Abstract
The clinical heterogeneity of MDS results from the combinatorial diversity of seemingly random, and thus individually unique, clone mutation hierarchies. Identifying genotype/phenotype associations is thus difficult. Indeed, to date, prognostic utility exists for mutations in only a few genes. We studied whether subsequent mutations are acquired randomly or follow certain rules/preferences, and whether the initial vs. secondary alterations affect the clinical and morphologic features.
We collected samples from 1,809 pts. with lower- and higher-risk MDS, and MDS/MPN. Multi-targeted deep sequencing (TS) for 60 myeloid genes and paired WES for a subset of 225 pts., was performed. All somatic mutations and copy number alterations were subjected to a bioanalytic clonal hierarchy (copy-number-adjusted VAF-based) ranking. Acknowledging resolution limitations of hierarchical positions of "sweeping" mutations, we used a conservative cut off (5% of delta VAF) for minimal difference between VAFs to identify 1st hit "dominant" mutations from subsequent hits; referred as to secondary mutations.
We identified 2,722 somatic mutations in WES cases (92% of pts. had secondary hits). Of which 563 dominant mutations (480 genes) were accompanied by 2,159 secondary mutations (1,590 genes). After confirming congruency of TS vs. WES (TS covered 96% of WES spectrum), we proceeded with further analysis using TS. The most common founder mutant genes, e.g., SF3B1, TET2, DNMT3A, SRSF2, and ASXL1 were exclusive, while secondary mutations often coexisted. No genes were mutated in a dominant fashion exclusively, but some mutations were solely subclonal, e.g ., FLT3 and K/NRAS . The association between primary and secondary hits was not random; for example, SF3B1 dominant hits were followed by secondary events in TET2, JAK2, and DNMT3A . In contrast, in cases with TET2 as dominant hits, the most frequent secondary hits were TET2, ASXL1, and SRSF2 . For certain genes dominant or subclonal hits had a different impact on outcome. SRSF2 and U2AF1 exerted differential impact between the dominant and secondary hits by univariate Cox regression analysis. The dominant and secondary hits of 5 genes (TP53, RUNX1, STAG2, ASXL1 and SF3B1) had the same significant impact on survival. In multivariate analysis of mutations of above 5 genes , SRSF2 and U2AF1, both dominant and secondary mutations of RUNX1 and TP53 independently influenced poor prognosis.
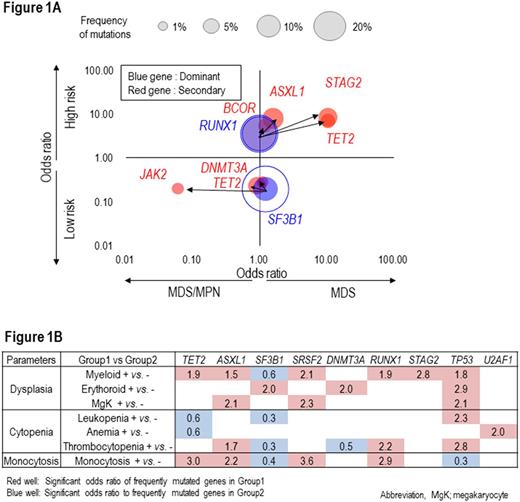
We applied 2-dimensional regression plots to assess how the presence of specific hits affects the odds of progression (low risk, vs. high risk of progression) and phenotypic features (dysplasia vs. myeloproliferative). These analyses revealed many important relationships as to what degree ancestral hits predetermine the phenotype and which secondary hits contribute to progression vs. morphologic features. In general, accumulating secondary mutations promoted progression but some subclonal events also affected phenotypic features. STAG2 and SRFS2 bothincreased the propensity of progression, but STAG2 mutations steered morphology towards dysplasia, while SRSF2 hits steered towards myeloproliferative features. Similarly, SF3B1 mutations were significantly associated with a low risk of progression, while RUNX1 and ASXL1 mutations were associated with a higher progression risk. Specific combinations of dominant and secondary mutations led to particular diagnoses. For instance, if pts with dominant RUNX1 mutations acquired secondary STAG2 and TET2 mutations, MDS was progressive. In contrast, pts. with dominant SF3B1 mutations who acquired secondary JAK2 mutations were more likely to have a myeloproliferative non-progressive phenotype (Fig.1A). Subclonal step wise acquisition of mutations correlated with multi lineage dysplasia. Individual hits imparted more circumscribe impact (e.g., SF3B1 on erythroid dysplasia, TET2 on myeloid dysplasia, ASXL1/SRSF2/TP53 on megakaryocytic dysplasia) (Fig.1B).Similar inferences can be made with regarding the impact of mutations on myeloproliferative features. For detailed analyses of these effects on morphological feature see abstract (How somatic mutations carve morphologic imprint in MDS?)
In conclusion, we report here the first comprehensive analysis of the impact of ancestral mutations in the context of invariant secondary hits on specific, morphologic features and odds of progression in myeloid neoplasms.
Makishima: Yasuda Medical Foundation: Research Funding. Sekeres: Celgene: Membership on an entity's Board of Directors or advisory committees. Haferlach: MLL Munich Leukemia Laboratory: Employment, Equity Ownership.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal