In this issue of Blood, Stark et al report that the platelet-derived high-mobility group box 1 protein (HMGB1) orchestrates an inflammation-induced venous thrombosis.1
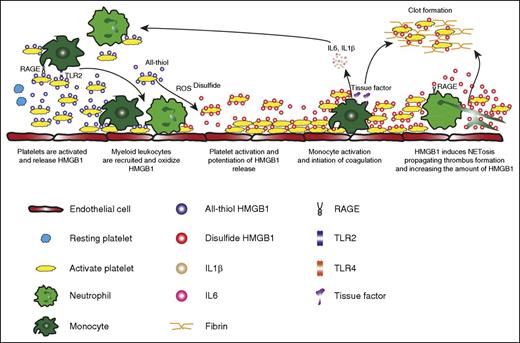
Platelet-derived disulfide HMGB1 as central mediator of deep venous thrombosis (DVT). Platelets adhere to the vessel wall and present reduced HMGB1 on their surface upon activation. This causes the recruitment of monocytes to the developing thrombus, which upon activation release reactive oxygen species (ROS) resulting in the oxidation of HMGB1, thereby rendering it prothrombotic. In an autocrine/paracrine manner, disulfide HMGB1 promotes further platelet activation and aggregation, potentiating the amount of released HMGB1 within the thrombus. This results in the accumulation of monocytes through RAGE and TLR2, inducing the expression of tissue factor and cytokines, setting in motion a vicious circle of coagulation and inflammation eventually leading to obstructive thrombus formation within the vein. Finally, disulfide HMGB1 induces the formation of prothrombotic neutrophil extracellular traps mediated by RAGE, which in turn expose more HMGB1 on extracellular DNA strands and promote DVT propagation. See supplemental Figure 5 in the article by Stark et al that begins on page 2435.
Platelet-derived disulfide HMGB1 as central mediator of deep venous thrombosis (DVT). Platelets adhere to the vessel wall and present reduced HMGB1 on their surface upon activation. This causes the recruitment of monocytes to the developing thrombus, which upon activation release reactive oxygen species (ROS) resulting in the oxidation of HMGB1, thereby rendering it prothrombotic. In an autocrine/paracrine manner, disulfide HMGB1 promotes further platelet activation and aggregation, potentiating the amount of released HMGB1 within the thrombus. This results in the accumulation of monocytes through RAGE and TLR2, inducing the expression of tissue factor and cytokines, setting in motion a vicious circle of coagulation and inflammation eventually leading to obstructive thrombus formation within the vein. Finally, disulfide HMGB1 induces the formation of prothrombotic neutrophil extracellular traps mediated by RAGE, which in turn expose more HMGB1 on extracellular DNA strands and promote DVT propagation. See supplemental Figure 5 in the article by Stark et al that begins on page 2435.
HMGB1 is a ubiquitous nuclear protein present in the majority of eukaryotic cells. It was discovered as a protein that maintains nucleosome structure and regulates gene transcription.2 It was later discovered that HMGB1 can also be released from cells into the extracellular space. Bioactive HMGB1 can passively diffuse from necrotic but not apoptotic cells.3 HMGB1 can also undergo acetylation in the nucleus and be actively released by secretory lysosomes from a variety of immune and nonimmune cells. Once secreted, HMGB1 acts as a danger-associated molecular patterns molecule, triggering inflammation and contributing to the pathology of various inflammatory and autoimmune diseases via activation of receptor for advanced glycation end products (RAGE), TLR2, and TLR4 receptors.2,3
In 2000, Rouhiainen and colleagues demonstrated that, although lacking a nucleus, platelets express HMGB1, and upon platelet activation, HMGB1 is both exported to the cell membrane and released from the cells.4 Later, it was proposed that platelet-derived microparticles loaded with HMGB1 may contribute to the chronic microvascular injury and endothelial activation observed in systemic sclerosis patients.5 Vogel and colleagues were the first to demonstrate the contribution of platelet HMGB1 to thrombosis. Ferric chloride–induced thrombus formation in mesenteric arteries was attenuated in mice with a platelet-specific deletion of HMGB1, and blood from these mice was less thrombogenic in a collagen-coated flow chamber system.6 Furthermore, HMGB1 expression on the platelet surface was significantly upregulated in trauma patients, and platelet-specific deletion of HMGB1 reduced platelet aggregation, microvascular thrombosis, and inflammation as well as organ damage in a mouse model of trauma /hemorrhagic shock.6
Stark and colleagues identified activated platelets as the main source of HMGB1 in murine venous thrombosis. In a series of elegant experiments, the authors demonstrated that HMGB1 released from platelets promotes thrombosis in the inferior vena cava stenosis model by coordinating the crosstalk between platelets, monocytes, and neutrophils. HMGB1 is known to undergo extensive posttranslational modifications that modulate its functions. Interestingly, Stark and colleagues showed that the oxidized, disulfide isoform of HMGB1 is the most important for platelet aggregation, monocyte tissue factor expression, and neutrophil extracellular trap formation and subsequent development of venous thrombosis (see figure).
Venous thromboembolism, including DVT and pulmonary embolism, is the third leading cause of cardiovascular death in the world after myocardial infarction and stroke.7 Anticoagulation, using vitamin K antagonists, heparins, or direct oral anticoagulants, is the primary treatment in patients with venous thromboembolism, both to limit existing thrombus growth and to prevent recurrence. Recent studies on the development of new anticoagulants with a reduced risk of bleeding identified novel therapeutic targets, including factors FXIa, FXIIa, and FXIIIa.7 Can HMGB1 join that list? Given its important role in chemokine and cytokine expression, prolonged inhibition of HMGB1 could affect the innate immune response to infection. Therefore, targeting HMGB1 may be better suited for the short-term treatment of acute venous thrombosis rather than extended prophylaxis. In addition, platelet-specific deletion of HMGB1 significantly prolonged the tail bleeding time in mice, suggesting that HMGB1 might be important for hemostasis.6 As an added advantage, targeting HMGB1 might have simultaneous anti-inflammatory and antithrombotic effects as well as be effective for the treatment of both venous and arterial thrombosis. Indeed, recent clinical trials demonstrated that a low dose of aspirin, conventionally regarded as an agent that prevents arterial thrombosis, reduced the rate of recurrence of venous thromboembolism.8 It is important to note that salicylic acid, a metabolite of aspirin, was recently shown to inhibit HMGB1, including the disulfide isoform.9
In summary, venous thrombosis can be added to the growing list of pathologic conditions modulated by extracellular HMGB1. The current study by Stark and colleagues highlighted the redox isoform-specific contribution of platelet HMGB1 to sterile inflammation-induced venous thrombosis. Future development of isoform-specific inhibitors of HMGB1 may mitigate excessive inflammation and thrombosis without affecting the beneficial effects of HMGB1 in the resolution of inflammation and tissue repair.10
Conflict-of-interest disclosure: The author declares no competing financial interests.