Abstract
Langerhans Cell Histiocytosis (LCH) and Erdheim-Chester Disease (ECD) are systemic histiocytic disorders characterized by infiltration of histiocytes in multiple tissues of the body leading to organ compromise. Although the underlying etiology of these conditions has long been enigmatic, recent investigations have determined that LCH and ECD are clonal disorders of myeloid-derived precursor cells with a high frequency of BRAFV600E mutations (40-60% of patients). Moreover, treatment of BRAF-mutant LCH and ECD patients with the BRAF inhibitor vemurafenib has demonstrated dramatic efficacy.
The above data underline the importance of accurately identifying BRAF mutational status in patients with systemic LCH and ECD. Unfortunately, the scant histiocyte content and marked stromal contamination, which are a hallmark of these disorders, make mutation detection in tissue biopsies challenging. Moreover,the propensity of histiocytic lesions to involve difficult to biopsy locations frequently necessitates the use of bone biopsies further limiting the availability of suitable tumor material for BRAF genotyping. We therefore initiated a prospective, blinded, multicenter study of BRAFV600E mutation detection in the cell-free DNA (cfDNA) from plasma and urine of histiocytosis patients to determine the sensitivity/specificity of cfDNA mutation detection compared with tissue biopsy and to track disease burden serially with therapy.
Between January 2013 and June 2014, 30 consecutive patients with ECD (n=25) and LCH (n=5) were enrolled from Memorial Sloan Kettering Cancer Center and MD Anderson Cancer Center. Initial BRAF tissue mutation testing on tissue biopsies was performed by a variety of methods as part of routine care in CLIA-certified molecular diagnostic laboratories. We applied a droplet-digitial PCR assay (RainDrop ddPCR) for quantitative detection of the BRAFV600E mutation in plasma and urine cell-free cfDNA in all patients.
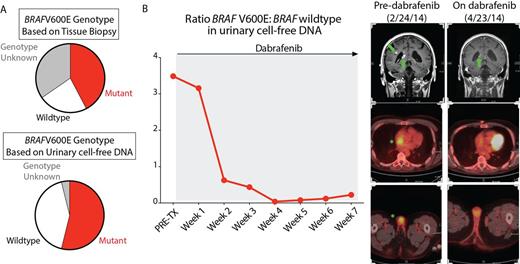
Of 30 patients enrolled, initial tissue BRAFV600E genotyping identified 15 patients to be mutant, 6 patients as wildtype, and 9 as indeterminate. There was 100% concordance between tissue and urinary cfDNA genotype in samples from treatment naïve patients (sensitivity 92.9%, specificity 100%, positive predictive value 100%, negative predictive value 85.7% compared to tissue biopsy detection). Urinary cfDNA analysis identified 5 patients as being BRAFV600E mutant that were not known to have the BRAF mutation previously (Figure A). Subsequent tissue biopsy was performed in 2 of these patients and identified the BRAFV600E mutation, allowing both patients to enroll in an ongoing phase II study of vemurafenib. Results from plasma cfDNA for identifying the BRAFV600E mutation were comparable to urinary cfDNA results
Next examining serial samples during therapy, a significant decrease in the cfDNA BRAFV600E:BRAF wildtype ratio was seen with therapy compared with pretreatment samples (p<0.0001). In all 10 patients treated with a BRAF inhibitor, serial urinary cfDNA analysis revealed progressive decrements in the BRAFV600E allele burden. Weekly serial urinary cfDNA analysis throughout the course of BRAF inhibitor therapy revealed that the decline in mutant cfDNA burden in response to BRAF inhibitors was consistent with radiographic disease improvement (Figure B). Moreover, in at least one patient where successful RAF inhibitor therapy was discontinued for toxicity, urinary cfDNA BRAFV600E burden increased after vemurafenib discontinuation which mirrored radiographic evidence of disease recurrence. Finally, 2 patients treated with the IL1-receptor antagonist anakinra exhibited substantial decreases in BRAFV600E burden, highlighting an effect of IL-1 receptor antagonism in BRAF-mutant histiocytes.
These results indicate that cfDNA BRAFV600E mutational analysis in plasma and urine provides a convenient and reliable method of detecting mutational status and can serve as a non-invasive biomarker to monitor response to therapy in LCH and ECD. Moreover, the dynamic results achieved with cfDNA analysis allowed for monitoring of disease recurrence with treatment cessation. Of note, this study represents the largest prospective study of adult ECD patients to date. Moreover, these data represent the first evidence of the effect of RAF inhibition as well as IL1-receptor antagonism on the BRAF mutant clone in patients with histiocytosis.
Vibat:Trovagene Inc.: Employment. Hassaine:Trovagene Inc.: Employment. Poole:Trovagene Inc.: Employment. Lu:Trovagene Inc.: Employment. Erlander:Trovagene Inc.: Employment. Janku:Trovagene Inc.: Consultancy, Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal