Abstract
Background: Antithymocyte globulin (ATG)-based immunosuppressive therapy (IST) has been successfully used as the first-line treatment for severe / very severe aplastic anemia (SAA/VSAA) patients if no HLA-matched sibling donor was eligible for HSCT as a first choice. It was reported rabbit ATG (rATG) produced more profound immunosuppressive activity compared to horse ATG (hATG). However, recent clinical studies indicated that the stronger lympholytic activity did not mean that rATG was more effective. Most experiences from adult SAA/VSAA implied the efficacy of rATG was worse than hATG. However, susceptibility of children to intensive IST might not be exactly the same as adult patients, long-term efficacy of rATG in historic studies for children with SAA/VSAA was still elusive.
Purpose: This study includes the largest cohort of pediatric AA patients treated with first-line rATG+CSA regimen published to date after a median follow-up of 69 months, aiming to assess the long-term outcome of rATG for children, and to identify the significant prior factors in clinical decision making.
Methods: We reviewed 231 SAA/VSAA patients under 18 years old assigned to rATG+CSA from February 2000 to May 2014 in Department of Pediatrics, the Blood Diseases Hospital & Institute of Hematology, CAMS & PUMC. Response was evaluated 3, 6, 9, 12 24, 36 and 60 months after IST. We separately defined SAA-II as a specific type of gradually progressed SAA from a NSAA status within a longer period for at least 6 months. Multivariate logistic regression models were used to evaluate the effects of variables on the responses at different time points. Multivariate Cox model analysis of overall survival (OS) and failure-free survival (FFS) was calculated for variables with a log rank P value less than 0.1 in Kaplan-Meier analysis.
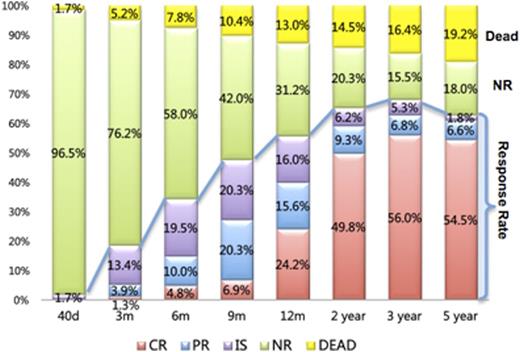
Results: Of the overall patients, the total responded patients were 79(34.3%), 110(51.6%), and 129 (60.6%) at 6, 9, 12 months following IST, respectively. Intriguingly, 22 patients achieved delayed response between 12 months and 24months after IST, which increased the overall response rate by 10.2%, afterwards the rate reached a plateau by 3 years with the best response rate of 74.6% (Figure 1). Differences in baseline clinical parameters pre-IST were associated with response to IST. Absolute neutrophil count (ANC) less than 0.1*109/L was associated with an unfavorable early response rate at 6 months (P=0.009); absolute lymphocyte count (ALC) less than 1.6*109/L was a significant predictor for better response by 6 months and 12 months in multivariate analysis [6 months, P=0.033 vs. 12 months, P=0.021]. Lower absolute reticulocyte count (ARC no more than 18.5*109/L) predicted worse late IST response by 2 years and 3 years. In our large series of cohort, 5-year OS and FFS were 82.7% and 61.9%. Patients with VSAA as a significantly unfavorable prognostic factor had a much lower probability of 5-year survival when compared to patients diagnosed with SAA (76.4% vs. 87.2%, P<0.001, Figure2A). In multivariate analysis, SAA-II (P=0.021, Figure2B), and a pretreatment lower ARC (P=0.020, Figure2C) were independent unfavorable prognostic factors for FFS, but moderate PNH clone size (more than 5%) was verified as a good predictor for FFS (P=0.006, Figure 2D). At the last follow-up, twelve of the 135 responders relapsed after IST, meanwhile eight patients in responders and seven patients in non-responders experienced clonal evolution after IST, corresponding to cumulative incidences at 5.2% of relapse and 6.5% of evolution, which were obviously lower than previous reports.
Conclusions: The combination of rATG and CSA was confirmed as an effective first-line therapy for children with SAA/VSAA in our cohort. We discerned a protracted recovery but an ultimately comparable long-term outcome of rATG. Baseline blood parameters (ANC, ALC, ARC) were predictive factors of response rate. Intensive supportive care may be necessarily pivotal to survival in cases of VSAA. Importantly, moderate PNH clone might be beneficial to FFS. Besides, for those who experienced gradually progressed disease course, early HSCT might be a more preferable option than receiving IST although further validation remains to be done.
Prognostic factors for overall survival (OS) and failure-free survival (FFS)
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.
This icon denotes a clinically relevant abstract


This feature is available to Subscribers Only
Sign In or Create an Account Close Modal