Abstract
Background: Aggressive B-cell non-Hodgkin lymphomas (B-NHL) harboring MYC oncogene abnormalities carry a poor prognosis and frequently relapse after standard front-line therapy. However, little is known about the outcomes following these relapses. Multiple series have suggested that survival is very poor, but detailed descriptions are generally lacking. We sought to examine this under-described yet frequent scenario.
Methods: We performed a retrospective analysis of our center’s experience under an IRB-approved protocol. Patients included those with MYC copy number alteration (CN) or gene rearrangement (GR) as detected by fluorescence in situ hybridization and/or metaphase cytogenetics that did not meet diagnostic criteria for Burkitt lymphoma and who experienced initial treatment failure (ITF): relapse after or unable to achieve complete remission (CR) following upfront therapy. Frequencies of characteristics between groups were compared using a chi-square test. Kaplan-Meier plots and Cox proportional hazard models were used to investigate associations between variables. Clinical follow-up was updated as of June 2014.
Results: We identified 43 pts with relapsed/refractory B-NHL and a MYC gene abnormality, 31 (72%) of which were MYC GR. Of these 31, 18 (64%) were “double-hit” lymphomas with concurrent BCL2 GR, plus another 2 (7%) that also possessed BCL6 GR (“triple-hit”; BCL2/BCL6 data missing from 3). There were no significant differences in overall survival (OS) from ITF between MYC CN, MYC-only GR, and double-/triple-hit (p = 0.73). Additional clinical features at initial diagnosis included median age of 60 years (yrs) (range: 19-72 yrs), stage III-IV disease in 84% (n = 36), elevated LDH in 83% (n = 24; missing data from 14), and B symptoms in 42% (n = 18). Median time from diagnosis until ITF was 6 months (mo) (range: 1-41 mo). Median follow-up from ITF of surviving pts was 8 mo (range: 2-66 mo).
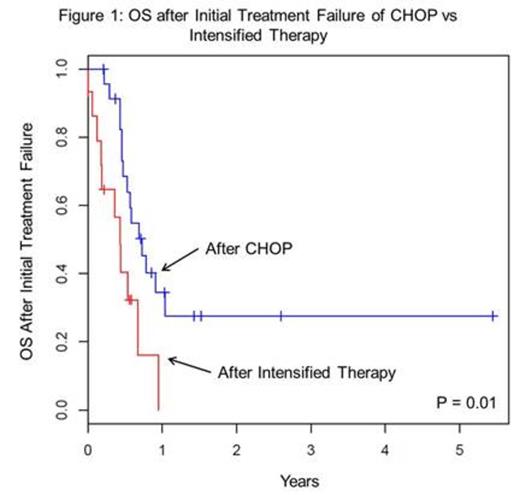
Initial chemotherapy consisted of CHOP in 56% (n = 24), while 35% (n = 15) received a more intense regimen: hyperCVAD (n = 7), dose-adjusted EPOCH (n = 6), or CODOX-M/IVAC (n = 2); all but 1 pt received rituximab (R) with initial therapy. There was no significant difference in the use of CHOP vs intensified regimens with regard to the presence of MYC CN (33% vs 20%, respectively) or MYC GR (67% vs 80%, respectively; p = 0.37). However, 1-yr OS from ITF among those who received CHOP upfront was significantly higher than those who received intensified upfront therapy (34% vs 0%, p = 0.01; Figure 1). CHOP was used more frequently among those who later received ICE salvage (n = 21) than cytarabine (Ara-C)-based salvage (n = 10; 71% vs 40%, respectively; p = 0.09). The rates of subsequent stem cell transplantation (SCT) following ICE and Ara-C were comparable (57% vs 40%, respectively; p = 0.37). R was included in all but 1 pt who received Ara-C. Pts who received ICE had a significantly higher 1-yr OS from ITF than those treated with Ara-C (28% vs 8%, p = 0.01; not shown).
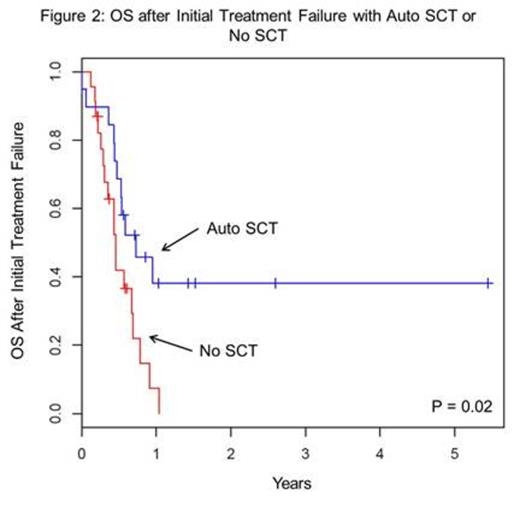
1-yr OS from ITF was significantly better among the 17 pts (40%) who were able to undergo autologous (auto) SCT than the 22 (51%) who did not (38% vs 7%, respectively; p = 0.02; Figure 2). Four pts (9%) received an allogeneic (allo) SCT for their initial salvage SCT and were analyzed separately: Three pts relapsed at 1, 4, and 8 mo post-SCT, while the fourth died in CR 3 yrs later. Rates of SCT use were comparable for pts who initially received CHOP (50%) and those treated with intensified initial therapy (60%; p = 0.54). All but 1 pt in this cohort were treated with the intent of undergoing SCT.
Six pts (14%) had confirmed central nervous system (CNS) relapse, 1 of whom received CNS prophylaxis during initial therapy. Among these, 4 died within 6 mo of ITF and 1 was lost to follow-up with progressive disease only 2 mo after ITF. The remaining pt underwent auto SCT in CR and is alive but with only 1 mo of follow-up.
Conclusions: We have described the outcomes of one of the largest cohorts of relapsed/refractory MYC+ B-NHL reported to date. Though intensified initial therapy may improve outcomes upfront, pts for whom these regimens fail do exceedingly poorly. Pts unable to undergo SCT also have exceptionally dismal outcomes. These subsets of pts in particular should be offered novel therapies in the context of clinical trials. Further, more aggressive initial therapies including consolidative SCT are worth studying since the outcomes after ITF are so poor.
Gopal:Seattle Genetics: Consultancy, Honoraria, Research Funding, Speakers Bureau; Takeda/Millennium: Honoraria, Research Funding, Speakers Bureau; Gilead: Research Funding; Janssen: Research Funding; Pfizer: Consultancy, Honoraria, Research Funding; BMS: Research Funding; Piramal: Research Funding; Teva: Research Funding; Spectrum: Research Funding; BioMarin: Research Funding; Merck: Research Funding; Emergent: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal