Abstract
Cardiopulmonary complications are an important contributor to morbidity and mortality in patients with sickle cell anemia (SCA). We have recently discovered that patients with SCA develop cardiomyopathy with restrictive pattern, characterized by diastolic dysfunction with progressive left atrial dilation, superimposed on the anemia-associated hyperdynamic physiology causing hypertrophied, dilated ventricles (Niss et al, JACC: Cardiovascular Imaging, 2016). Using a longitudinal systematic analysis of mouse models of SCA versus mice with iron-deficiency anemia, we found that this unique cardiomyopathy is indeed not due to chronic anemia and is associated with upregulation of genes related to oxidative stress pathways (Bakeer et al, PNAS, in press). These findings along with the known fact of increased inflammation and oxidative stress in SCA led us to postulate that NADPH oxidase (Nox)-mediated reactive oxygen species (ROS) may be an important pathogenic mechanism underlying cardiomyopathy in SCA and consequently mice lacking Nox-mediated ROS might be relatively protected from cardiac dysfunction.
To investigate the role of NADPH oxidase in SCA cardiovascular pathophysiology, we bred the knock-in humanized SCA murine model Hbbtm2(HBG1,HBB*)Tow (where mouse α- and β-globin genes have been replaced by the human α- and βS globin genes; commonly known as UAB mice and herein called SS mice) with mice knocked out for the p22phox subunit of NADPH oxidase (a common subunit for all mouse Nox isoforms). We evaluated the phenotype of SS;p22phox-/- mice in blood and determined their cardiac function as compared to SS (p22phox+/+) littermate control mice. Hemolysis and ensuing reticulocytosis did not appear significantly improved in the SS;p22phox-/- mice; red blood cell (RBC) ROS was also stable likely due to the major contribution of mitochondrial ROS in the reticulocytes. Baseline ROS levels in the neutrophils of SS;p22phox-/-mice were similar to the levels in SS mice, but inducible ROS was almost eliminated as expected with deficiency of the p22phox subunit and inactivation of all NADPH oxidase isoforms.
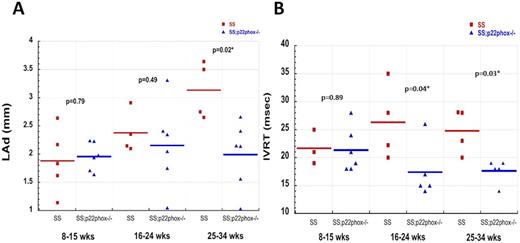
Starting at 8 weeks of age, cardiac structure and function were assessed on age-matched SS and SS;p22phox-/- mice by serial echocardiography. We studied 3 timepoints: 8-15 weeks of age, 16-24 weeks, and 25-34 weeks. SS mice developed progressively increased left atrial dimension (LAd) starting at 16 weeks of age while SS;p22phox-/- mice had stable LAd, with values similar to WT (Figure 1A). This difference became more pronounced with aging (P=0.02 at 25-34 weeks). Isovolumic relaxation time (IVRT), the time interval between closure of the aortic valve and opening of the mitral valve, was prolonged in the SS mice older than 16 weeks of age, consistent with diastolic dysfunction. In contrast, SS;p22phox-/- mice had no change in IVRT (Figure 1B). Also consistent with diastolic dysfunction, the ratio of transmitral E and e-(MV IVS E/e-) was increased in the SS group vs SS;p22phox-/-. Finally, the SS mice showed elevated left ventricular (LV) mass and decreased LV shortening fraction by 22-34 weeks whereas these parameters were preserved in the SS;p22phox-/-mice.
Histopathology studies were performed to evaluate changes in the cardiac tissue. In the SS mice, H&E, picrosirius red and Masson trichrome staining showed not only significant LV hypertrophy and dilation but also significant interstitial fibrosis. Immunostaining for the extracellular matrix proteins collagen and fibronectin, showed extensive deposition of these proteins in the SS mouse heart. In contrast, SS; p22phox-/-mouse hearts were relatively spared.
In conclusion, our data show that SS;p22phox-/-mice demonstrate better preserved diastolic and systolic heart function compared to SS mice, and decreased heart tissue damage. These findings suggest that therapeutic manuevers aimed at decreasing oxidative stress in SCA may be an effective strategy to counter SCA cardiomyopathy.
Echocardiographic parameters evaluating diastolic function of the heart of SS vs SS;p22phox-/-mice. A. Left atrial dimension (LAd). B. Isovolumic relaxation time (IVRT). SS: Hbbtm2(HBG1,HBB*)Tow (UAB) with wild-type p22phox; SS;p22phox-/-: UAB mice with targetted deletion of p22phox.(statistical analysis was performed using Wilcoxon rank-sum test; JMP 9.0 SAS Institute, Cary, North Carolina)
Echocardiographic parameters evaluating diastolic function of the heart of SS vs SS;p22phox-/-mice. A. Left atrial dimension (LAd). B. Isovolumic relaxation time (IVRT). SS: Hbbtm2(HBG1,HBB*)Tow (UAB) with wild-type p22phox; SS;p22phox-/-: UAB mice with targetted deletion of p22phox.(statistical analysis was performed using Wilcoxon rank-sum test; JMP 9.0 SAS Institute, Cary, North Carolina)
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal