Abstract
During erythropoiesis, heme production becomes dramatically increased to support the production of oxygen-carrying hemoglobin. Heme biosynthesis requires an eight-step enzymatic cascade culminating with the insertion of ferrous iron into protoporphyrin IX by the mitochondrial enzyme ferrochelatase (FECH). This last step is the rate-limiting step in red blood cells (RBCs) and represents a critical regulatory juncture in RBC biology and human blood diseases. Most notably, in humans, FECH mutations are strongly associated with a disorder called erythropoietic protoporphyria (EPP) that typically presents with mild anemia and porphyria resulting from inadequate heme production and upstream accumulation of porphyrin intermediates, respectively. Disease severity can be highly heterogeneous, suggesting that additional modulators of FECH likely exist. However, despite its implications in EPP and other human blood disorders, very little is known regarding such regulatory mechanisms.
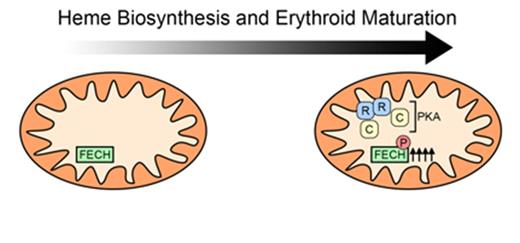
Here, using complementary biochemical and genetics approaches, we identify FECH as a direct physiologic target for protein kinase A (PKA) phosphorylation during erythroid maturation. Quantitative proteomics revealed that PKA becomes enriched in mitochondria of differentiating murine erythroid cells. Phosphorylation of a highly conserved Thr116 in the catalytic domain of human FECH by PKA increases its activity resulting in elevated hemoglobinization of erythroblasts. We examined the importance of this phosphorylation in vivo by taking advantage of CRISPR/Cas9-mediated genome editing to knock-in the analogous Thr115Ala substitution into the endogenous Fech gene in murine RBCs. This approach allowed us to examine FECH function in a more physiologic context. Murine erythroid cells harboring only Thr115Ala Fech show reduced ferrous iron incorporation into protoporphyrin IX and, consequently, compromised hemoglobinization. In primary murine erythroid cells, we demonstrate that a distinct PKA gene expression signature is induced early in erythropoiesis. This suggests that the PKA pathway is engaged by physiologic signaling mechanisms during RBC development.
Together, our results support a model where in maturing RBCs, PKA becomes enriched in the mitochondria where it phosphorylates FECH. Phosphorylation of FECH is required for full activity to support elevated heme biosynthesis and hemoglobin production. Our data also uncover PKA as a novel mechanism that links early erythroid signaling pathways to regulate heme biosynthesis and suggests that porphyrin accumulation can result not only from defective porphyrin transport but also aberrant cell signaling.
Bauer:Biogen: Research Funding; Editas Medicine: Consultancy.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal