Abstract
Introduction: [18 F]-Fludarabine is a promising novel positron emission tomography (PET) radiotracer for lymphoid malignancies. The rationale for its development is the high selectivity of fludarabine uptake within lymphoid cells, irrespective of their cycle activity, and the fluorine atom within the molecule, which is replaced by [18 F] and leads to a positron emitting biomarker. Pre-clinical studies (Dhilly et al, Mol Imaging Biol 2014,16:118-26 ; Hovhannisyan et al, EJNMMI Res 2015,5:23) showed a marked tumor uptake in lymphoma-bearing mice. The aim of this study was to describe anatomical sites with abnormal [18F]-fludarabine uptake in DLBCL patients. This study was designed as a clinical proof of concept.
Methods: [18F]-Fludarabine was produced according to a method already described (Guillouet et al, Mol Imaging Biol 2014,16:28-35). [18F]-Fludarabine PET/CT (Discovery RX VCT 64, GE Healthcare) was performed in 5 histologically confirmed and treatment naïve DLBCL patients (65 ± 8 years old). Successive partial body PET scans (skull vertex to mid-thigh) were acquired for 250 min after intravenous injection of [18F]-fludarabine with an activity of 4MBq/kg. PET images were analyzed drawing VOIs over the uptake sites on a late scan and projected onto all co-registered scans of the same subject. The intensity of tracer uptake was evaluated with standardized uptake value (SUVmax). The performance of [18F]fludarabine PET/CT was visually compared with conventional assessments (high-resolution CT) and [18F]-FDG PET.
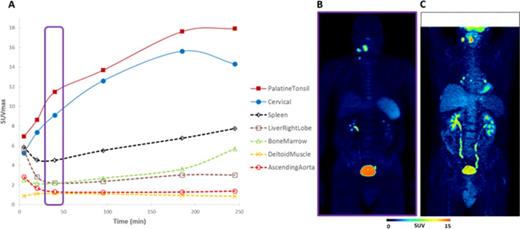
Results: CT and [18F]-FDG PET staging of the 5 patients was I to IV. No adverse event was recorded during and after the procedure. In all 5 patients, the uptake of [18F]-fludarabine coincides with sites expected to be involved following conventional staging. SUVmax were significantly higher in involved sites in comparison with non-lymphoma sites (Figure 1). As previously demonstrated in an animal model, we found no uptake in the cardiac muscle and brain in contrast to [18F]-FDG PET. Bone marrow was histologically normal in the 5 patients and [18F]-fludarabine PET displayed no hypermetabolism. A progressive splenic uptake (x4 to x8 at 250 min) was observed in every patient. The possibility of an unexpected splenic infiltration appears to be a better explanation than uptake by physiologically normal lymphoid tissue. Comparison of [18F]-FDG and [18F]-fludarabine PET showed discrepancies in 2 patients. The first had bilateral hilar [18F]-FDG uptakes (Figure 1C), not present in [18F]-fludarabine PET, which persisted on [18F]-FDG PET after completion of the treatment and disappearance of all suspected pathological sites. This pattern suggests a non-specific [18F]-FDG hypermetabolism. The second patient had a right testis positive [18F]-FDG PET, not evident with [18F]-fludarabine PET. Histology of the testis confirmed the presence of NHL. The interpretation of this would require further extensive data.
Conclusion: [18F]-Fludarabine PET/CT appears to be a promising tool to diagnose discordance and follow-up NHL. These preliminary results showed a clear specificity of this novel radiotracer for lymphoma tissues and support the development of this innovative biomarker for lymphoproliferative diseases. Studies for a more detailed comparison with [18F]-FDG PET are in progress.
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.

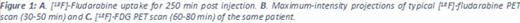
This feature is available to Subscribers Only
Sign In or Create an Account Close Modal