Abstract
Background
The platelet storage lesion is mitigated by refrigeration in platelet additive solutions (PAS), but questions remain regarding effects of cold storage (4°C) on mitochondrial function. Understanding underlying mechanisms responsible for enhanced hemostatic function of 4°C-stored PAS platelets will be critical for such improvements in platelet transfusion. We hypothesized that storage of platelet concentrates at 4°C leads to GPIIb-IIIa activation and thus aggregate formation through fibrinogen binding, and that this could be prevented by storing platelets in platelet additive solution (PAS) without compromising platelet function.
Methods
Apheresis platelets in plasma (AP) or apheresis platelets in PAS were stored at 22°C or 4°C for up to 15 days. Measurements include platelet counts, blood gases, flow cytometry analysis, intracellular reactive oxygen species (ROS) levels, and high resolution respirometry. To explore the effect of fibrinogen on storage, platelets were treated with abciximab to inhibit GPIIb-IIIa binding and aggregation. Donor fibrinogen levels were measured before being collected in PAS; samples were separated into minibags for comparison of control 4°C PAS-stored platelets to 4°C PAS-stored platelets with fibrinogen added back to original donor levels (PAS-FBN).
Results
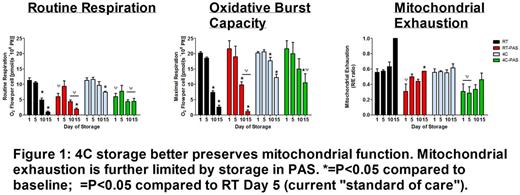
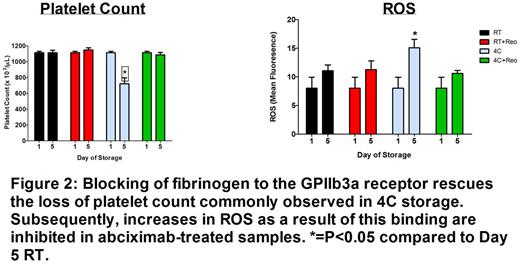
Mitochondrial respiration was lower in platelets stored at 4°C compared to 22°C in both AP and PAS; however, PAS-stored platelets retained lower mitochondrial respiration over the time course. Additionally, maximal mitochondrial oxygen utilization (oxidative burst capacity) was better preserved in 4°C-stored platelets; PAS demonstrated less overall mitochondrial exhaustion at 4°C compared to 4°C-AP samples (Figure 1). PAS-FBN resulted in reduced oxidative burst capacity over 15 days of storage which was consistent with loss of platelet function observed through flow cytometry analysis including increased phosphatidylserine expression, microparticles, and GPIb shedding. Mitochondrial intracellular ROS levels were lower in all 4°C-stored samples compared to their respective RT-stored groups. PAS-FBN resulted in significantly lower platelet counts (Day 5: BL= 1.1x106±.04plt/µL, PAS = 1.1 x106±.06plt/µL, PAS-FBN = .61 x106±.06plt/µL; P<0.001) compared to 4°C PAS-stored platelets and displayed marked increases in visible aggregates in the storage bag. PAS-stored platelets resulted in significantly lower levels of intracellular ROS compared to AP at both RT and 4°C conditions and PAS-FBN (P<0.05). 4°C-AP-stored samples treated with abciximab (AP-Ab) rescued the loss in platelet count commonly encountered in 4°C-AP storage (Day 5: BL = 1112±18, AP-4°C = 720±77, AP-Ab = 1085±28;P<0.001) and resulted in less intracellular ROS compared to untreated 4°C-AP samples at Day 5 (Figure 2).
Discussion
Mitochondrial damage and ROS production may contribute to loss of platelet viability during storage, whereas cold storage is known to preserve platelet function. Our previous work has shown that use of PAS in stored platelets prevents aggregate formation, enhances platelet function, results in less oxidant stress, and preserves both mitochondrial function and potential compared to AP-stored platelets. Here we demonstrate that reduction in fibrinogen levels of 4°C PAS -stored platelets plays a key role in the underlying mechanism for PAS-stored platelet enhancement. Taken together, these data indicate that cold storage combined with PAS preserves platelet viability both by maintaining energy potential and preventing degradation due to enhanced ROS.
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal