Abstract
Introduction: Primary graft failure (PGF) is a potentially life threatening complication following hematopoietic cell transplantation. Patients undergoing cord blood transplantation (CBT) are at higher risk for PGF and also experience delayed hematopoietic recovery. The ability to distinguish between PGF and delayed engraftment is critical for correct clinical management. Limited data exists analyzing the kinetics of engraftment of specific circulating cell lineages within 14 days after double CBT (dCBT). Herein, we investigate the potential for real time immunophenotyping (RTIP) of day 7 and day 14 post-transplant peripheral blood (PB) samples as an effective and economically feasible tool to distinguish PGF versus delayed engraftment.
Methods: Between Feb 2013 and Jun 2014, 26 patients underwent a myeloablative dCBT at our institute. Heparinized PB samples (30 ml) were obtained from patients on days 7 and 14 post-transplant. PB mononuclear cells (PBMCs) were isolated by density gradient separation and total cell number was counted. RTIP with 9-color flow cytometry was performed at each time point using isolated fresh PBMC.
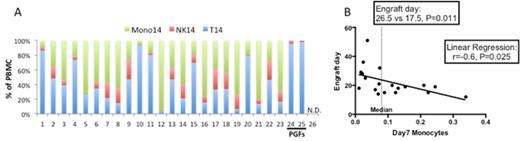
Results: Median time to engraftment was 19 days (range 12-51) in 23 evaluable patients. The remaining 3 patients had no documented hematopoietic recovery by routine daily CBCs. Two patients were declared PGFs when day 21 PB chimerism results demonstrated 100% host T cells followed by confirmation of 0% donor engraftment on day 28. The third patient never had sufficient quantity of circulating cells to obtain RTIP results and died on day 28 with no hematopoietic recovery. Excluding this patient, we were able to quantify the absolute number of PBMC at day 7 (median: 3.1/µl; 95% CI: 2.6-4.6) and day 14 (median: 26/µl; 95% CI: 33-103) in all other patients. There was an increase in the absolute number of PBMC from day 7 to 14 in all patients except the two who experienced PGF. Furthermore, RTIP of day 7 PBMC revealed a predominance of T cells that were donor-derived, while day 14 RTIP of PBMC demonstrated a decreased frequency of T cells and increased frequency of predominantly donor-derived monocytes and NK cells. In contrast to the engrafting patients, the two PGF patients displayed a markedly different pattern in RTIP with minimal evidence of circulating monocytes in the day 14 samples (Fig 1A). Importantly, RTIP demonstrated that day 7 PBMC contained a higher frequency of CD14+CD16- monocytes and CD56brightCD16- NK cells than were infused, suggesting these specific cells were generated de novo and were not representative of cells infused with the graft (data not shown).
Correlations between day 7 or day 14 cell subset numbers and time to engraftment were analyzed. The median absolute number of monocytes at day 7 was 0.075/µl (95% CI: 0.03-0.153). Patients with day 7 monocyte counts above the median demonstrated earlier engraftment than patients below the median (17.5 vs 26.5 days; p= 0.011). Using linear regression with engraftment as the variable of interest and the absolute number of day 7 monocytes as the predictor, the coefficient was 0.6 (95% CI: 0.075- 0.78, P=0.025) (Fig 1B). As expected, higher day 14 monocytes also correlated with earlier engraftment. With respect to the NK cell subset, the absolute number of NK cells at day 7 was not significantly correlated with engraftment time, but day 14 NK cell numbers were predictive of engraftment kinetics. The median number of NK cells at day 14 was 2.78/µl (95% CI: 1.87-5.79). Patients with day 14 NK counts above the median had earlier engraftment than those below the median (16 vs 26.5 days; (P=0.0034). The regression coefficient was 0.5 (95% CI, 0.077- 0.77; P=0.024). Finally, although total T cell numbers at day 7 and day 14 had no correlation with engraftment time, the two evaluable PGF patients had a greater inversion of CD4:CD8 ratio (0.038 & 0.058) than others (median: 0.71, 95% CI: 0.22-1.42) at day 14.
Conclusions: This study provides the first clear evidence that RTIP of PBMC at days 7 and 14 can detect the kinetics of circulating de novo generated monocytes and NK cells and also reflects the in vivo immune environment in patients following dCBT. Importantly, detecting and measuring specific cell subsets using RTIP permits earlier identification of patients at high risk of graft failure versus patients with delayed engraftment and warrants further development as a clinically feasible diagnostic method to guide clinical intervention.
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.
This icon denotes a clinically relevant abstract

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal