interactions, whereas the stability of the last-formed parts of the thrombus involves additional and as-yet-unidentified mechanisms.
GpIbα-VWF interactions are required to achieve rapid vessel lumen closure
To further investigate these mechanisms, we dissected step-by-step the formation of occlusive thrombus following 20% FeCl3 application on the MCA. Previous studies have demonstrated that thrombus formation at arterial shear rate starts by a platelet adhesion phase, which involves GpIbα-VWF interactions.8 The second phase involves platelet aggregation, in which platelet tethering to the developing thrombus is also initiated by GpIbα-VWF, but subsequent firm platelet cross-linking is mediated by GpIIb/IIIa and its ligands.8 We first wanted to confirm these different steps in the present model of thrombosis. Thus, we performed FeCl3-induced injury of the MCA of WT and VWF−/− mice under continuous Doppler monitoring. In contrast to WT animals, VWF−/− mice were protected from vascular occlusion, and no ischemic lesion was detectable at 24 hours (supplemental Figure 4).
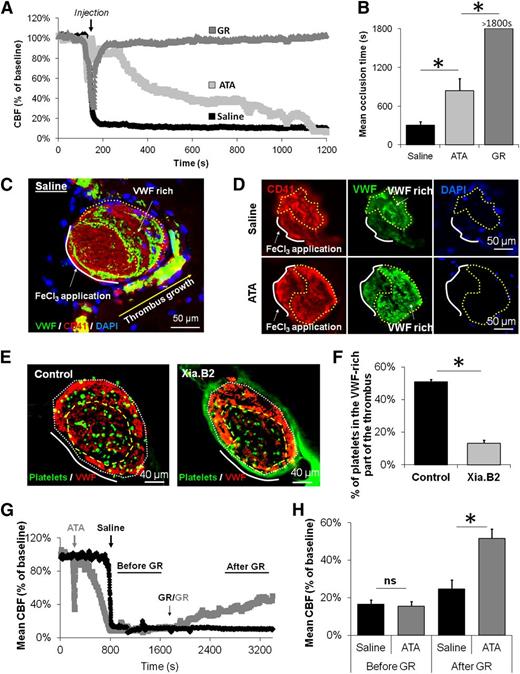
Next, to confirm the roles of GpIIb/IIIa and GpIbα-VWF interactions in early platelet adhesion and aggregation, WT mice were injected with either a GpIbα-VWF inhibitor (ATA, which blocks the GpIbα binding domain of VWF22 ) (supplemental Figure 5A-B) or with a GR 2 minutes before FeCl3 application. Unlike saline pretreated animals, mice pretreated with ATA or GR were protected from vascular occlusion (supplemental Figure 6A-B), demonstrating that both GpIbα-VWF interactions and GpIIb/IIIa are necessary for the formation of an occlusive thrombus in this model. To study the role of GpIbα-VWF interactions and GpIIb/IIIa without interfering with the platelet adhesion phase, we injected GR or ATA after the platelet adhesion phase, during thrombus formation (which was considered ongoing when the CBF dropped below 50% of the basal value (∼4.5 min after FeCl3 application) (Figure 2A). In these conditions, GpIIb/IIIa inhibition caused disaggregation of the developing thrombus and prevented arterial occlusion, confirming that the core of the thrombus can be disaggregated using GpIIb/IIIa inhibitors. Conversely, inhibition of GpIbα-VWF interactions did not prevent formation of stable (>60 minutes) occlusive thrombus, but the time to occlusion increased considerably (Figure 2A-B). This confirms that GpIbα-VWF interactions are necessary to achieve rapid vessel lumen closure after endothelial damage.
GpIbα-VWF interactions are required to achieve rapid vessel lumen closure and participate in thrombus resistance to GpIIb/IIIa inhibitors. (A) Representative laser Doppler profiles of mice treated with saline, ATA, or GR during thrombus formation. The black arrow indicates the time of injection of the different inhibitors. (B) Mean CBF at the end of the monitoring period (30 minutes after FeCl3 application) of mice treated with saline, ATA, or GR during thrombus formation (n = 4 mice/group). (C) Representative immunohistological staining for VWF, CD41 (platelet marker), and 4′6 diamidino-2-phenylindole nuclear staining of MCA of saline-treated mice 20 minutes after FeCl3-induced MCAo (representative of 10 mice). (D) Representative immunohistological stainings (CD41, VWF, and 4′6 diamidino-2-phenylindole) of MCA from mice that received either saline or ATA during thrombus formation. The yellow dotted line delineates the VWF-rich part of the thrombus. (E) Representative immunohistological images of the MCA 20 minutes after thrombosis in mice that received fluorescently labeled platelets intravenously 10 minutes before FeCl3 application (representative of 5 mice). In the Xia.B2 group, platelets were incubated with anti-GpIbα Fab (40 µg/mL) before being injected. (F) Quantitative analysis of the proportion of platelets in the VWF-rich part of the thrombus after injection of control or GpIbα-blocked, fluorescently labeled platelets (n = 4 mice/group). (G) Representative laser Doppler profiles of mice treated with saline or ATA during thrombus formation and with GR 20 minutes after MCAo. (H) Mean CBF measured in both groups before and after GR treatment (n = 5 mice/group). ns, not significant.
GpIbα-VWF interactions are required to achieve rapid vessel lumen closure and participate in thrombus resistance to GpIIb/IIIa inhibitors. (A) Representative laser Doppler profiles of mice treated with saline, ATA, or GR during thrombus formation. The black arrow indicates the time of injection of the different inhibitors. (B) Mean CBF at the end of the monitoring period (30 minutes after FeCl3 application) of mice treated with saline, ATA, or GR during thrombus formation (n = 4 mice/group). (C) Representative immunohistological staining for VWF, CD41 (platelet marker), and 4′6 diamidino-2-phenylindole nuclear staining of MCA of saline-treated mice 20 minutes after FeCl3-induced MCAo (representative of 10 mice). (D) Representative immunohistological stainings (CD41, VWF, and 4′6 diamidino-2-phenylindole) of MCA from mice that received either saline or ATA during thrombus formation. The yellow dotted line delineates the VWF-rich part of the thrombus. (E) Representative immunohistological images of the MCA 20 minutes after thrombosis in mice that received fluorescently labeled platelets intravenously 10 minutes before FeCl3 application (representative of 5 mice). In the Xia.B2 group, platelets were incubated with anti-GpIbα Fab (40 µg/mL) before being injected. (F) Quantitative analysis of the proportion of platelets in the VWF-rich part of the thrombus after injection of control or GpIbα-blocked, fluorescently labeled platelets (n = 4 mice/group). (G) Representative laser Doppler profiles of mice treated with saline or ATA during thrombus formation and with GR 20 minutes after MCAo. (H) Mean CBF measured in both groups before and after GR treatment (n = 5 mice/group). ns, not significant.
GpIbα-VWF is involved in platelet recruitment and cross-linking during vessel lumen closure
Thus, to further investigate the role of GpIbα-VWF during vessel lumen closure, we performed immunohistological analyses of occlusive thrombi. Immunohistological staining revealed 2 different parts forming occlusive thrombi: one that was poor in VWF and located proximal to the injured vessel wall, and the second one rich in VWF and located distally at the opposite of the injured vessel wall (Figure 2C). Hence, in MCA sections acquired after thrombus formation, abundance of VWF was maximal during vessel closure and thus appeared to be high in areas of thrombus development under higher shear rates. Conversely, fibrin-fibrinogen rich areas were preferentially located around the base of the thrombus in areas constituted at lower shear rates (supplemental Figure 7).
If animals were pretreated with inhibitors of either GpIIb/IIIa or GpIbα-VWF before FeCl3 application, immunohistological staining confirmed that thrombus development was prevented (data not shown). If the inhibitors were administered during thrombus formation, occlusive thrombosis was prevented in GpIIb/IIIa inhibitor-treated animals but occurred in GpIbα-VWF inhibitor-treated mice (Figure 2D). Additionally, when platelets preincubated with Fab fragments of an antibody blocking the VWF binding site of GpIbα (Xia.B2)23 were injected into recipient mice, they were less efficiently recruited into the VWF-rich part of the thrombus than control Fab-treated platelets (Figure 2E-F). Together with the results shown in Figure 2A-B, these results demonstrate that initial thrombus development requires GpIIb/IIIa and that GpIbα-VWF interactions play a role during vessel lumen closure. Interestingly, we also demonstrated that occlusive thrombi formed in the presence of inhibitors of GpIbα-VWF interactions were efficiently disaggregated by administration of GpIIb/IIIa inhibitors 20 minutes after thrombus formation (Figure 2G-H), whereas thrombi formed in the absence of inhibitors of GpIbα-VWF interactions were resistant. These data demonstrate that GpIbα-VWF interactions mediate thrombus resistance to GpIIb/IIIa inhibitors, suggesting that the role of GpIbα-VWF interactions goes beyond platelet recruitment and extends to platelet cross-linking during vessel lumen closure.
Inhibition of GpIbα-VWF interactions restores vessel patency after occlusive thrombosis
According to this hypothesis, inhibition of GpIbα-VWF interactions should disengage the VWF that cross-links platelets in the VWF-rich part of the thrombus and thus should restore vessel patency. To address this question, we performed immunohistological analyses of thrombi from animals euthanatized 10 minutes after intravenous administration of GpIbα-VWF inhibitors (20 minutes after arterial occlusion). Interestingly, treatment with GpIbα-VWF inhibitors specifically disaggregated the last-formed VWF-rich part of the thrombi. Indeed, the MCA was reopened, and the VWF-rich part of the thrombus appeared 2.7-fold smaller than in saline-treated animals (Figure 3A-D). In mice treated with GpIIb/IIIa inhibitors, thrombi were still present and similar to those in saline-treated animals (data not shown).
Inhibition of GpIbα-VWF interactions restores vessel patency after occlusive thrombosis. (A) Representative immunohistological staining (of 5 mice) of the MCA 10 minutes after ATA treatment (injected 20 minutes after MCAo). Quantification of the relative areas (in percentage of the total MCA area) of the whole thrombus (B), the VWF-poor (C), and the VWF-rich areas (D) in mice brains 10 minutes after administration of either saline or ATA (n = 4 mice/group). ns, not significant.
Inhibition of GpIbα-VWF interactions restores vessel patency after occlusive thrombosis. (A) Representative immunohistological staining (of 5 mice) of the MCA 10 minutes after ATA treatment (injected 20 minutes after MCAo). Quantification of the relative areas (in percentage of the total MCA area) of the whole thrombus (B), the VWF-poor (C), and the VWF-rich areas (D) in mice brains 10 minutes after administration of either saline or ATA (n = 4 mice/group). ns, not significant.
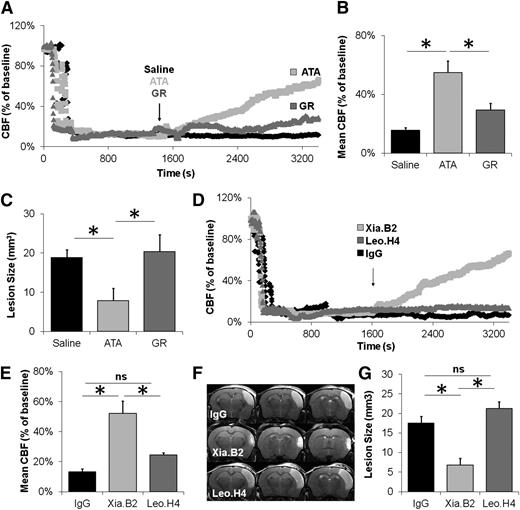
To further demonstrate the thrombolytic efficiency of GpIbα-VWF inhibitors, we compared the efficiency of ATA and GR to restore vessel patency and reduce brain damage following occlusive thrombosis of the MCA. These inhibitors were injected independently 20 minutes after complete formation of a FeCl3-induced occlusive thrombus. Doppler flowmetry and magnetic resonance imaging showed that only inhibition of GpIbα-VWF interactions significantly rescued CBF and reduced volumes of ischemic lesion at 24 hours (Figure 4A-C). Importantly, in mice subjected to permanent MCAo (by electrocoagulation), neither ATA nor GR influenced ischemic lesion sizes, confirming that both agents have no relevant parenchymal effects (supplemental Figure 5C-D).
Inhibition of GpIbα-VWF interactions improves stroke outcome after occlusive thrombosis. (A) Representative laser Doppler profiles of mice treated with saline, ATA, or GR 20 minutes after occlusive thrombus formation. (B) Mean CBF (as assessed by laser Doppler flowmetry 60 min after MCAo) in brains of mice treated with saline, ATA, or GR 20 minutes after MCAo (n = 10-15 mice/group). (C) Corresponding mean ischemic lesion sizes at 24 h post-MCAo (n = 10-15 mice/group). (D) Representative laser Doppler profiles of brains of mice treated with Fab from control IgG, Xia.B2 (2.5 mg/kg, anti-GpIbα), or Leo.H4 (2.5 mg/kg, anti-GpIIb/IIIa) after thrombus formation. (E) Corresponding mean CBF (as assessed by laser Doppler flowmetry 60 minutes after MCAo; n = 6 mice/group). (F) Representative T2-weighted images of Fab from control IgG-, Xia.B2-, and Leo.H4-treated mice 24 hours after FeCl3 application (quantification of lesion sizes is presented in G, n = 6 mice/group). ns, not significant.
Inhibition of GpIbα-VWF interactions improves stroke outcome after occlusive thrombosis. (A) Representative laser Doppler profiles of mice treated with saline, ATA, or GR 20 minutes after occlusive thrombus formation. (B) Mean CBF (as assessed by laser Doppler flowmetry 60 min after MCAo) in brains of mice treated with saline, ATA, or GR 20 minutes after MCAo (n = 10-15 mice/group). (C) Corresponding mean ischemic lesion sizes at 24 h post-MCAo (n = 10-15 mice/group). (D) Representative laser Doppler profiles of brains of mice treated with Fab from control IgG, Xia.B2 (2.5 mg/kg, anti-GpIbα), or Leo.H4 (2.5 mg/kg, anti-GpIIb/IIIa) after thrombus formation. (E) Corresponding mean CBF (as assessed by laser Doppler flowmetry 60 minutes after MCAo; n = 6 mice/group). (F) Representative T2-weighted images of Fab from control IgG-, Xia.B2-, and Leo.H4-treated mice 24 hours after FeCl3 application (quantification of lesion sizes is presented in G, n = 6 mice/group). ns, not significant.
To provide additional evidence that disrupting the GpIbα-VWF interactions leads to restoration of vessel patency following occlusive thrombosis, we injected Fab from monoclonal antibodies targeting either GpIIb/IIIa (Leo.H4, 2.5 mg/kg, >95% receptor blockade13 ) or the VWF-binding domain of GpIbα (Xia.B2, 2.5 mg/kg) 20 minutes after MCAo. Although both antibody-derived Fab prevented thrombus formation when they were administered before endothelial damage (supplemental Figure 6C-D), only Fab targeting GpIbα significantly improved CBF and reduced ischemic lesion size compared with control Fab-treated animals (Figure 4D-G). Thus, targeting the GpIbα-binding A1 domain of VWF with ATA or targeting the VWF-binding domain of GpIbα with Xia.B2 Fab were both effective in disaggregating occlusive thrombi and in restoring vessel patency.
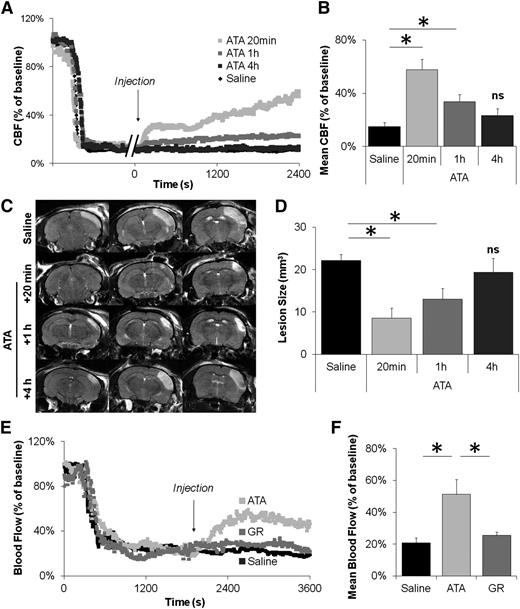
Thereafter, we aimed to determine the therapeutic window of GpIbα-VWF interaction inhibition. To this aim, we injected ATA either 20 minutes, 1 hour, or 4 hours after occlusive thrombus formation and compared the resulting CBF and ischemic lesion size with saline-treated animals. Our results demonstrate that inhibition of GpIbα-VWF interactions remains effective at least 1 hour after occlusive thrombosis in terms of CBF rescue and reduction of ischemic lesion size (Figure 5A-D). In contrast, 4 hours after arterial occlusion, thrombi became resistant to ATA, and the ischemic lesion size was similar in ATA- and saline-treated animals, revealing a therapeutic time-window for GpIbα-VWF inhibition.
Blockade of the GpIbα-VWF interactions remains efficient one hour after thrombus formation in the MCA and after occlusive thrombosis of the CCA. (A) Representative laser Doppler profiles of mice treated with ATA (20 mg/kg) 20 minutes, 1 hour, or 4 hours post-MCAo. Control animals received saline 1 hour post-MCAo. (B) Mean CBF of mice treated with saline or ATA at different time points. (C) Representative T2-weighted images of saline- and ATA-treated mice 24 hours after FeCl3 application. (D) Corresponding quantification of lesion sizes. (E) Representative laser Doppler profiles of mice treated with saline, ATA (20 mg/kg), or GR (10 mg/kg) 20 minutes postocclusion of the CCA. (F) Mean blood flow of mice treated with saline, ATA, or GR 20 minutes postocclusion of the CCA (n = 4/group). ns, not significant.
Blockade of the GpIbα-VWF interactions remains efficient one hour after thrombus formation in the MCA and after occlusive thrombosis of the CCA. (A) Representative laser Doppler profiles of mice treated with ATA (20 mg/kg) 20 minutes, 1 hour, or 4 hours post-MCAo. Control animals received saline 1 hour post-MCAo. (B) Mean CBF of mice treated with saline or ATA at different time points. (C) Representative T2-weighted images of saline- and ATA-treated mice 24 hours after FeCl3 application. (D) Corresponding quantification of lesion sizes. (E) Representative laser Doppler profiles of mice treated with saline, ATA (20 mg/kg), or GR (10 mg/kg) 20 minutes postocclusion of the CCA. (F) Mean blood flow of mice treated with saline, ATA, or GR 20 minutes postocclusion of the CCA (n = 4/group). ns, not significant.
Then, we investigated whether GpIbα-VWF interaction inhibitors remain efficient in large arteries. According to CFD simulations, the shear rate was predicted to be slightly higher in the CCA than in the MCA, without striking differences in the wall shear rate profiles (supplemental Fig 8), suggesting that the resulting thrombus structures should be similar in the 2 arteries. Subsequently, we induced occlusive thrombosis of the CCA in mice by topical application of FeCl3, as previously described.28 Again, whereas the resulting thrombi were resistant to GpIIb/IIIa inhibitors, inhibition of GpIbα-VWF interactions 20 minutes after carotid occlusion restored arterial patency (Figure 5E-F). Magnetic resonance angiograms confirmed these results by revealing a restored arterial lumen in all ATA-treated mice but a persistent occlusion in all saline- and GR-treated mice 24 hours after treatment administration (supplemental Figure 9).
Inhibition of GpIbα-VWF interactions is more efficient than conventional antithrombotic treatments
Thereafter, we wanted to compare the efficacy of GpIbα-VWF blockade in the FeCl3 model on the MCA to other available antithrombotic treatments. Surprisingly, among the fibrinolytic (supplemental Figure 10A-C), antiplatelet, and anticoagulant treatments tested (supplemental Figure 10D-G and 11), only administration of 2000 IU/kg of unfractionated heparin (UFH) restored vessel patency and improved ischemic lesion size when injected 20 minutes after occlusion, notwithstanding a high mortality rate at 24 hours (57%; supplemental Figure 10H) probably explained by UFH-induced internal hemorrhages (not shown). As UFH and other anticoagulants have distinct effects on thrombus stability, these results imply that UFH acts independently of thrombin or Factor X inactivation to restore vessel patency. Interestingly, this thrombolytic effect can be explained by the fact that, besides its anticoagulant effects, UFH is also a potent inhibitor of GpIbα-VWF interactions.29
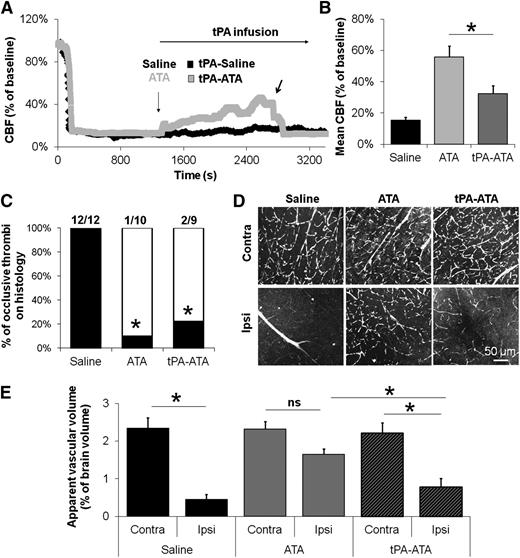
Then, we looked for potential interactions between GpIbα-VWF inhibitors and the 2 approved pharmacological treatments for acute ischemic stroke, aspirin, and tissue-type plasminogen activator (tPA). Whereas aspirin treatment did not influence the thrombolytic effects of GpIbα-VWF inhibitors (supplemental Figure 12), the combination of tPA infusion with GpIbα-VWF inhibitor administration attenuated the effects of GpIbα-VWF inhibitor treatment alone, as evidenced from the smaller beneficial effects of the combination therapy in terms of CBF and ischemic lesion size (Figure 6A-B and supplemental Figure 13). Notably, Doppler flowmetry analysis suggested generation of large emboli from the main thrombus in mice treated with both ATA and tPA (Figure 6A, arrow). Immunohistological and microperfusion experiments confirmed this finding, by revealing an altered distal microcirculation in ATA-tPA treated mice, despite lysis of the main thrombus (Figure 6C-E). These data suggest that fibrinolysis promotes detachment of the thrombus from the vessel wall once vessel patency has been restored by GpIbα-VWF inhibitors.
Fibrinolysis during GpIbα-VWF interaction blockade promotes embolization of large platelet aggregates from the thrombus and impairs distal microcirculation. (A) Representative laser Doppler profiles of mice treated with saline or ATA followed by continuous infusion of tPA. The black arrow indicates a sudden drop in CBF during tPA infusion, probably due to embolism. (B) Mean CBF at the end of the monitoring period (40 minutes posttreatment, n = 10-11). (C) Proportion of animals with occlusive thrombi on histology 1 hour after treatment injection (n = 9-12). (D) Representative multiphoton images of Evan’s blue-injected mice 1 hour after saline, ATA, or tPA-ATA treatment. Both ipsilateral and contralateral sides are represented. (E) Mean apparent vascular volume in saline-, ATA-, and tPA-ATA–treated mice 1 hour after administration (n = 4/group). ns, not significant.
Fibrinolysis during GpIbα-VWF interaction blockade promotes embolization of large platelet aggregates from the thrombus and impairs distal microcirculation. (A) Representative laser Doppler profiles of mice treated with saline or ATA followed by continuous infusion of tPA. The black arrow indicates a sudden drop in CBF during tPA infusion, probably due to embolism. (B) Mean CBF at the end of the monitoring period (40 minutes posttreatment, n = 10-11). (C) Proportion of animals with occlusive thrombi on histology 1 hour after treatment injection (n = 9-12). (D) Representative multiphoton images of Evan’s blue-injected mice 1 hour after saline, ATA, or tPA-ATA treatment. Both ipsilateral and contralateral sides are represented. (E) Mean apparent vascular volume in saline-, ATA-, and tPA-ATA–treated mice 1 hour after administration (n = 4/group). ns, not significant.
Discussion
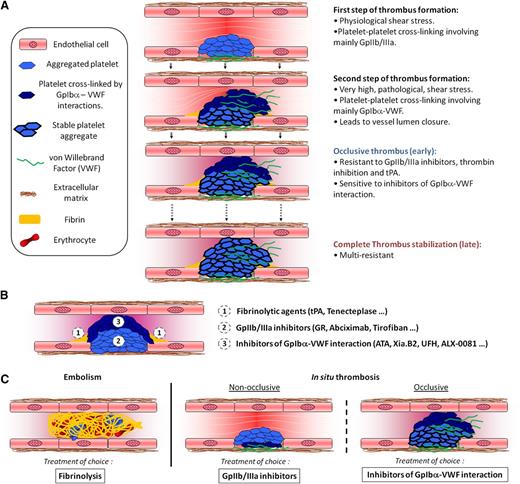
The main finding of the current study is that GpIbα-VWF inhibitors restore vessel patency after occlusive thrombosis by specifically disaggregating the external layer of occlusive thrombi. In contrast, the clot-busting effect of GpIIb/IIIa inhibition is restricted to the core of the thrombus. Overall, our study integrates the different reported mechanisms of platelet aggregation into one unified model to explain the formation and the resulting structure of occlusive thrombi (Figure 7A). Our study extends the hierarchical model proposed by Stalker et al30 by demonstrating that once the thrombus occludes ∼50% of the artery, platelet cross-linking in its external layer occurs in a GpIbα-VWF dependent manner. This model helps reconcile the lack of efficiency of GpIIb/IIIa inhibitors to treat acute occlusive thrombosis,4 despite their efficiency to disaggregate fresh platelet aggregates31 and nonocclusive thrombi.6 This model also provides an explanation for the thrombolytic effect of GpIbα-VWF blockade after occlusive thrombosis in guinea pigs and confirms this finding in another model of MCA thrombosis.10 In fact, our results demonstrate that each part of the occlusive thrombi is responsive to specific thrombolytic agents (Figure 7B), suggesting that thrombolytic strategies should be adapted to thrombus subtype (Figure 7C).
A multistep process of occlusive thrombosis. (A) First, platelets aggregate through involvement of their GpIIb/IIIa receptors, resulting in partial occlusion of the blood vessel and a locally increased shear rate (Step 1). At this early stage, the nonocclusive thrombi are responsive to treatment with GpIIb/IIIa inhibitors. Subsequently, when the shear stress becomes elevated, platelet aggregation to the developing thrombus becomes GpIbα-VWF dependent, until closure of the vessel lumen (Step 2). At this later stage, the occlusive thrombi are resistant to GpIIb/IIIa inhibitors or tPA and only responsive to inhibitors of the GpIbα-VWF interaction. Then, the thrombus progressively stabilizes (>1 hour) and finally becomes resistant to inhibitors of the GpIbα-VWF interactions (Step 3). Additionally, all over the thrombotic process, the low shear rates at the base of the thrombus (both upstream and downstream) favor the formation of fibrin networks, which anchor the thrombus to the vessel wall. (B) Each of the 3 regions in occlusive thrombi is responsive to a specific thrombolytic agent. (C) Therefore, the most efficient thrombolytic strategy may depend on the thrombus developmental stage and on the mechanism responsible for its formation.
A multistep process of occlusive thrombosis. (A) First, platelets aggregate through involvement of their GpIIb/IIIa receptors, resulting in partial occlusion of the blood vessel and a locally increased shear rate (Step 1). At this early stage, the nonocclusive thrombi are responsive to treatment with GpIIb/IIIa inhibitors. Subsequently, when the shear stress becomes elevated, platelet aggregation to the developing thrombus becomes GpIbα-VWF dependent, until closure of the vessel lumen (Step 2). At this later stage, the occlusive thrombi are resistant to GpIIb/IIIa inhibitors or tPA and only responsive to inhibitors of the GpIbα-VWF interaction. Then, the thrombus progressively stabilizes (>1 hour) and finally becomes resistant to inhibitors of the GpIbα-VWF interactions (Step 3). Additionally, all over the thrombotic process, the low shear rates at the base of the thrombus (both upstream and downstream) favor the formation of fibrin networks, which anchor the thrombus to the vessel wall. (B) Each of the 3 regions in occlusive thrombi is responsive to a specific thrombolytic agent. (C) Therefore, the most efficient thrombolytic strategy may depend on the thrombus developmental stage and on the mechanism responsible for its formation.
Notably, our study further highlights the GpIbα-VWF axis as a relevant therapeutic target for acute occlusive thrombosis. We were not able to investigate the potential pro-hemorrhagic effect of GpIbα-VWF blockade, because no hemorrhage was detected in our stroke model. However, in previous studies of ischemic stroke, blockade of GpIbα-VWF interactions did not increase the rate of intracranial hemorrhage,10,14 suggesting that this therapeutic strategy would be safer than current thrombolytic treatments. Because several inhibitors of GpIbα-VWF interactions are currently under preclinical and clinical investigations as thromboprophylactic drugs,32,33 they constitute promising candidates for the pharmacological treatment of acute occlusive thrombotic disorders. Whether part of the beneficial effect of GpIbα-VWF blockade observed in the present study is due to protection from reperfusion injury remains, however, to be investigated.
Although we did not demonstrate that platelet cross-linking is exclusively dependent on GpIbα-VWF interactions in the outer part of the thrombus, our results provide evidence that blockade of this interaction is sufficient to induce thrombus disaggregation. Future studies may look at the platelet activation state during vessel lumen closure to determine whether they remain in a quiescent state and thus exclude any participation of GpIIb/IIIa receptors in this process. This hypothesis is supported by previous studies reporting discoid and activation-independent platelet aggregation in specific rheological conditions.9,16
One potential weakness of GpIbα-VWF blockade remains its limited efficient therapeutic window, because the thrombus becomes rapidly resistant to GpIbα-VWF inhibitors, probably by multiple concomitant mechanisms.34 Future insights into these mechanisms may lead to the development of more efficient strategies for late reperfusion. Moreover, our data suggest that thromboprophylactic GpIbα-VWF inhibition should be associated with inhibition of GpIIb/IIIa-VWF interactions to fully prevent occlusive thrombotic events. Indeed, although platelets in the last-forming part of the thrombus preferentially aggregate by GpIbα-VWF–dependent mechanisms, we demonstrated that, once engaged, complete thrombus formation can occur despite the presence of GpIbα-VWF inhibitors (Figure 2A-B). High VWF content and sensitivity to GpIIb/IIIa inhibitors of the resulting thrombus (Figure 2D-H) suggest the involvement of compensatory mechanisms mediated by GpIIb/IIIa-VWF interactions to achieve vessel lumen closure in these settings.
Although an additive beneficial effect of coadministration of tPA and an inhibitor of GpIbα-VWF interactions may have been expected, this in fact partially alleviates the beneficial effects of GpIbα-VWF inhibition by impairing distal microperfusion (Figure 6). We first observed that concomitant GpIbα-VWF blockade and tPA administration induce embolization and a sudden drop in CBF (as assessed by laser Doppler flowmetry) (Figure 6A-B). Then, we demonstrated that distal microcirculation was impaired in those animals, although the MCA was reopened (Figure 6C-E). Fibrinolysis may weaken the anchorage of the thrombus to the vessel wall and promote embolization of large platelet aggregates. This hypothesis is supported by recent clinical data revealing that thrombolysis improves initial arterial patency but impairs late myocardial perfusion by increasing distal thrombus burden in patients with acute myocardial infarction.35
One potential limitation of the present study is the use of small pharmacological inhibitors that might be less specific than monoclonal antibody fragments. However, GR is a well-characterized and widely used GpIIb/IIIa inhibitor (tirofiban analog).36,37 ATA is a polyanionic and polyaromatic compound having multiple long-term effects on apoptosis,38 angiogenesis,39 and protein interaction,40 which could constitute confounding effects. Nevertheless, its described effects on platelet aggregation are limited to blockade of GpIbα-VWF interactions, and we performed additional experiments to ensure that its effects in the present study are related to neither coagulation inhibition nor parenchymal neuroprotection. Moreover, we confirmed our major findings with Fab from monoclonal antibodies. Altogether, our data unveil GpIbα and VWF as 2 key players mediating arterial occlusion and thrombus stability. GpIbα-VWF inhibitors represent, therefore, an alternative to fibrinolytics and GpIIb/IIIa inhibitors to induce acute reperfusion in patients presenting myocardial infarction or ischemic stroke. Our results, however, suggest that GpIbα-VWF will be efficient only for lysis of recent in situ-formed clots in conditions of high shear rates and in the absence of tPA co-treatment. These conditions would be fulfilled in patients presenting acute myocardial infarction who benefit from facilitated percutaneous coronary intervention or in patients with symptomatic intracranial atherosclerosis who cannot benefit from tPA administration.
The online version of this article contains a data supplement.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 USC section 1734.
Acknowledgments
We thank Dr Anuradha Alahari for help in editing the manuscript.
This work was supported by the Institut National de la Santé Et de la Recherche Médicale, the Conseil Régional de Basse-Normandie, and the Eurostroke-Arise Program (FP7/2007-2013-201024).
Authorship
Contribution: A.L.B., M.G., C.O., and D.V. designed the study and wrote the manuscript; A.L.B., M.G., and S.M.D.L. performed experiments; A.L.B., E.L., and A.M. participated in the development of experimental models; Y.R. supervised platelet aggregation studies and participated in data analysis; S.G. participated in CFD analysis; C.V.D. provided critical reagents and participated in the study design; E.M. supervised the histological studies; and D.V. supervised the study.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: D. Vivien, Inserm UMR-S U919, Université de Caen Basse-Normandie, GIP Cyceron, Bd H. Becquerel, BP5229, 14074 Caen, France; e-mail: vivien@cyceron.fr.
References
Author notes
A.L.B., M.G., C.O., and D.V. contributed equally to this study.