Abstract
Background: Acute Graft-versus-host disease (aGVHD) is a leading cause of morbidity and mortality following allogeneic haematopoietic stem cell transplantation (HSCT). The diagnosis and management of aGVHD relies on clinical criteria and graded according to the number and extent of organ involvement. Clinical diagnosis of aGVHD is one of exclusion and usually requires tissue biopsy to aid diagnosis. The aim of this study was to evaluate the clinical utility of a biomarker panel to help identify individuals at risk of developing aGVHD and its ability to differentiate severity at onset of acute graft-versus-host disease (aGVHD) following T-cell depleted allogeneic HSCT.
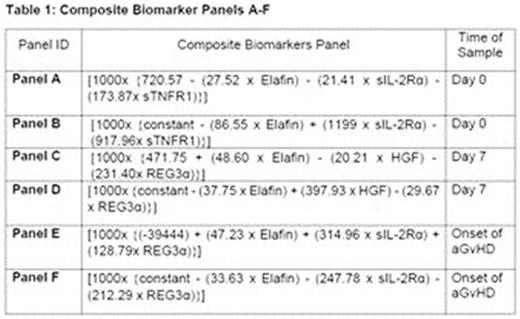
Methods: This retrospective study of T-cell depleted reduced intensity allogeneic HSCT patients in our centre included 12 biopsy confirmed aGVHD patients (Grade I-IV; Modified Glucksberg criteria) and 14 patients as negative controls without any evidence of aGVHD, matched for age, sex, underlying haematological disease, time since transplant and conditioning regimen, within 100 days of transplantation. Serum samples on Day 0, Day +7 and at onset of aGVHD were analysed by ELISA for Elafin, regenerating islet-derived 3-α(REG3α), soluble tumour necrosis factor receptor 1 (sTNFR1), soluble interleukin-2 receptor-α (sIL-2Rα) and hepatocyte growth factor (HGF). Biomarker data was combined as composite panels A-F (Table 1) using binary and ordinal logistic regression analysis as best fit model. Receiver Operating Characteristic curves (ROC) analysis was performed to study sensitivity and specificity of the composite panel for diagnosis of aGVHD.
Results: Composite biomarker panel B (Elafin+ sIL-2Rα + sTNFR1) and panel D (Elafin+ HGF + REG3α) at Day 0 and at Day 7 post-transplant differentiated significantly between biopsy confirmed severe aGVHD (Grade III-IV) and patients with no GVHD respectively (p=0.02). Panel F (Elafin+ sIL-2Rα + REG3α) strongly differentiated between various grades of aGVHD (Grade I-IV) at time of aGVHD onset (p<0.01), potentially predictive of disease severity. Composite panels A (Elafin+ sIL-2R + sTNFR1) and C (Elafin+ HGF + REG3) analysed in samples at Day 0 and Day +7 could not differentiate between patients with aGVHD (all grades) and no GVHD (p=0.54 and p=0.84 respectively), however Panel E (Elafin+ sIL-2Rα + REG3α) significantly differentiated between patients with no GVHD and biopsy confirmed aGVHD at time of onset of clinical aGVHD (p=0.02). This is suggestive of diagnostic utility of this composite biomarker panel in aGVHD diagnosis. The area under the curve (AUC) for this panel at time of onset was 0.65 with specificity, sensitivity, positive predictive value and negative predictive value of 100%, 55.6%, 100% and 78.9% respectively (p=0.03)
Conclusions: This pilot data supports the usefulness of these composite biomarker panels in the prediction of severity and diagnosis of acute GVHD in patients undergoing T-cell depleted reduced intensity allogeneic HSCT. Further larger studies are needed to validate their use in the clinical management of aGVHD.
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal