Abstract

Background: The use of haploidentical hematopoietic cell transplantation (HCT) using post- transplant cyclophosphamide (PT-Cy), calcineurin inhibitors (CNI) and mycophenolate as graft-versus-host disease (GVHD) prophylaxis is rapidly increasing in patients (pts) lacking suitable HLA-matched donors. Herein we compare outcomes of haploidentical HCT using this GVHD prophylaxis with 8/8 allele-level MUD HCT.
Methods: Included are 917 adult (>18) lymphoma pts who underwent allogeneic HCT between 2008 and 2013. All pts received non-myeloablative or reduced-intensity conditioning regimens. The study cohort was divided into 3 groups; haploidentical (n=185), MUD without (w/o) antithymocyte globulin (ATG; n=491) and MUD with (w/) ATG (n=291). The primary end-point was overall survival (OS). Secondary endpoints included cumulative incidence (Cum-Inc) of acute GVHD, chronic GVHD, non-relapse mortality (NRM), relapse/progression (rel/prog) and progression-free survival (PFS). The study had an 83% power to detect an 11% difference in OS.
Results: The baseline characteristics are shown in Table 1. Pts in the haploidentical group received conditioning with Flu/CY/2Gy TBI and PT-Cy + CNI and mycophenolate as GVHD prophylaxis, while the two MUD cohorts received fludarabine-based (+ an alkylator or 2GyTBI) conditioning and CNI-based GVHD prophylaxis. Graft source was bone marrow in 93% of the haploidentical pts and peripheral blood in 94% and 91% of MUD w/o ATG and MUD w/ ATG pts, respectively.
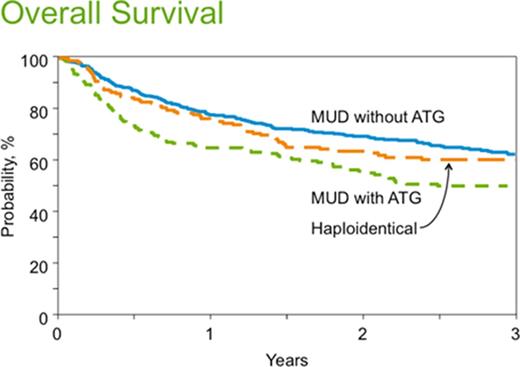
The 28-day neutrophil recovery and platelet recovery were 94%, 97%, 97% (p=0.32) and 63%, 89%, 84% (p<0.001) in the haploidentical, MUD w/o ATG and MUD w/ ATG groups respectively. Cum-Inc of grade II-IV acute GVHD at day100 and chronic GVHD at 1 year was 27%, 40% and 49% (p=0.07) and 13%, 51% and 33% (p<0.001) in the haploidentical, MUD w/o ATG and MUD w/ ATG groups, respectively. On multivariate analysis (MVA) higher risk of chronic GVHD was seen in MUD w/o ATG (RR=5.85, 95%CI 3.96-8.64; p<0.0001) and MUD w/ ATG (RR=3.64, 95%CI 2.37-5.59; p<0.0001) groups relative to the haploidentical cohort. The 3 year NRM was 17%, 22% and 26% in the haploidentical, MUD w/o ATG and MUD w/ ATG groups (p=0.08), respectively. On MVA a trend towards higher NRM was noted in MUD w/ ATG cohort, RR 1.54 (95%CI 0.98 - 2.41, p=0.06), relative to the haploidentical group. Among the haploidentical, MUD w/o ATG and MUD w/ ATG cohorts the 3 year Cum-Inc of rel/prog was 36% vs. 28% vs. 36%, PFS was 47% vs. 49% vs. 38% and OS was 60%, 62% and 50% (Figure), respectively. MVA demonstrated no significant difference between the three groups in terms of rel/prog (p=0.27) and PFS (p=0.07). Compared to the haploidentical group, the two MUD groups did not have a significantly different mortality risk (inverse of OS; p>0.05), but compared to MUD w/ ATG, the MUD w/o ATG pts had a reduced mortality risk (RR=0.67; p=0.001). We tested for a transplant center effect on survival and found none.
Conclusion: With lower-intensity conditioning regimens the early (up to 3 years) survival outcomes are comparable between conventional MUD transplants (w/ or w/o ATG) and haploidentical HCT with PT-Cy approach. Chronic GVHD was significantly lower with haploidentical HCT. Prospective, randomized confirmation of these findings is necessary before wide spread adoption of haploidentical HCT over MUD transplants in lymphomas.
| . | Haploidentical N=185 (%) . | MUD w/o ATG N=491 (%) . | MUD w/ ATG N=241 (%) . | p-value . |
|---|---|---|---|---|
| Age @ HCT, median (range) | 55 (18-75) | 55 (19-74) | 55 (20-73) | 0.13 |
| Male sex | 118 (64) | 301 (61) | 163 (68) | 0.25 |
| White race | 149 (81) | 469 (96) | 227 (94) | <0.001 |
| KPS ≥ 90 | 145 (78) | 311 (63) | 153 (63) | <0.001 |
| HCT-CI≥3 | 55 (30) | 175 (36) | 88 (37) | <0.001 |
| Histology NHL Hodgkin | 139 (75) 46 (25) | 386 (79) 105 (21) | 193 (80) 48 (20) | <0.001 |
| Months from diagnosis to HCT, median (range) | 31 (<1-255) | 34 (<1-342) | 32 (4-460) | 0.19 |
| High LDH @ HCT | 16 (31) | 31 (33) | 11 (27) | <0.001 |
| BM +ve @ HCT | 6 (12) | 5 (5) | 2 (5) | 0.35 |
| Extranodal disease @ HCT | 18 (35) | 20 (21) | 6 (15) | 0.11 |
| Prior lines of therapy, median (range) | 3 (1-7) | 3 (1-12) | 3 (1-8) | 0.41 |
| Remission @ HCT CR PR Refractory Untreated / missing | 72 (39) 99 (54) 10 (5) 4 (2) | 215 (44) 215 (44) 55 (11) 6 (1) | 100 (41) 96 (40) 40 (17) 5 (2) | 0.02 |
| Disease Risk Index Low Intermediate High | 45 (24) 126 (68) 13 (7) | 199 (41) 263 (49) 48 (10) | 75 (31) 129 (54) 37 (15) | <0.001 |
| Median follow-up, months (range) | 36 (5-73) | 35 (4-74) | 35 (<1-75) |
| . | Haploidentical N=185 (%) . | MUD w/o ATG N=491 (%) . | MUD w/ ATG N=241 (%) . | p-value . |
|---|---|---|---|---|
| Age @ HCT, median (range) | 55 (18-75) | 55 (19-74) | 55 (20-73) | 0.13 |
| Male sex | 118 (64) | 301 (61) | 163 (68) | 0.25 |
| White race | 149 (81) | 469 (96) | 227 (94) | <0.001 |
| KPS ≥ 90 | 145 (78) | 311 (63) | 153 (63) | <0.001 |
| HCT-CI≥3 | 55 (30) | 175 (36) | 88 (37) | <0.001 |
| Histology NHL Hodgkin | 139 (75) 46 (25) | 386 (79) 105 (21) | 193 (80) 48 (20) | <0.001 |
| Months from diagnosis to HCT, median (range) | 31 (<1-255) | 34 (<1-342) | 32 (4-460) | 0.19 |
| High LDH @ HCT | 16 (31) | 31 (33) | 11 (27) | <0.001 |
| BM +ve @ HCT | 6 (12) | 5 (5) | 2 (5) | 0.35 |
| Extranodal disease @ HCT | 18 (35) | 20 (21) | 6 (15) | 0.11 |
| Prior lines of therapy, median (range) | 3 (1-7) | 3 (1-12) | 3 (1-8) | 0.41 |
| Remission @ HCT CR PR Refractory Untreated / missing | 72 (39) 99 (54) 10 (5) 4 (2) | 215 (44) 215 (44) 55 (11) 6 (1) | 100 (41) 96 (40) 40 (17) 5 (2) | 0.02 |
| Disease Risk Index Low Intermediate High | 45 (24) 126 (68) 13 (7) | 199 (41) 263 (49) 48 (10) | 75 (31) 129 (54) 37 (15) | <0.001 |
| Median follow-up, months (range) | 36 (5-73) | 35 (4-74) | 35 (<1-75) |
Armand:Infinity: Consultancy, Research Funding; Merck: Consultancy, Research Funding; BMS: Research Funding; Sequenta, Inc.: Research Funding. Smith:Celgene: Consultancy; Pharmacyclics: Consultancy. Sureda:Seattle Genetics Inc.: Research Funding; Takeda: Consultancy, Honoraria, Speakers Bureau.
Author notes
Asterisk with author names denotes non-ASH members.

This icon denotes a clinically relevant abstract

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal