Abstract
β thalassemia carriers are usually symptom free with microcytic hypochromic red cells and a raised HbA2 level. However, an increased output of α globin through co-inheritance of extra α globin genes, converts a typically asymptomatic β thalassemia carrier state to that of thalassemia intermedia. We describe 3 families with 3 unique head-to-tail duplications of the a-globin cluster in which all the probands have thalassemia intermedia ranging from moderately severe anemia with splenomegaly, to transfusion-dependence.
In Family 1, both father (Chinese) and son (Chinese and Anglo-Saxon English) were heterozygous for the HBB:c.316-197C>T β thalassemia variant but had moderately severe anemia (Hb 67 to 91 g/L) with splenomegaly; they were both transfusion-independent. In Family 2, the father (Syrian) had normal hematology, while mother (Iraqi) had a hematologic profile (Hb 110 g/L, RBC 5.68x1012/L, MCH 19.4 pg, MCV 58.9 fL and HbA2 4.8%) typical of heterozygous β0 thalassemia. The proband presented at age 5 years with severe anemia precipitated by an infection, that needed a blood transfusion. She continued to need intermittent blood transfusion while an older sister with a hematologic profile of Hb 75 g/L, RBC 3.04x1012/L, MCH 19.6 pg, MCV 65.6 fL, and mild spenomegaly, remained transfusion-independent. In Family 3 (Greek Cypriot), both parents were asymptomatic; the father was heterozygous for the HBB:c.93-21G>A β thalassemia variant with a normal a globin genotype (aa/aa), and the mother had normal HbA2 levels. In contrast, both their daughter and son who had inherited father's β thalassemia, had moderately severe anemia and needed intermittent blood transfusion. In all 3 families, MLPA suggested duplication of the whole alpha globin cluster but could not differentiate the different duplications.
Next generation sequencing using Agilent SureSelect bait capture, targeted sequence analysis to the two globin loci. Sequence alignment to the reference sequence was performed using NextGene (SoftGenetits, USA). Analysis of the β globin gene sequence identified the β thalassemia-causing variant in each family. Comparison of the sequence coverage across the a loci on chromosome 16 between each case and normal controls, showed that where duplications had occurred, there was proportionally more sequence captured, similar to SNP or CGH array analysis. At the point where the sequence coverage increased, a duplication breakpoint was suspected, and the aligned sequence reads were examined in detail. In Family 1, individual sequence reads matched part of the reference sequence, BLAT query in UCSC showed that the two halves of the read aligned at either end of the duplication, indicating they were sequences that spanned the head-to-tail breakpoint. This was confirmed by gap-PCR and Sanger sequence analysis. In the other two families, breakpoints were identified within repetitive regions which could not be captured by the baits and were therefore not covered by the captured sequence. The high resolution of the coverage map allowed precise characterisation of the duplications by gap-PCR and Sanger sequencing analysis of the breakpoint amplicons.
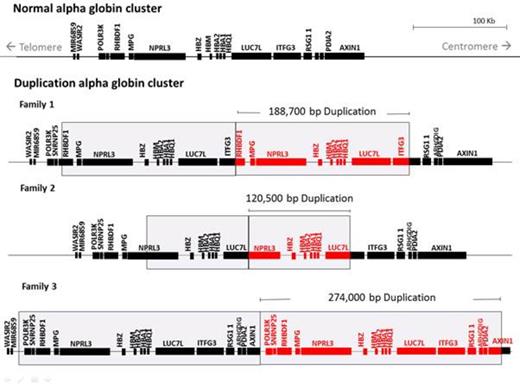
Duplication of the a globin cluster was encompassed in 188.7 kb in family 1, 120.5 kb in family 2, and 274 kb in family 3 (Figure 1). Both father and son in Family 1 were heterozygous for the duplicated a globin cluster (aa/aa, aa) and HBB:c.316-197C>T mutation. Both siblings in Family 2 were heterozygous for mother's β thalassemia c.135delC variant and father's duplicated a globin cluster. In family 3, the mother had a 3.7 kb a deletion variant and a duplicated a globin cluster (-a3.7/aa, aa), a total of 5 a globin genes. Both the daughter and son had inherited mother's duplicated a globin cluster with father's β thalassemia variant.
These families clearly show that a duplicated a globin cluster does not have a discernible phenotype on its own but is readily detectable when co-inherited with a β thalassemia variant. In all 3 cases, the a globin cluster duplications are in a head-to-tail orientation and occurred in repeats. These have most likely formed by non-homologous recombination events involving repetitive Alu sequences interspersed throughout the region. Whether these events occur more commonly in this region or if the region tolerates these changes better, allowing them to accumulate, remains to be resolved.
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal