Abstract
Background
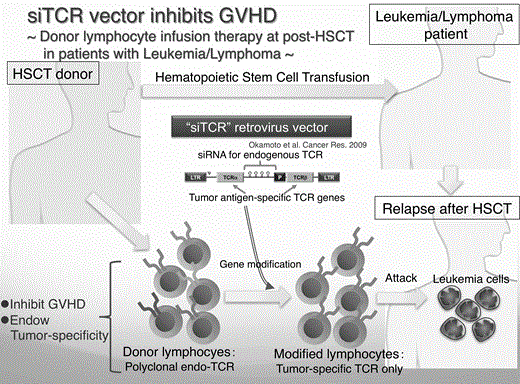
The patients with relapsed hematological malignancy after allogeneic hematopoetic stem cell transplantation (HSCT) can be treated by donor lymphocyte infusion (DLI). However, the efficacy is often limited in association with the development of Graft-versus-Host Disease (GVHD) as a serious adverse event. By the use of the novel retrovirus vector that specifically silences endogenous T cell receptor (TCR) in gene-engineered T cells, here we demonstrate the development of DLI with lymphocytes engineered to express tumor-specific TCR that exhibit increased tumor-specificity in combination with decreased GVHD-inducing potential (Summarized in the figure).
Methods
Human PBMC were transduced with a high affinity TCR specific to a cancer/testis antigen, NY-ESO-1, by the retrovirus vector with siRNA specific to the endogenous TCR (siTCR vector). Resulting TCR gene-transduced T cells were examined for the expression of their endogenous TCR by flow cytometry and for their reactivity to allogeneic LCL by 3H uptake proliferation assay. Immunodeficient NOG mice were innoculated with a NY-ESO-1 expressing human tumor cell line NW-MEL-38, received TCR gene-transduced T cells, and monitored for the tumor growth and the development of GVHD.
Results
Human lymphocytes that were genetically engineered to express a high affinity NY-ESO-1-specific TCR with siTCR vector showed significantly reduced expression of endogenous TCR associated with dramatically diminished reactivity to allogeneic LCL. When adoptively transferred into NOG mice, control non-engineered T cells failed to control the growth of human tumor cells despite the fact that the lymphocytes were allogeneic to the tumor cells and developed lethal GVHD including severe bone-marrow destruction. The development of GVHD was dependent on both the CD4+ and CD8+ T cells suggesting that the simple purification of CD8+ T cells was not a sufficient enough strategy to prevent GVHD. In contrast, adoptive transfer of NY-ESO-1-specific TCR gene-transduced T cells induced complete tumor regression without the development of GVHD.
Conclusions
The results here suggest that the allogeneic T cells transduced with a tumor-specific TCR by siTCR vector showed increased tumor-reactivity with diminished GVHD potential. These T cells will be applicable to the donor lymphocytes infusion therapy after allogeneic stem cell transplantation for the treatment of hematological malignacy. Currently we plan a clinical trial of DLI with siTCR technology for the patients with relapsed NY-ESO-1 positive adult T cell leukemia/lymphoma after HSCT.
Ikeda:Takara Bio Inc.: Research Funding. Kawamura:Takara Bio Inc.: Employment. Imai:Takara Bio Inc.: Research Funding. Okamoto:CDM Center, Takara Bio Inc.: Employment. Mineno:Takara Bio Inc.: Membership on an entity's Board of Directors or advisory committees. Takesako:Takara Bio Inc.: Membership on an entity's Board of Directors or advisory committees. Shiku:Takara Bio Inc.: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal