Abstract
BACKGROUND: Activating mutations of the FLT3 gene are common in acute myeloid leukemia (AML), with approximately 25-30% of cases containing an internal tandem duplication (ITD) in the juxtamembrane domain. The presence of a FLT3-ITD mutation is associated with a poor prognosis, the severity of which, can be modulated by the combination of co-occuring mutations. In animal models, expression of Flt3-ITD by transgenesis, bone marrow transplantation or gene knock-in does not lead to an acute leukemia but a myeloproliferative disease, resembling CMML, suggesting a requirement for additional co-operating mutations (Lee et al, Cancer Cell. 2007, 12: 367). This is supported by in vivo models which demonstrate that the combination of the Flt3-ITD mutation with other genetic lesions leads to the development of an acute leukemia in mice (Chen et al, Genes Dev. 2013, 27: 1974).
AIMS: To identify and characterise novel genes that alter Flt3-ITD induced MPN by using an N-ethyl-N-Nitroso-urea (ENU) mutagenesis strategy in mice with an Flt3-ITD homozygous knock-in background.
METHODS: An autosomal dominant screen for Flt3-ENU co-operating mutations was carried out at the Australian Phenomics Facility by mating ENU-mutagenised male mice homozygous for the Flt3-ITD knock-in allele to homozygous Flt3-ITD females. G1 mice were screened for changes in blood cell parameters indicative of an altered disease state (compared to that induced by Flt3-ITD alone). Mice with blood cell parameters outside two standard deviations of the relevant G1 mean were identified as potential mutation carriers and bred to Flt3-ITD homozygous knock-in mice to test for heritability of the phenotype. Where pedigrees were generated demonstrating heritable phenotypes, multiple affected and unaffected littermates were subject to exome sequencing and analysis to identify a list of candidate gene mutations segregating with the disease phenotype.
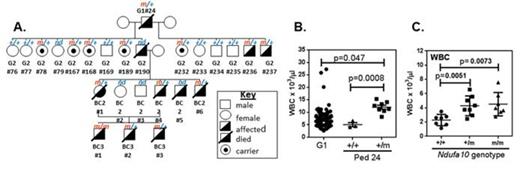
RESULTS: 150 G1 mice were screened, leading to the identification of four pedigrees with heritable phenotypes marked by an exacerbated MPN. Exome sequencing has identified a short list of 3 genes for one pedigree (pedigree 37) that includes a mutation in Neurofibromatosis 1 (Nf1), a gene known to be involved in the induction of juvenile myelomonocytic leukemia and frequently lost in AML (Parkin et al,Clin Cancer Res 2010, 16:4135). In another pedigree (pedigree 24) we identified a mutation in Ndufa10 as the single candidate segregating with the phenotype (Figure 1A-B). Ndufa10 encodes a subunit of the mitochondrial respiratory complex I, which is the first and largest complex in the mitochondrial electron transport chain. Importantly, germline mutations in this gene lead to a complex I deficiency syndrome in humans, indicating that it is a critical subunit of this complex. We hypothesise that mutation of Ndufa10 leads to altered cellular metabolism in hematopoietic stem and progenitor cells which contributes to exacerbation of the MPN, possibly through an alteration in production of reactive oxygen species and a shift in the balance between glycolysis and oxidative phosphorylation. Breeding of the Ndufa10 mutation onto a non Flt3-ITD background shows that action of the mutation is not dependent on the presence of Flt3-ITD, as these mice also have altered blood counts, including increased WBC (Figure 1C).
CONCLUSIONS: It is possible to identify mutations that exacerbate Flt3-ITD induced MPN through mutagenesis and an efficient blood screening strategy. In addition, using this strategy, we have identified novel mutations that act independently of Flt3-ITD to induce changes in the haematological compartment. Translation of these findings to human AML may indicate pathways that will be targets for new and complementary treatments in AML.
A. Flt3-ITD Pedigree 24 indicating affected mice and genotyping for the Ndufa10 mutation. +/+=Ndufa10 wt, m/+=Ndufa10 heterozygous mutant. B. WBC counts for male mice from Pedigree 24 at 15-17 weeks (+/+, n=3; m/+, n=8). WBC from ENU G1 mice are shown as a comparison (G1, n=75). C. WBC counts for male mice on a wildtype C57 background at 16-18 weeks. +/+=Ndufa10 wt (n=7), m/+=Ndufa10 heterozygous mutant (n=8), m/m=Ndufa10 homozygous mutant (7).
A. Flt3-ITD Pedigree 24 indicating affected mice and genotyping for the Ndufa10 mutation. +/+=Ndufa10 wt, m/+=Ndufa10 heterozygous mutant. B. WBC counts for male mice from Pedigree 24 at 15-17 weeks (+/+, n=3; m/+, n=8). WBC from ENU G1 mice are shown as a comparison (G1, n=75). C. WBC counts for male mice on a wildtype C57 background at 16-18 weeks. +/+=Ndufa10 wt (n=7), m/+=Ndufa10 heterozygous mutant (n=8), m/m=Ndufa10 homozygous mutant (7).
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal