Abstract
Background: MM patients with enhanced immune responses following AHSCT are known to have better long-term disease control. While it has been shown that early responses are mediated by NK cells and alternate inhibitory/stimulatory pathways, including costimulatory molecules, the characterization of these costimulatory molecule profiles in MM patients post AHSCT is not well established. To address this issue, we conducted a pilot study designed to characterize NK cells and function after AHSCT in MM patients through the expression of a cytolytic receptor (CD16), an antibody-dependent cell mediated cytotoxic (ADCC) receptor, and through NK cells functional status based on interferon gamma (IFN-γ) expression as well as the expression of stimulatory (4-1BB) and inhibitory (PD-1 and CTLA-4) molecules. The aims are to characterize the inhibitory and activating costimulatory pathways involved in immune reconstitution after autologous transplantation and to describe changes in the immune signature profile post transplant in response to maintenance therapy and disease status.
Methods: A prospective consecutive case-series of 37 MM patients who underwent AHSCT, median age 59.6 years (36-71.4), were enrolled. Peripheral blood samples were collected pre-AHSCT and again on days 14, 30, 60, 90, 180 and 360 post-AHSCT. IMWG criteria were used for response assessment. NK cells and NK costimulatory molecules were evaluated by flowcytometry using two, six color panels of antibodies. Kruskal-Wallis (KW) test using Graphpad Software was used to plot data/assess differences. The specific time trend for each molecule was modeled using a repeated measures model with unstructured covariance using SAS v9.3. Models were evaluated graphically by taking a scatter plot of data for each molecule and then fit using categorical time, d60-90 response and their interaction as covariates. Since NK expression values were positively skewed, the log-transformed expression + 0.1 (to account for 0) were used as model outcomes. Model outcomes were assessed as absolute number of NK/uL, percent of NK cells, and MFI (mean fluorescence intensity) of NK cell receptor expression. Statistical significance was set at α=0.05 for all analyses.
Results:
| Disease Status . | d60-90 . | d180* . | d360* . |
|---|---|---|---|
| PR or worse (≤ PR) | 18 | 14 | 17 |
| Very Good Partial Response or better (≥VGPR) | 19 | 22 | 19 |
| Disease Status . | d60-90 . | d180* . | d360* . |
|---|---|---|---|
| PR or worse (≤ PR) | 18 | 14 | 17 |
| Very Good Partial Response or better (≥VGPR) | 19 | 22 | 19 |
* One patient - in evaluable
The patients were grouped into ≤ PR (PR + SD+PD: n=18) and ≥VGPR (VGPR +CR: n=19) based on their d60-90 disease status.
· Median percent NK cell was highest (20% of total lymphocytes) at d14 (KW p=0.0001) but returned at d60 to baseline value (11.6% of total lymphocytes).
· The median percent of NK cells post-AHSCT was significantly higher in ≥VGPR group than the ≤ PR group at D 90, 180 and 360 (p= 0.01, <0.01 & 0.02 respectively).
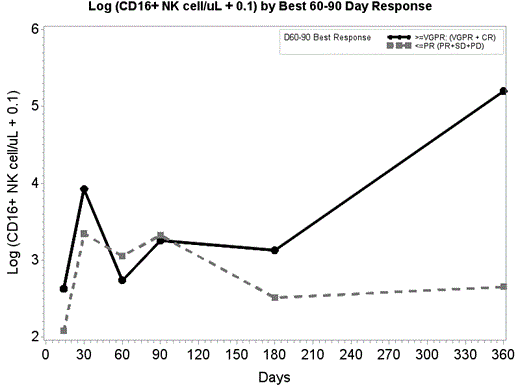
· The total number of NK cells expressing CD16 was not significantly different by group at d14 through d180. However, at d360, CD16 expression that was 13 times higher in ≥VGPR patients than the ≤PR group (p=.0053) (Figure 1).
· The differences in the percentage of NK expressing IFN-γ, were significant between the two groups at d30 (p=.007) and d60 (p=.006). At both time points, ≥VGPR had on average 74% fewer NK IFN-γ cells than ≤PR group.
· There were no significant difference between ≥VGPR and ≤PR groups at different time points, the average expression of PD1 by MFI in ≥VGPR group was 55 % higher than ≤PR group (p=.0126). The average expression of MFI for PD1 + NK cells in all patients were higher at d180 (2.9 fold, p=0.003) and d360 (3.0 fold, p=0.0006) when compared to d14.
· The percent NK/ 4-1BB+ cells were on average 26% lower in ≥VGPR patients than ≤ PR patients (p=0.0156) and the average expression in all patients was significantly higher at d360 (84%, p=0.0115) when compared to d14.
Conclusion: After AHSCT, NK cell recovery was reached at day 60 and was higher overall in the ≥VGPR compared to the ≤PR group. The better disease response group ≥VGPR had higher CD16 expression at d360 but lower IFN-γ at d30 and 60 than the ≤ PR group. Higher CD16 expression (responsible for ADCC killing) and higher NK cells number seem to predict better disease response as well as a low level of IFN-g and 4-1BB expression showing a lower stress response by NK cells.
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal