Abstract
A toxicity reduced conditioning regimen containing Treosulfan (Treo), fludarabine (Flu), thiotepa for high risk Thal Major (TM) has been used since 2009 at our centre that has significantly improved transplant outcomes of these patients compared to the historical cohort of patients receiving busulfan/ cyclophosphamide based myeloablative regimen (Mathews et al, 2013). Limited knowledge is available on the pharmacokinetics (PK), pharmacogenetics (PG) and pharmacodynamics of fludarabine and treosulfan, especially in non-malignant hematological disorders like TM. We describe here the PK of Flu and Treo in patients with TM undergoing HSCT, the factors influencing the inter-individual variability in PK and the role of these factors on HSCT outcome.
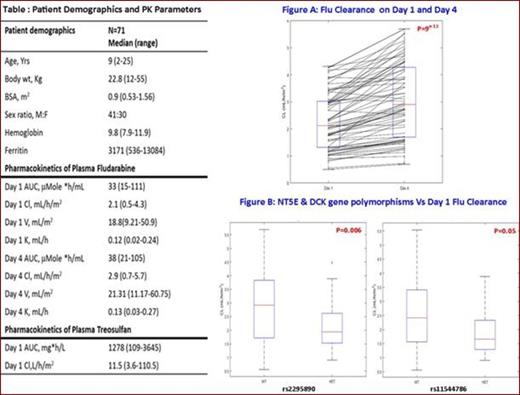
Seventy one patients diagnosed with TM undergoing HSCT with Flu/Treo based conditioning regimen between January 2012 and January 2015 were included (Table: Patient demographics). Selected functional polymorphisms in the NT5E, DCK, hENT1 and GST genes that are involved in fludarabine or treosulfan metabolism were screened. All patients received Flu 40mg/m2/day x 4 days as an 1hr infusion on days 1 and 4 and Treo as 14g/m2/day x 3 days at the rate 5g/hr. Plasma was separated from the peripheral blood collected at predetermined time points after the infusion of Flu and Treo PK analysis. Plasma Flu was analyzed using a LC-MS/MS method and the concentration was expressed as mMole/ml while Treo was analyzed using a HPLC-RI method and concentration was expressed as mg/L. Flu and Treo PK was estimated using nonlinear mixed effects modeling via Monolix 4.3.3. The covariates tested for both PK were: age, sex, body weight, BSA, ferritin, and polymorphisms in NT5E, hENT1, dCK and GST genes. The PK parameters AUC, CL, V and k were estimated on day 1 for Treo and on day 1 and day 4 for Flu (Table). The influence of Flu and Treo PK and PG on graft rejection, early transplant related mortality (TRM) & chimerism status was estimated using logistic regression analysis.
Wide inter-individual variation in Flu and Treo PK was noted (7 and 9 fold Vs 5 and 8 fold respectively for Day 1 & 4 Flu AUC & Cl; 33 & 31 fold variation in Treo AUC and Cl) (Table). Flu CL was significantly higher on day 4 compared to day1 (Figure A). The variation in Flu PK was explained by genetic variants in NT5E and dCK. Patients having variant genotype for the SNPs in NT5E (rs2295890) and dCK (rs11544786) showed significantly lower plasma Flu clearance compared to those with wild type genotype (p=0.006 & p=0.05 respectively) (Figure B). This is consistent with our previous report in patients with aplastic anemia undergoing HSCT (Mohanan et al. 2014; Blood: 124 (21)). None of the genetic variants in the GST genes explained the variation in Treo PK.
Day21 mortality was seen in 6/71 patients (8.5%) and graft rejection in 3/66 evaluable patients (4.5%). Analysis of the influence of PK and PG variables on transplant outcome showed significantly high first dose Flu AUC to be associated with D21 mortality upon Univariate analysis (median 42.5, range 32.1-63.7 compared to 31.8, range 15.2-111 mMole*h/mL, in those with and without TRM respectively; p=0.043); none of these parameters were significantly associated with graft rejection or mixed chimerism. There was no association between Treo PK parameters and graft rejection or TRM. The influence of Flu and Treo PK on regimen related toxicity is yet to be evaluated. The lack of the influence of PK on transplant outcome could be due to lower incidence of rejection and TRM in this cohort. Further analysis in a larger cohort of patients will be done once we enroll more patients for PK analysis.
Our results demonstrate that Flu PK is influenced by genetic variants in NT5E and dCK, the enzymes involved in Flu biotransformation. The relationship between high-plasma Flu exposure and TRM and given the fact that multiple factors influence TRM, we can extrapolate that the plasma Flu AUC may be a surrogate marker of overall preparative regimen intensity as reported previously (Long-Boyle et al, Bone Marrow Transplant, 2011). The lack of association of genetic variants in GST genes in explaining the inter-patient variability in treosulfan exposure suggests the involvement of other drug metabolizing genes on treosulfan PK. We are currently evaluating the role of genetic variants in a large panel of drug metabolizing genes on explaining this inter-individual variability in Treo PK.
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal