Key Points
T cells transduced with a CD5 CAR demonstrate limited and transient fratricide and expand ex vivo.
CD5 CAR T cells eliminate T-ALL blasts in vitro and control disease progression in xenograft T-ALL mouse models.
Abstract
Options for targeted therapy of T-cell malignancies remain scarce. Recent clinical trials demonstrated that chimeric antigen receptors (CARs) can effectively redirect T lymphocytes to eradicate lymphoid malignancies of B-cell origin. However, T-lineage neoplasms remain a more challenging task for CAR T cells due to shared expression of most targetable surface antigens between normal and malignant T cells, potentially leading to fratricide of CAR T cells or profound immunodeficiency. Here, we report that T cells transduced with a CAR targeting CD5, a common surface marker of normal and neoplastic T cells, undergo only limited fratricide and can be expanded long-term ex vivo. These CD5 CAR T cells effectively eliminate malignant T-cell acute lymphoblastic leukemia (T-ALL) and T-cell lymphoma lines in vitro and significantly inhibit disease progression in xenograft mouse models of T-ALL. These data support the therapeutic potential of CD5 CAR in patients with T-cell neoplasms.
Introduction
Prognosis for patients with primary chemotherapy-refractory or relapsed lymphoid malignancies remains poor.1-7 Chemotherapy treatment, although greatly improving disease-free survival, may result in significant short-term and long-term toxicities, substantiating the need for novel targeted therapies. Recent studies in patients with B-lymphoid malignancies have demonstrated the remarkable potency of chimeric antigen receptors (CARs) that can redirect T cells to the CD19 antigen present on normal and malignant B cells with complete response rates of >90% even in patients with refractory or relapsed disease.8-10 Such response rates, however, are accompanied by elimination of the normal B-cell population. The concern that loss of normal T lymphocytes would produce a more profound immunodeficiency than loss of B cells has impeded parallel approaches that would treat T-cell malignancies by targeting an antigen consistently expressed by both normal and malignant T cells. Moreover, any CAR T cell that targeted a tumor antigen shared between normal and malignant T cells might lead to fratricide of CAR T cells, thus jeopardizing their therapeutic efficacy.
CD5 is one of the characteristic surface markers of malignant T cells, present in ∼80% of T-cell acute lymphoblastic leukemia (T-ALL) and T-cell lymphoma.11,12 In addition, CD5 is often expressed in B-cell lymphoma. Expression of CD5 by normal cells is restricted to components of the immune system: thymocytes, peripheral T cells, and a minor subpopulation of B lymphocytes (B-1 cells).13,14 CD5 is a negative regulator of T-cell receptor (TCR) signaling15-17 implicated in promoting survival of normal and malignant human lymphocytes,18-21 and was validated as a tumor target antigen in earlier clinical trials using immunotoxin-conjugated CD5 antibodies.22-24 These clinical trials demonstrated efficient depletion of malignant T cells in patients with cutaneous T-cell lymphoma and T-ALL.
We hypothesized T cells expressing a novel CD5-targeting CAR could mount a sustained anti-CD5 response. We found that the biological properties of the CD5 antigen allow CD5 CAR T cells to produce potent antitumor activity against T-ALL and T-lymphoma cells in vitro and in vivo while limiting T-cell fratricide and sparing responses to viral antigens.
Materials and methods
CD5 CAR design
Anti-CD5 single chain variable fragment (scFv) was created using commercial gene synthesis and cloned into a backbone of a 2nd generation (κ chain-specific) CAR.25 For the in vivo studies, the CH2 portion of the immunoglobulin (Ig)G Fc spacer was removed. A truncated version of CD5 CAR (ΔCD5 CAR) was created by deleting cytoplasmic domains. Transduction and expansion of T cells was performed as described before.26 Efficiency of transduction routinely exceeded 90%. For some experiments, activated T cells were transduced with a green fluorescent protein (GFP)-encoding retrovirus to obtain GFP+ autologous T cells.
Sequential killing assay
CD5 CAR T cells were plated with GFP+ Jurkat cells in 96-well flat bottom plates at a 1:2 effector to target ratio (E:T) (25 000 CAR T and 50 000 Jurkat cells per well in cytotoxic T lymphocyte media). Some 72 hours later, cells were collected and counted with flow cytometry using CountBright counting beads and 7-AAD. CD5 CAR T cells were then replated and reconstituted with fresh Jurkat-GFP cells to restore initial E:T ratio. Cell counting and replating was repeated after 72 hours with a total of 4 iterations. No exogenous cytokines were added.
Statistical analysis
Unpaired 2-tailed Student t test was used to determine statistical significance. Statistical analysis of the Kaplan–Meier survival curves was done using log rank (Mantel–Cox) test. Data are presented as mean ± standard deviation (SD) unless noted otherwise. All P values were calculated with Prism 6 (GraphPad). Additional methods are listed in supplemental Methods, available on the Blood Web site.
Results
CD5 CAR-transduced T cells expand and downregulate CD5 from cell surface
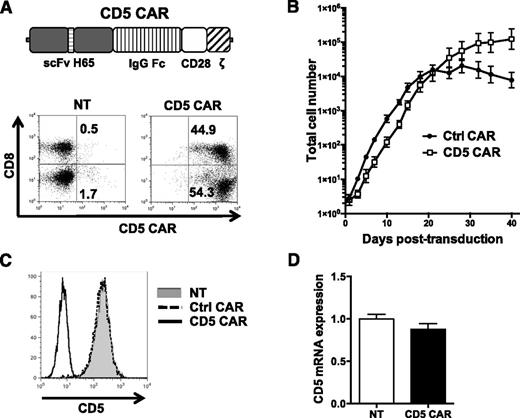
We designed a CD5 CAR consisting of anti-CD5 scFv (derived from clone H6524 ), an IgG Fc spacer, and intracellular signaling domains from CD28 and TCR ζ chain (Figure 1A). After transduction, both CD4+ and CD8+ T cells expressed the CD5 CAR (Figure 1A). Initial expansion of CD5 CAR-transduced T cells was delayed by 2 to 3 days due to transient fratricide but subsequent expansion superseded that of T cells transduced with a control CD19 CAR (Figure 1B).
CD5 CAR T cells expand and downregulate CD5. (A) Schematic structure of CD5 CAR and transduction efficiency of primary activated T cells. (B) Expansion of activated T cells transduced with either Ctrl CAR or CD5 CAR. Data denote mean ± SD from 4 donors. (C) Surface expression of CD5 on NT T cells or T cells transduced with Ctrl CAR or CD5 CAR at 7 days post-activation. (D) Relative expression of CD5 messenger RNA in NT activated T cells or T cells transduced with CD5 CAR at 7 days post-stimulation. Ctrl CAR, control CAR; NT, nontransduced.
CD5 CAR T cells expand and downregulate CD5. (A) Schematic structure of CD5 CAR and transduction efficiency of primary activated T cells. (B) Expansion of activated T cells transduced with either Ctrl CAR or CD5 CAR. Data denote mean ± SD from 4 donors. (C) Surface expression of CD5 on NT T cells or T cells transduced with Ctrl CAR or CD5 CAR at 7 days post-activation. (D) Relative expression of CD5 messenger RNA in NT activated T cells or T cells transduced with CD5 CAR at 7 days post-stimulation. Ctrl CAR, control CAR; NT, nontransduced.
Expression of CD5 CAR in T cells was stable for >25 days posttransduction (supplemental Figure 1). CAR expression was associated with loss of CD5 (Figure 1C), possibly facilitating the expansion of CD5 CAR T cells and limiting fratricide. Both CD4+ and CD8+ T cells showed similar loss of surface CD5 expression (supplemental Figure 2A), which was not a result of preferential survival of CD5-negative T cells, as overall transcription of the CD5 gene was unaltered (Figure 1D). Hence, CD5 downregulation occurred at the translational and/or posttranslational level. Loss of CD5 from their cell surface did not impair the cytotoxicity of CAR T cells (supplemental Figure 2B) nor did it compromise their cytokine production (supplemental Figure 3) or overall T-cell proliferation (supplemental Figure 4), so that CD5 expression is not required for these (CAR) T-cell functions.
CD5 CAR T cells produce limited and transient fratricide
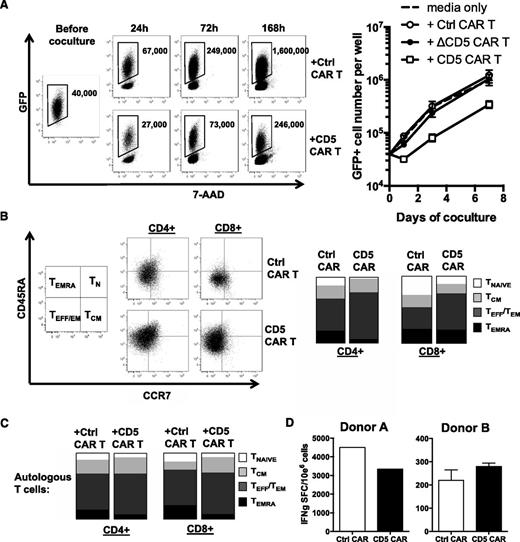
To assess the extent of fratricide among CD5 CAR T cells against autologous T cells, we compared expansion of autologous GFP+-activated T cells cocultured with CD5 CAR- or control CAR-transduced T cells for 7 days. We observed a transient decline in the number of autologous GFP+ T cells after 24 hours of coculture with CD5 CAR T cells followed by sustained expansion (Figure 2A). As expected, autologous T cells expanded in coculture with T cells transduced with either control CAR or truncated CD5 CAR (ΔCD5 CAR, lacking cytoplasmic activation domains) (Figure 2A).
CD5 CAR T cells produce limited fratricide and spare VSTs. (A) Autologous GFP+ T cells were mixed with T cells transduced with Ctrl CAR, truncated CD5 CAR (ΔCD5 CAR, without intracellular signaling domains), or full length CD5 CAR at 1:2 E:T ratio and cocultured for 7 days. Numbers in dot plots denote cell counts of gated GFP+ autologous T cells per well at indicated time points (left). Graph (right) summarizes data from 4 donors ± SD. (B) Phenotype of activated T cells 10 days after transduction with Ctrl CAR or CD5 CAR. Naïve T cells (TNAIVE, CD45RA+ CCR7+), central memory (TCM, CD45RA− CCR7+), effector and effector-memory (TEFF/TEM, CD45RA− CCR7−), and TEMRA (CD45RA+ CCR7−) subsets are shown on representative dot plots with gating strategy (left) and as mean data from 3 donors (right). (C) Phenotype of autologous GFP+ T cells after coculture with Ctrl CAR- or CD5 CAR-transduced T cells for 24 hours. Data shown as mean average from 3 donors. (D) Autologous GFP+ T cells were cocultured with Ctrl CAR T or CD5 CAR T cells for 72 hours and purified by cell sorting. Frequency of T cells specific for cytomegalovirus, Epstein-Barr virus, and adenovirus among sorted cells was measured by IFN-γ ELISPOT.
CD5 CAR T cells produce limited fratricide and spare VSTs. (A) Autologous GFP+ T cells were mixed with T cells transduced with Ctrl CAR, truncated CD5 CAR (ΔCD5 CAR, without intracellular signaling domains), or full length CD5 CAR at 1:2 E:T ratio and cocultured for 7 days. Numbers in dot plots denote cell counts of gated GFP+ autologous T cells per well at indicated time points (left). Graph (right) summarizes data from 4 donors ± SD. (B) Phenotype of activated T cells 10 days after transduction with Ctrl CAR or CD5 CAR. Naïve T cells (TNAIVE, CD45RA+ CCR7+), central memory (TCM, CD45RA− CCR7+), effector and effector-memory (TEFF/TEM, CD45RA− CCR7−), and TEMRA (CD45RA+ CCR7−) subsets are shown on representative dot plots with gating strategy (left) and as mean data from 3 donors (right). (C) Phenotype of autologous GFP+ T cells after coculture with Ctrl CAR- or CD5 CAR-transduced T cells for 24 hours. Data shown as mean average from 3 donors. (D) Autologous GFP+ T cells were cocultured with Ctrl CAR T or CD5 CAR T cells for 72 hours and purified by cell sorting. Frequency of T cells specific for cytomegalovirus, Epstein-Barr virus, and adenovirus among sorted cells was measured by IFN-γ ELISPOT.
Following expansion, CD5 CAR T cells had a low frequency of naïve phenotype cells (CD45RA+ CCR7high) and were enriched for effector and effector memory cells (CD45RA− CCR7low) compared with T cells transduced with a control CAR (Figure 2B). To discover whether the prevalence of effector and effector memory populations in CD5 CAR T cells results from preferential survival of those subsets during the initial period of fratricide, we analyzed the phenotype of autologous T cells cocultured with CD5 CAR T cells for 24 hours to identify populations that were preferentially targeted by CD5 CAR T cells. We found that the frequency of autologous T cells with a naïve phenotype was halved following coculture, whereas central and effector memory cells were less sensitive to fratricide (Figure 2C). This difference in sensitivity is consistent with the observed phenotype of CD5 CAR T cells (Figure 2B). Although naïve and central memory T cells had higher CD5 expression than effector memory cells (supplemental Figure 5A), all T-cell subsets downregulated CD5 with similar kinetics upon coculture with CD5 CAR T cells (supplemental Figure 5B). Therefore, it is unlikely that the differential killing of T-cell subsets is due to the differential availability of CD5 on the cell surface. Instead, it parallels the intrinsically enhanced resistance of central and effector memory subsets to other routes of self-directed cytotoxicity.27,28
We then sought to determine if selective fratricide by CD5 CAR T cells would eliminate virus-specific T cells (VSTs), and thus potentially compromise anti-viral immunity. We cocultured autologous GFP+ T cells with CD5 CAR T cells for 72 hours, then purified GFP+ T cells by cell sorting, and used interferon (IFN)-γ enzyme-linked immunospot (ELISPOT) assay to analyze the frequency of T cells reactive to a pool of peptide antigens derived from cytomegalovirus, Epstein-Barr virus, and adenoviruses. We observed no significant change in the frequency of tri-virus–specific autologous T cells after coculture with CD5 CAR T cells compared with control CAR (Figure 2D). These data demonstrate that the limited and transient fratricide of CD5 CAR T cells does not preclude expansion of memory VSTs.
CD5 CAR T cells effectively recognize and eliminate malignant T-cell lines in vitro
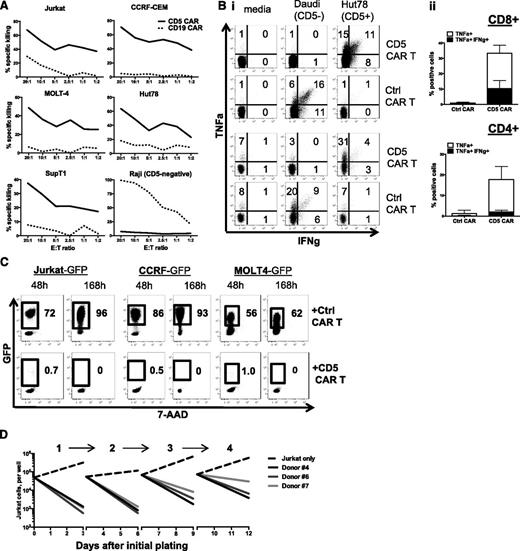
We then evaluated the capacity of CD5 CAR T cells to eradicate CD5+ T-ALL and T-lymphoma cell lines. Compared with control CD19 CAR T cells, CD5 CAR T cells demonstrated significant cytotoxicity against 5 different T-cell lines: Jurkat, CCRF-CEM, MOLT4, Hut78, and SupT1 (Figure 3A). The CD5− B-cell line Raji was not recognized by CD5 CAR T cells (Figure 3A), indicating the selectivity of the CD5 CAR T cells. Both CD4+ and CD8+ CD5 CAR T cells had significant production of IFN-γ and tumor necrosis factor (TNF)-α when cocultured with CD5+ target cells Hut78, but not with the CD5− B-cell line Daudi (Figure 3B).
CD5 CAR T cells eliminate malignant T cells in vitro. (A) Cytotoxicity of CD19 CAR- and CD5 CAR-transduced T cells against T-ALL and T-lymphoma cell lines was assessed in a 5-hour Cr release assay. CD19+CD5− Raji cells (bottom right panel) were used as a negative control for CD5 CAR and positive control for CD19 CAR T cells. (B) Panel i: production of IFN-γ and TNF-α by CD4+ (top) and CD8+ (bottom) T cells transduced with CD19 CAR or CD5 CAR was measured by intracellular cytokine staining. Panel ii: bar graphs show mean ± SD from 3 donors. (C) Long-term coculture of CAR T cells with GFP+ target cell lines Jurkat, CCRF, and MOLT4 at an initial E:T ratio 1:4. Numbers in dot plots denote percentage of target GFP+ cells at indicated time points. (D) Sequential killing of GFP+ Jurkat cells by CD5 CAR T cells. Graph indicates number of target Jurkat cells per well at the beginning and the end of each cycle of cell killing. Data from 3 individual donors are shown.
CD5 CAR T cells eliminate malignant T cells in vitro. (A) Cytotoxicity of CD19 CAR- and CD5 CAR-transduced T cells against T-ALL and T-lymphoma cell lines was assessed in a 5-hour Cr release assay. CD19+CD5− Raji cells (bottom right panel) were used as a negative control for CD5 CAR and positive control for CD19 CAR T cells. (B) Panel i: production of IFN-γ and TNF-α by CD4+ (top) and CD8+ (bottom) T cells transduced with CD19 CAR or CD5 CAR was measured by intracellular cytokine staining. Panel ii: bar graphs show mean ± SD from 3 donors. (C) Long-term coculture of CAR T cells with GFP+ target cell lines Jurkat, CCRF, and MOLT4 at an initial E:T ratio 1:4. Numbers in dot plots denote percentage of target GFP+ cells at indicated time points. (D) Sequential killing of GFP+ Jurkat cells by CD5 CAR T cells. Graph indicates number of target Jurkat cells per well at the beginning and the end of each cycle of cell killing. Data from 3 individual donors are shown.
To assess the ability of CD5 CAR T cells to suppress tumor cell line growth in longer coculture, we cocultured CAR T cells with GFP-expressing Jurkat, CCRF, or MOLT4 cells and analyzed the survival of the target cells at multiple time points. CD5 CAR T cells effectively eliminated >95% of target cells after 48 hours (Figure 3C), and after 7 days of coculture no measurable target cells remained (Figure 3C and supplemental Figure 6). Unlike normal T cells, therefore, the malignant T-cell lines were highly susceptible to CD5 CAR T cells.
The effectiveness of cytotoxic T-cell therapy likely requires the effector cells to sequentially kill multiple target cells. Therefore, we tested the capacity of CD5 CAR T cells to eliminate tumor cells in a sequential killing assay, in which fresh Jurkat cells were added to CD5 CAR T cells every 3 days to restore an E:T of 1:2. We found CD5 CAR T cells could eradicate freshly replenished Jurkat cells for at least 4 iterations (Figure 3D).
Mechanisms of differential killing of normal and malignant T cells
Enhanced cytotoxicity against leukemic cells might result from increased or more stable CD5 expression on the cell surface of malignant T cells. However, surface expression of CD5 was comparable in all leukemic cell lines and activated T cells (supplemental Figure 7A-B). Ligand binding induces rapid internalization of CD5 protein even without crosslinking.29,30 We assessed the kinetics of CD5 internalization in normal and malignant T cells after mixing them with CD5 CAR T cells. No difference in kinetics of internalization was observed (supplemental Figure 7C-D), suggesting that the differential killing was unrelated to disparate persistence of CD5 antigen expression. We then investigated the susceptibility of target cells that had downregulated CD5 from their surface to subsequent cytotoxicity on re-exposure to CD5 CAR T cells. T cells were transduced with a truncated CD5 CAR (ΔCD5 CAR) that lacks intracellular signaling domains but can still trigger downmodulation of CD5 from the cell surface (supplemental Figure 8A). We incubated GFP+ malignant (Jurkat and CCRF) and normal T cells with ΔCD5 CAR T cells to downregulate CD5, and then added T cells transduced with full-length CD5 CAR. We continued to observe >85% killing of CD5low leukemic cells after adding fresh CD5 CAR T cells, likely reflecting re-expression of the targeted antigen over time. Cytotoxicity against autologous T cells was reduced from 29% (without prior CD5 downregulation) to 10% (after coculture with ΔCD5 CAR T cells) (supplemental Figure 8B).
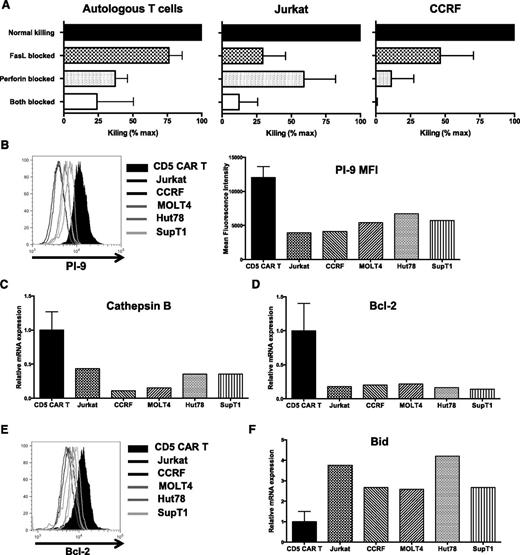
T-cell cytotoxicity is primarily mediated by two separate mechanisms: perforin/granzyme secretion and Fas/FasL-mediated apoptosis. We therefore determined which pathways contribute to the disparate cytotoxicity of CD5 CAR T cells against normal and malignant target cells. We cocultured CD5 CAR T cells with autologous T cells or Jurkat cells in the presence of brefeldin A and anti-FasL antibodies (cooperatively blocking the FasL pathway), or concanamycin A (CMA) and EGTA (inhibiting the perforin pathway), or all four agents. We then measured apoptosis of target cells by Annexin V staining to determine residual cytotoxic effector activity. Blocking the FasL pathway did not substantially alter the extent of cytotoxicity against autologous T cells, but perforin inhibition with CMA/EGTA reduced fratricide by 63% (Figure 4A, left), suggesting the predominance of this pathway in fratricidal killing. In contrast, cytotoxicity against Jurkat and CCRF cells was substantially decreased upon blocking FasL (Figure 4A, middle and right), highlighting the importance of this mechanism in killing malignant T cells in addition to the perforin-dependent pathway. Therefore, CD5 CAR T cells use both perforin- and Fas-mediated pathways to eliminate Jurkat and CCRF cells, whereas fratricide is predominantly mediated by a perforin-dependent mechanism.
Multiple mechanisms contribute to resistance to fratricide. (A) Inhibition of cytotoxicity of CD5 CAR T cells against autologous T cells and Jurkat cells by blocking either FasL (brefeldin A + aFasL), perforin (CMA + EGTA), or both pathways. Cell death was measured by Annexin V after 2 hours of coculture. (B) Expression of PI-9 protein in CD5 CAR T cells and malignant T-cell lines was measured by intracellular staining and flow cytometry (left). Bar graphs show MFI of PI-9 (right). (C) Expression of cathepsin B transcript in CD5 CAR T cells and target cell lines was measured by quantitative polymerase chain reaction. (D) Levels of Bcl-2 transcript in CD5 CAR T cells and target cell lines. (E) Protein expression of Bcl-2 was measured by intracellular staining and flow cytometry. (F) Bid expression in CD5 CAR T cells and target cell lines was measured by quantitative polymerase chain reaction. Error bars denote SD for 3 different T cell donors. MFI, mean fluorescence intensity.
Multiple mechanisms contribute to resistance to fratricide. (A) Inhibition of cytotoxicity of CD5 CAR T cells against autologous T cells and Jurkat cells by blocking either FasL (brefeldin A + aFasL), perforin (CMA + EGTA), or both pathways. Cell death was measured by Annexin V after 2 hours of coculture. (B) Expression of PI-9 protein in CD5 CAR T cells and malignant T-cell lines was measured by intracellular staining and flow cytometry (left). Bar graphs show MFI of PI-9 (right). (C) Expression of cathepsin B transcript in CD5 CAR T cells and target cell lines was measured by quantitative polymerase chain reaction. (D) Levels of Bcl-2 transcript in CD5 CAR T cells and target cell lines. (E) Protein expression of Bcl-2 was measured by intracellular staining and flow cytometry. (F) Bid expression in CD5 CAR T cells and target cell lines was measured by quantitative polymerase chain reaction. Error bars denote SD for 3 different T cell donors. MFI, mean fluorescence intensity.
Effector T cells employ several mechanisms to protect themselves against autolysis, including overexpression of the serine protease inhibitor PI-9, a specific inhibitor of granzyme B,27 and cathepsin B that provides resistance to perforin.28 We found both PI-9 (Figure 4B) and cathepsin B (Figure 4C) to be upregulated in CD5 CAR T cells compared with T-ALL cell lines, providing a means by which CD5 CAR T cells can resist perforin/granzyme-mediated fratricide.
Fas-mediated apoptosis is triggered by caspase 8-mediated cleavage of the pro-apoptotic protein Bid and is inhibited by Bcl-2.31 Although both normal and malignant T-cell lines expressed Fas on the cell surface (supplemental Figure 9), levels of Bcl-2 were significantly higher in CD5 CAR T cells (Figure 4D-E). Conversely, malignant T cells expressed more Bid (Figure 4F), which correlates with the enhanced sensitivity of T-ALL and T-lymphoma cell lines to Fas-mediated cell death.
CD5 CAR T cells recognize and kill primary T-ALL cells
We next evaluated the ability of CD5 CAR T cells to respond to and kill primary tumor cells from T-ALL patients. We detected cytokine production and degranulation by CD5 CAR T cells in response to primary T-ALL blasts from several patients (Figure 5A-B). In the CD5-positive tumors, we observed no correlation between the level of CD5 expression on tumor cells and the intensity of cytokine production by CAR T cells (supplemental Figure 10). The CD5-negative T-ALL #300, by contrast, elicited minimal production of IFN-γ (Figure 5B).
CD5 CAR T cells recognize and kill primary T-ALL cells. (A) Production of IFN-g upon coculture with different primary T-ALL samples was assessed by intracellular cytokine staining. Numbers indicate percent or CAR+ T cells positive for IFN-g. (B) Production of IFN-g (left), TNFa (middle), and expression of CD107a (right) by CD5 CAR T cells upon mixing with thawed T-ALL blasts from 2 patients (T-ALL #295 and #315). Bar graphs depict frequency of cytokine-producing CD4+ and CD8+ T cells as average ± SEM from 4 donors. (C) Cytotoxicity of CD5 CAR T cells against fresh primary T-ALL blasts isolated from peripheral blood mononuclear cells of a T-ALL patient #394 was measured in a 5-hour Cr release assay. (D) Protein expression of PI-9 and (E) Bcl-2 in T-ALL blasts from donor #394 was measured by intracellular staining and flow cytometry. Expression histogram in Jurkat cells is shown with a dotted line (left). Bar graphs depict corresponding MFI compared with CD5 CAR T cells (mean ± SD from 3 donors) and Jurkat T-ALL cell line (right).
CD5 CAR T cells recognize and kill primary T-ALL cells. (A) Production of IFN-g upon coculture with different primary T-ALL samples was assessed by intracellular cytokine staining. Numbers indicate percent or CAR+ T cells positive for IFN-g. (B) Production of IFN-g (left), TNFa (middle), and expression of CD107a (right) by CD5 CAR T cells upon mixing with thawed T-ALL blasts from 2 patients (T-ALL #295 and #315). Bar graphs depict frequency of cytokine-producing CD4+ and CD8+ T cells as average ± SEM from 4 donors. (C) Cytotoxicity of CD5 CAR T cells against fresh primary T-ALL blasts isolated from peripheral blood mononuclear cells of a T-ALL patient #394 was measured in a 5-hour Cr release assay. (D) Protein expression of PI-9 and (E) Bcl-2 in T-ALL blasts from donor #394 was measured by intracellular staining and flow cytometry. Expression histogram in Jurkat cells is shown with a dotted line (left). Bar graphs depict corresponding MFI compared with CD5 CAR T cells (mean ± SD from 3 donors) and Jurkat T-ALL cell line (right).
To assess the cytotoxicity of CD5 CAR T cells against primary tumor cells, peripheral blood mononuclear cells of a T-ALL patient were purified and used as targets in a 5-hour chromium (Cr) release assay with normal donor T cells transduced with either CD5 CAR or a mock retrovirus. We observed substantial cytotoxicity with CD5 CAR T cells and minimal alloreactive killing from mock-transduced T cells (Figure 5C). Sensitivity of primary tumor cells to CD5 CAR T cells was associated with low expression of PI-9 and bcl-2, as was also seen in Jurkat cells (Figure 5D-E). These data further strengthen the hypothesis that CD5 CAR T cells have a significant therapeutic potential to eradicate primary T-cell malignancies.
Efficient control of T-ALL progression by CD5 CAR T cells in vivo
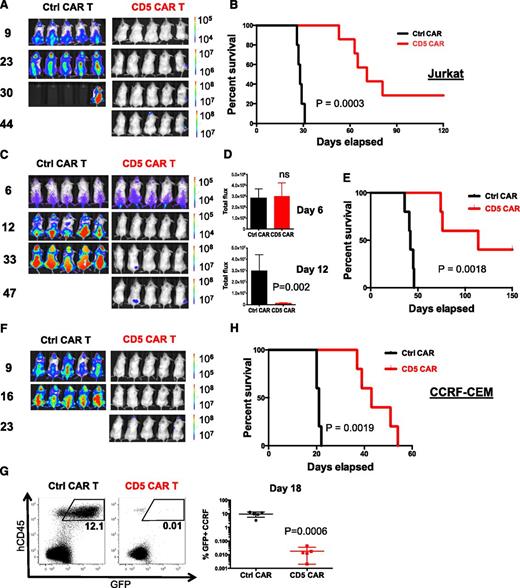
The ability of CAR T cells to suppress or eliminate malignant cells in vivo in xenograft mouse models may be an important predictor of their therapeutic efficacy in patients. We therefore established a xenograft mouse model of disseminated T-ALL by IV engrafting nonobese diabetic-severe combined immunodeficiency γ-chain–deficient mice with firefly luciferase-expressing Jurkat cells and evaluated the capacity of CD5 CAR T cells (injected on day 3 and day 6 after tumor implantation) to control disease progression by recording in vivo luminescence. Upon IV injection, Jurkat cells established disseminated leukemia expanding preferentially in the spine, femur, head, and pelvis,32 with limited numbers of cells in peripheral blood. Mice receiving control CD19 CAR T cells developed rapid disease progression and were euthanized by day 30 (Figure 6A-B). In contrast, mice receiving CD5 CAR T cells were significantly protected from rapid progression and their median survival was prolonged by >150% (28 days in control CAR group vs 71 days in CD5 CAR group; P = .0003) (Figure 6A-B). We also observed superior tumor control by T cells transduced with a CD5 CAR harboring only the CH3 portion of the IgG1 spacer compared with T cells expressing a full-spacer CAR (supplemental Figure 11), consistent with prior reports.33-35
CD5 CAR T cells control progression of T-ALL in xenograft mouse models. (A) Jurkat-FFluc cells (3 × 106 per mouse) were IV injected followed by IV injection of CAR T cells (10 × 106 per mouse) on days 3 and 6 postimplantation. Tumor burden was assessed by IVIS imaging at indicated time points. (B) Kaplan–Meier survival curve; mice were euthanized after developing hind limb paralysis. (C) Eradication of systemic disease by CD5 CAR T cells. Mice were engrafted with Jurkat-FFluc cells, which established systemic disease by day 6. (D) Total luminescence from Jurkat cells recorded on day 6 (prior to CAR T-cell injection) and day 12. (E) Kaplan–Meier survival curve for eradication of systemic disease. (F) CCRF-CEM–FFluc cells (1 × 106 per mouse) were IV injected followed by IV injection of CAR T cells (10 × 106 per mouse) on day 3 and 6 post-implantation. Tumor burden was assessed by IVIS imaging at indicated time points. (G) Relative frequency CCRF-GFP in peripheral blood of mice on day 18 post-engraftment is shown on representative dot plots. (H) Kaplan–Meier survival curve for the CCRF model. P values are shown for each experiment.
CD5 CAR T cells control progression of T-ALL in xenograft mouse models. (A) Jurkat-FFluc cells (3 × 106 per mouse) were IV injected followed by IV injection of CAR T cells (10 × 106 per mouse) on days 3 and 6 postimplantation. Tumor burden was assessed by IVIS imaging at indicated time points. (B) Kaplan–Meier survival curve; mice were euthanized after developing hind limb paralysis. (C) Eradication of systemic disease by CD5 CAR T cells. Mice were engrafted with Jurkat-FFluc cells, which established systemic disease by day 6. (D) Total luminescence from Jurkat cells recorded on day 6 (prior to CAR T-cell injection) and day 12. (E) Kaplan–Meier survival curve for eradication of systemic disease. (F) CCRF-CEM–FFluc cells (1 × 106 per mouse) were IV injected followed by IV injection of CAR T cells (10 × 106 per mouse) on day 3 and 6 post-implantation. Tumor burden was assessed by IVIS imaging at indicated time points. (G) Relative frequency CCRF-GFP in peripheral blood of mice on day 18 post-engraftment is shown on representative dot plots. (H) Kaplan–Meier survival curve for the CCRF model. P values are shown for each experiment.
We next evaluated the capacity of CD5 CAR T cells to control an established tumor by injecting CAR T cells later, on day 6 and 9 post-implantation. By day 6, the overall tumor burden in control and CD5 CAR groups was similar. In the group receiving the CD5 CAR T-cell injection, however, the disease was significantly reduced by day 12 (Figure 6C-D). Median survival of the mice was significantly extended from 42 days with control CAR to 114 days with CD5 CAR (P = .0018) (Figure 6E). No benefit was seen from CD19 CAR T cells despite their higher frequency in peripheral blood after infusion (supplemental Figure 12A,C). The lower CD4:CD8 ratio in CD5 CAR T cells (supplemental Figure 12B,D) likely resulted from better expansion of effector/memory CD8+ T cells or increased susceptibility of CD4+ T cells to fratricide by CD5 CAR T cells.
We established a second xenograft mouse model of an aggressive T-ALL by IV engrafting CCRF-CEM cells to produce a predominantly leukemic distribution of tumor with cells also detected in lymphoid organs. We tested the ability of CD5 CAR T cells administered on day 3 and day 6 post-engraftment to suppress leukemia progression. Although the control mice succumbed to the disease by day 22, injection of CD5 CAR T cells significantly reduced tumor burden and disease progression (Figure 6F). This benefit correlated with decreased frequency of CCRF cells in peripheral blood of mice 18 days post-engraftment (Figure 6G) and extended median survival to 43 days vs 21 days in control mice (P = .0019) (Figure 6H). Re-emerging tumor cells in mice receiving CD5 CAR T cells still expressed CD5 (supplemental Figure 13), suggesting that the lack of complete tumor eradication resulted from suboptimal persistence of CAR T cells in mouse hosts rather than from antigen escape.
Discussion
We show that activated human T cells transduced with a CD5 CAR can specifically recognize and kill malignant T-cell lines and primary T-ALL blasts. Although expansion of CD5 CAR T cells was preceded by transient fratricide, the extent of self-killing was limited. The fratricide also induced the disappearance of CD5 from the cell surface and selected for a resistant differentiated population. CD5 CAR T cells could be expanded in vitro where they recognized and eradicated CD5+ malignant T cells and efficiently controlled disease progression in xenograft mouse models.
Monoclonal anti-CD5 antibodies conjugated with the ricin A chain toxin have been used in clinical trials to eliminate malignant23,36 or self-reactive37,38 T cells. No major irreversible toxicities were reported; some toxicities were attributed to nonspecific uptake of the toxin conjugate in the liver and kidneys, but there were no fatal events attributable to “on-target, off-organ” activity.22-24 There was, however, a lack of sustained effect, which correlated with limited persistence of murine antibodies and the development of human anti-mouse antibodies.22,23 In contrast, both the longevity and self-replication of CAR T cells should induce more durable antitumor responses. It is also possible, however, that such persistence will enhance off-target toxicities. Targeted therapy of lymphoid malignancies with CAR T cells can certainly affect normal lymphocyte populations, as demonstrated in current CD19 CAR trials, in which there is profound aplasia of normal B cells as a result of such “on-target, off-tumor” toxicity. Since CD5 CAR T cells display discernible, albeit limited, fratricide against nontransduced T cells, there is a concern that infusion of CD5 CAR T cells may further decrease T-cell counts in patients. CD5 CAR T cells may also target both peripheral T cells and, in younger patients, CD5hi thymocytes, which may impact generation and homeostasis of normal T cells. Although continuous infusion of Igs may compensate for the lack of humoral adaptive immunity in B-cell–depleted patients, eliminating peripheral T cells and thymocytes could have more substantial consequences that may render patients susceptible to opportunistic infections with viruses and other organisms, and be more difficult to overcome. Although killing of nontransduced normal T cells was limited in these preclinical studies, we do not yet know whether the severity of bystander killing of normal (CAR-negative) T cells will be more profound in humans in vivo. If necessary, it will be possible to incorporate strategies to limit the persistence of CD5 CAR T cells, such as equipping them with a suicide gene39 or a targetable surface marker,40 or limiting the duration of CD5 CAR expression by employing messenger RNA transfection instead of integrating vectors,41 should prolonged and profound aplasia of normal T cells follow administration of CD5 CAR T therapy. Of note, the resistance of activated VSTs to fratricide by CD5 CAR T cells may allow infusion of expanded multivirus-specific VSTs42 alongside with CAR T cells to provide means of immune protection against viral infections.
Notwithstanding the above concerns, our data suggest that the level of fratricide from CD5 CAR is limited. A recent study demonstrated that the profound fratricide by T cells transduced with a survivin-specific TCR43 could be substantially reduced by substituting an alternative survivin-specific TCR clone,44 suggesting that extensive fratricide requires exceeding a certain threshold of T-cell activation. CD5 is rapidly internalized upon binding to a ligand,29,30 limiting its availability on the cell surface and thereby reducing the avidity and duration of CAR-CD5 interactions. Combined with the natural resistance of mature effector T cells to perforin- and granzyme-mediated killing by overexpression of PI-9 and cathepsin B,27,28 these mechanisms associate with preferential survival and expansion of cytotoxic CD5 CAR T cells. Conversely, the lack of inherent resistance mechanisms and enhanced sensitivity to Fas-mediated apoptosis in T-precursor–derived malignant cells evidently makes them more susceptible targets for CD5 CAR T cells.
Our data also show that fratricide occurs primarily in the naïve compartment, leading to selective enrichment of differentiated effector and memory T cells in the CD5 CAR-transduced population. Such enrichment may also reflect constant signaling from CD5 CAR in self-reactive T cells, driving their terminal differentiation. The prevalence of effector and effector memory cells may limit the subsequent in vivo expansion and persistence of CD5 CAR T cells due to inferior self-renewal compared with central memory or naïve phenotype cytotoxic T lymphocytes.26,45 Nonetheless, the depletion of memory cells is not absolute, and virus reactive T cells are spared. Moreover, we found that CD5 CAR T cells were able to perform sequential killing and elimination of tumor cells in up to 4 iterations. The sequential killing capacity suggests the T cells can eliminate large numbers of tumor cells upon infusion.
In contrast to a complete elimination of malignant T cells in vitro, in most animals, CD5 CAR T cells failed to completely eradicate established disease. The tumor cells that ultimately re-emerged in these animals retained CD5 expression, indicating that tumor relapse did not result from antigen loss. In most mice, relapsed tumor originated from several anatomical locations such as the periodontal region, the bone marrow, and the central nervous system, that were previously shown to be the sites of persistent disease in a NALM-6 leukemia xenograft model.46,47 It is therefore more likely that failure to eradicate all xenograft reservoirs was due to the limited persistence of human CD5 CAR T cells in a mouse host, an issue commonly observed in other xenograft models using adoptive T-cell therapy.46,47 Further optimization of the signaling domain configuration in the CD5 CAR, co-expression of chimeric cytokine receptors, and “humanization” of scFv may all serve to prolong the antitumor effect in patients.
In conclusion, we demonstrate that a CD5 CAR can effectively redirect human T cells to target malignant CD5+ cells with limited fratricide. This approach may provide novel treatment options for patients with refractory or relapsed CD5+ T-cell neoplasms.
The online version of this article contains a data supplement.
There is an Inside Blood Commentary on this article in this issue.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 USC section 1734.
Acknowledgments
The authors thank G. Dotti for providing CAR backbone and CD19 CAR control, C. Rooney for critically reviewing the manuscript, T. Goltsova and J. Sederstrom for technical assistance with cell sorting, M. Schmück-Henneresse for assistance with intracellular cytokine staining, and C. Gillespie for editing the manuscript.
This study was supported by a grant from the National Institutes of Health (NIH), National Cancer Institute (NCI) (P50 CA126752). The authors also appreciate the support of shared resources in the Cancer Center (support grant NCIP30CA125123). R.H.R. was supported by NIH, National Heart, Lung, and Blood Institute training grant T32 HL092332. This study was funded by a research collaboration of the Center for Cell and Gene Therapy with Celgene and Bluebird Bio Inc.
Authorship
Contribution: M.M. conceived the CD5 CAR design, planned and performed the experiments, analyzed data, and wrote the manuscript; R.H.R. provided primary T-ALL samples; H.T. contributed to ELISPOT assays; M.K.B. directed the study, planned the experiments, and wrote the manuscript.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: Malcolm K. Brenner, Texas Children’s Hospital and Houston Methodist Hospital, Feigin Center, 1102 Bates Ave, Ste 1630, Houston, TX 77030; e-mail: mbrenner@bcm.edu.


This feature is available to Subscribers Only
Sign In or Create an Account Close Modal