Abstract
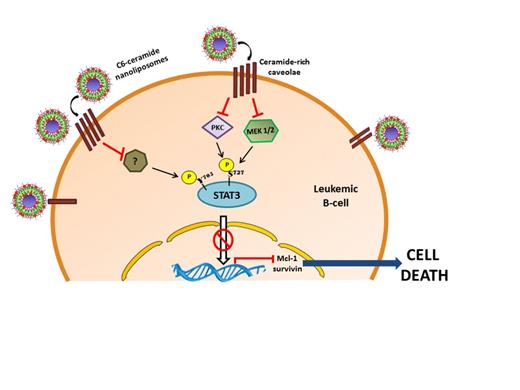
Chronic lymphocytic leukemia (CLL) is the most prevalent form of leukemia in adults in Western countries. Despite high incidence, its pathogenesis is still poorly understood and it remains incurable with current therapies. Ceramide is a tumor suppressor' lipid known to regulate differentiation, senescence and cell cycle arrest. Previous work from our laboratory has demonstrated the therapeutic efficacy of nanoliposomal C6-ceramide in several solid and non-solid tumor models. We have shown that nanoliposomal C6-ceramide selectively induces cell death in CLL cells by targeting the Warburg effect with no detrimental effects on normal peripheral blood mononuclear cells. Additionally, nanoliposomal C6-ceramide treatment results in tumor regression in an in vivo murine model of CLL. However, the mechanism and key molecular players mediating C6-ceramide-induced cell death in CLL remains unknown. Constitutive activation of signal transducer and activator of transcription 3 (STAT3) has been implicated in many cancers. Several reports in the literature have shown that STAT3 is constitutively phosphorylated on serine 727 (S727 p-STAT3) in CLL and STAT3 might be a therapeutic target in this disease. We hypothesize that STAT3 is a target of C6-ceramide in CLL cells.
We first validated STAT3 as a therapeutic target in CLL. STAT3 knockdown in CLL cells induced cell death. C6-ceramide treatment also induces necrotic cell death in these cells. Examination of STAT3 phosphorylation state demonstrates constitutive phosphorylation at S727 and Y705 residue in CLL cell lines and patient cells. C6-ceramide treatment suppressed the levels of Y705 p-STAT3 and S727 p-STAT3, while having no effect on total STAT3 levels. The downregulation of phosphorylation was specific to STAT3 and did not affect the other STATs. C6-ceramide treatment also led to the downregulation of several downstream targets of STAT3 like p21, cyclin D1, Mcl-1 and survivin. Furthermore, the suppression in STAT3 phosphorylation preceded induction of C6-ceramide-mediated cell death in CLL cells, suggesting that this event might be critical for cell death induction. Analysis of the upstream kinases revealed that C6-ceramide-induced suppression of S727 p-STAT3 might be, in part, a consequence of suppression of MEK1/2 kinase and protein kinase C (PKC). Ongoing studies will focus on identifying the specific phosphorylation site(s) that mediates cell death and examine the effect of C6-ceramide on upstream regulators of p-STAT3 in an effort to delineate the molecular mechanism of C6-ceramide-induced cell death in CLL. In conclusion, our data suggests that targeting STAT3 with nanoliposomal C6-ceramide is a potential therapy for CLL. This work is the first body of evidence demonstrating an effect of C6-ceramide on STAT3 phosphorylation.
Kester:Keystone Nano, Inc: Employment, Equity Ownership.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal