Abstract
INTRODUCTION
The prognosis for children with refractory acute myeloid leukemia (AML) treated with chemotherapy is dismal and data on the outcome after allogeneic haematopoietic stem cell transplant (SCT) are scanty with reported leukemia free survival (LFS) rates of 10-20%. Thus there is significant controversy about whether SCT is appropriate in such patients (pts). We performed a retrospective, national study to analyse outcomes and prognostic factors for children undergoing SCT for refractory AML in the UK.
METHODS
A retrospective analysis of all pts <18 years of age reported to the BSBMT registry who received their first allogeneic SCT between 2000-2012 for refractory AML (ie >5% blasts in the bone marrow (BM) or proven extramedullary disease (EM) was performed. Source data verification (SDV) was performed to ensure pts were indeed refractory. The primary end-point was 5 year LFS. Secondary end-points were Relapse Rate (RR), Treatment Related Mortality (TRM), Graft Versus Host Disease (GVHD) and Overall Survival (OS). The Kaplan Meyer method was used to estimate survival data and Fisher's exact and Mantel-Cox Log Rank tests were used to compare disease- transplant- and survival-related variables.
RESULTS
Following SDV, a total of 44 pts from 13 centres were included in the study. The median age at SCT was 11.5 yrs and the median number of prior lines of chemotherapy was 3. The median time from diagnosis to SCT was 197 days. 23 pts had primary refractory AML and 21 had relapsed refractory AML. 12 pts showed adverse risk cytogenetics, 26 standard risk and 6 favourable. EM disease was documented in 5 pts. 42 children had >5% myeloid blasts in the BM immediately prior to conditioning and refractory disease was confirmed by cytogenetics/molecular genetics in 23. 2 pts were in BM remission but had frank EM disease. 38 pts (86%) received myeloablative conditioning (14 TBI based) and 6 (14%) had reduced intensity conditioning (RIC). In vivo T cell depletion was used in 25 pts. 15 pts (35%) were transplanted from an HLA identical family donor, 15 from a matched unrelated donor and 14 (32%) a mismatched donor. BM was used as the stem cell source in 18 (41%), peripheral blood in 20 (46%) and cord blood in 6 cases (14%). Median follow up was 4 years 10 months.
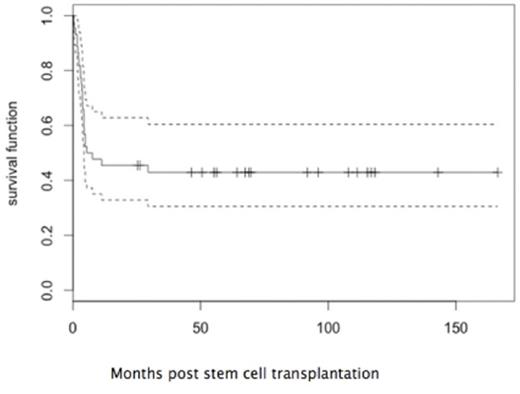
5 pts never achieved engraftment and had disease progression. The remaining 39 pts engrafted at a median of 15 days post-SCT. 30 pts (68%) achieved a complete remission (CR) following SCT. TRM at 1 year was 18% (5 infections, 1 cardiac failure, 1 GVHD-related). Acute GVHD occurred in 23 pts and was severe (grade ≥III) in 8 (19%). The incidence of chronic GvHD was low (1 limited, 2 extensive). Relapse was the major cause of treatment failure and occurred in 17 pts (39%) at a median 2.3 months post SCT. At last follow-up, 18 pts remain alive and in continuous complete remission (CCR). In this cohort, the 5 year OS and LFS were both 43% (95%CI 31-61%) (Figure1). Outcomes in pts with primary refractory disease (9/23, 39% in CCR) and those with relapsed refractory AML (9/21, 43% in CCR) were equivalent. Outcome for pts with cytogenetic confirmation of refractory disease was not statistically different (7/23, 30% in CCR) from the overall group. Pts transplanted with ≤30% blasts in the BM had improved outcomes (5-year LFS 52% vs 27%, p= 0.05). Likewise, the development of aGVHD of any grade was associated with a significantly better LFS (5-year LFS 56% vs 30%, p= 0.05). Cytogenetics including monosomy 7 (n=7) and molecular risk classification did not translate into a significant prognostic factor for relapse. Since RIC was used in only 6 pts, the impact of the intensity of conditioning cannot be determined.
CONCLUSIONS
This is the largest series of outcomes for SCT for refractory paediatric AML reported to date. Our data indicate that for selected pts, particularly those with a lower disease burden, SCT offers a realistic chance of salvage in both primary refractory and relapsed refractory AML (5 year LFS 43%) with acceptable toxicity. The association of aGVHD with improved LFS suggest a possible role in engineering a graft-versus-leukemia effect in this patient group.
Leukemia-free survival for pediatric patients with refractory acute myeloid leukemia undergoing allogeneic stem cell transplantation.
Leukemia-free survival for pediatric patients with refractory acute myeloid leukemia undergoing allogeneic stem cell transplantation.
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal