Abstract

Diffuse large B cell lymphoma (DLBCL) comprises two distinct molecular subtypes: germinal center B cell (GCB) subtype and activated B cell (ABC) subtype. The pathogenesis of ABC-DLBCL is characterized by two processes - the activation of NF-KB and a block in terminal B-cell differentiation. However, in GCB-DLBCL only a few biologically relevant pathways have been identified, which has hampered the development of targeted therapies with specific efficacy in this subtype.
Recurrent genetic alterations involved in PI3K-AKT signaling pathway have been identified in the patients with solid cancers, and dramatic responses have been observed to PI3K inhibitors in clinical trials. The prevalence and clinical significances of genetic alterations in PI3K-AKT pathway have not been well studied in DLBCL. Previous studies have reported that loss of PTEN expression was observed in almost half of GCB-DLBCL cases but were largely absent in the ABC-DLBCL. In addition, functional studies have recently shown that the inactivation of Gα13 signaling pathway genes may also activate AKT in germinal center driven lymphoma, raising the possibility that additional genes within these pathways are affected in GCB-DLBCL. Herein, we identified genetic alterations involved in the PI3K-AKT pathway and evaluated their clinical impact.
We analyzed biopsies from 347 patients newly diagnosed with de novo DLBCL uniformly treated with R-CHOP at the BC Cancer Agency. High-resolution copy number analyses were performed using Affymetrix SNP 6.0 arrays. Mutation status was determined using deep targeted re-sequencing of the coding exons of 61 genes with a Truseq Custom Amplicon assay (Illumina) and/or Fluidigm Access Array chips, and RNAseq. Immunohistochemical staining of phospho-AKT (pAKT) was performed on tissue microarrays (n=332). Cell-of-origin (COO) was assigned by gene expression (Lymph2Cx assay) in 323 cases - 183 GCB, 104 ABC and 36 unclassifiable.
GISTIC analysis revealed several COO-specific peaks with copy number changes. Among them, focal 10q23.3 deletion including PTEN was detected in GCB-DLBCL (q-value=1.7e-7), but not in ABC-DLBCL. We also identified two focal amplification peaks in GCB-DLBCL containing microRNAs MIR17HG (13q31.1;q=1.01e-24) and MIR21 (17q23.3; q=1.45e-6), which are known to down-regulate PTEN and result in activation of PI3K-AKT signaling. Furthermore, we detected recurrent INPP4B deletion (4q21.23) in GCB-DLBCL (q= 0.004) only. Of note, the lipid phosphatase INPP4B has been shown to play a role as a tumor suppressor that controls the levels of PI3K lipid products leading to AKT activation and metastasis of some solid cancers. This is consistent with the observation that PTEN and INPP4B deletions were individually associated with increased pAKT protein expression in our cohort (p=0.015 and p<0.0001, respectively).
With respect to mutations, GNA13, P2RY8 and ARHGEF1 were more frequently mutated in the GCB than ABC subtype (26% vs 0.6%; p<.0001, 25% vs 7%; p=.0002, and 8% vs 0.5%; p=0.008, respectively). Previously known mutations of PI3KCA and PI3KCB were not recurrently observed in GCB-DLBCL.
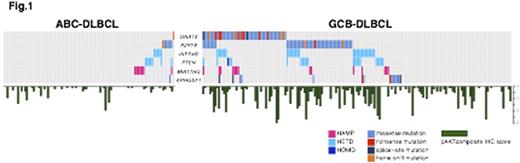
Based on these genetic data, we found that the PI3K-AKT signaling pathway was more commonly altered in GCB-DLBCL (64% (114/177)) than in ABC-DLBCL (23% (23/98) p<0.0001; Fig1). Consistent with this, pAKT protein expression was significantly higher in GCB-DLBCL than ABC-DLBCL (p<0.0001). The cases with higher pAKT protein expression (defined as the 25% highest pAKT protein expressors) were associated with significantly inferior outcome in GCB-DLBCL (5y-PFS 75% vs 59 %, p= 0.007 and 5y-OS 81% vs 64%, p=0.004; Fig2). At the genetic level PTEN and INPP4B deletions were individually associated with poor outcome in GCB-DLBCL (p=0.01 and p=0.045, respectively), and further, patients whose tumors harbored both genetic alterations had even worse prognosis (p= 0.004, Fig 3).
In conclusion, genetic alterations in the PI3K-AKT pathway were frequently observed in GCB-DLBCL and were associated with activation of the pathway and inferior outcomes. These results suggested that AKT and mTOR inhibitors might be beneficial in a greater proportion of GCB-DLBCL than expected.
The distribution of genetic alterations of PI3K-AKT pathway and pAKT protein expression.
The distribution of genetic alterations of PI3K-AKT pathway and pAKT protein expression.
KM curves according to pAKT protein expression (Fig 2) and PTEN and INPP4B deletions (Fig 3).
KM curves according to pAKT protein expression (Fig 2) and PTEN and INPP4B deletions (Fig 3).
Sehn:roche/genentech: Consultancy, Honoraria; amgen: Consultancy, Honoraria; seattle genetics: Consultancy, Honoraria; abbvie: Consultancy, Honoraria; TG therapeutics: Consultancy, Honoraria; celgene: Consultancy, Honoraria; lundbeck: Consultancy, Honoraria; janssen: Consultancy, Honoraria. Connors:F Hoffmann-La Roche: Research Funding; Millennium Takeda: Research Funding; Seattle Genetics: Research Funding; Bristol Myers Squib: Research Funding; NanoString Technologies: Research Funding. Scott:NanoString Technologies: Patents & Royalties: named inventor on a patent for molecular subtyping of DLBCL that has been licensed to NanoString Technologies.
Author notes
Asterisk with author names denotes non-ASH members.

This icon denotes a clinically relevant abstract


This feature is available to Subscribers Only
Sign In or Create an Account Close Modal