In this issue of Blood, Ren et al investigate mechanisms underlying a novel strategy to block multiple myeloma (MM) cell proliferation by targeting the heparin sulfate (HS) proteoglycan syndecan-1 and identify the Wnt/β-catenin pathway as a critical mediator of this approach. If validated, these findings could lead to new therapeutic strategies in MM that may be particularly effective in circumventing microenvironmental forms of resistance in this disorder.1
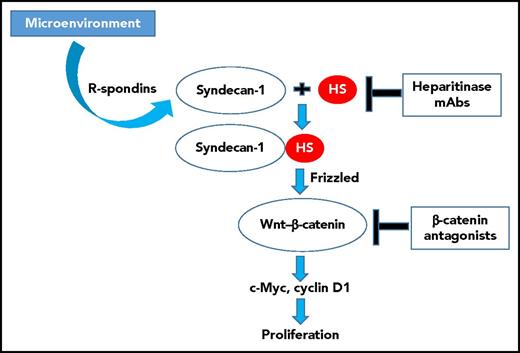
Microenvironmentally secreted proteins such as R-spondins collaborate with HS-decorated syndecan-1 to activate Wnt/ligands, leading via Frizzled to β-catenin–mediated induction of multiple downstream targets critical for MM cell growth and survival, including c-Myc and cyclin D1. Interruption of this process (eg, at the level of HS via monoclonal antibodies or heparitinase) can block this sequence of events, leading to inhibition of MM cell proliferation. It is possible that disabling this process at a more distal point (eg, by β-catenin antagonists) might recapitulate these effects. mAb, monoclonal antibody.
Microenvironmentally secreted proteins such as R-spondins collaborate with HS-decorated syndecan-1 to activate Wnt/ligands, leading via Frizzled to β-catenin–mediated induction of multiple downstream targets critical for MM cell growth and survival, including c-Myc and cyclin D1. Interruption of this process (eg, at the level of HS via monoclonal antibodies or heparitinase) can block this sequence of events, leading to inhibition of MM cell proliferation. It is possible that disabling this process at a more distal point (eg, by β-catenin antagonists) might recapitulate these effects. mAb, monoclonal antibody.
MM is an accumulative disease of mature plasma cells, and during the last several years, major therapeutic advances in this disease have emerged, including the introduction of novel proteasome inhibitors, immunomodulatory agents (IMiDs; eg, pomalidomide), and antibodies (eg, daratumumab, SLAM7).2 Nevertheless, despite these advances, the disease remains incurable for many patients, and new and more effective therapies are clearly needed. Notably, aside from intrinsic or acquired forms of drug refractoriness, stromal cell and other microenvironmental forms of resistance are widely believed to represent a continuing impediment to curative efforts.3
Syndecan-1 is a membrane-bound HS proteoglycan, which is an extracellular matrix or cell membrane–bound glycoprotein that binds to and regulates the activity of soluble protein ligands involved in regulation of cell signals related to growth, survival, differentiation, and tumorigenesis.4 It has been previously shown to be highly expressed in plasma cells and to play a role in mediating MM cell interactions with the bone marrow niche.5 Decoration of syndecan-1 by HS chains has been linked to multiple effects, including activation (via Frizzled) of the Wnt/β-catenin pathway, which operates upstream of c-Myc and cyclin D1, both of which are implicated in MM pathogenesis. In this context, R-spondins represent proteins secreted by the microenvironment that act as highly potent activators of Wnt/β-catenin signaling. Using genetic techniques, Ren et al found that loss of HS (by knocking down the HS copolymerase EXT1 by CRISPR/Cas9 or by coadministering heparitinase, which degrades HS) blocked Wnt/β-catenin pathway signaling and downregulated downstream targets in both MM cell lines and primary samples. Significantly, growth attenuation primarily reflected proliferation inhibition and could not be reversed by stromal cell coculture. Importantly, genetic manipulation via constitutively active β-catenin or c-Myc overexpression reduced antiproliferative effects of these interventions, demonstrating their functional significance in HS/syndecan-1 growth signaling. Of note, the paracrine effects of Wnt signaling could be recapitulated in stromal cells, which have been demonstrated to be a source of Wnt ligands.6 In support of this notion, the authors demonstrated that (pre)osteoclast-secreted R-spondins potently induced Wnt signaling and that this event could be antagonized by genetic or pharmacologic prevention of HS decoration of syndecan-1. The authors conclude that interventions disrupting HS-mediated activation of syndecan-1 and Wnt/β-catenin signaling pathway may represent a new and effective approach to circumventing bone marrow niche–related MM cell proliferation. A summary of the relevant pathways involved in these interactions is shown in the figure.
Although the findings by Ren et al could provide a novel framework for targeting MM by disabling a potentially important pathway implicated in microenvironmental signals, the study raises a number of questions, the resolution of which will be important in successfully translating this concept to the clinic. For example, it has long been recognized that stromal cell factors within the bone marrow niche confer resistance to a variety of agents in MM and other tumor types.7 Although disruption of HS-mediated syndecan-1 activation by microenvironmental factors could block MM cell proliferation, and potentially reverse resistance of such cells to diverse agents, it remains to be determined whether and to what extent this strategy would be effective against MM cells that exhibit intrinsic or acquired resistance to newer targeted agents such as IMiDs and proteasome inhibitors. If these mechanisms do not directly involve Wnt/β-catenin signaling, it is entirely possible that such cells would not be cross-resistant to a strategy targeting the HS/syndecan-1 axis. This possibility could be readily tested using established resistant cell models.
Another key issue is whether the in vitro findings described here can be translated into the in vivo setting. In fact, previous studies have suggested that syndecan-1 HS may be particularly important for MM cell growth in vivo.5 Whether the same downstream mechanisms involving Wnt/β-catenin signaling are operative in this setting remains to be determined.
The ability of targeting HS syndecan-1 to disable the Wnt/β-catenin pathway raises the question of whether this strategy might be particularly effective against primitive MM progenitors. This pathway has been implicated in stem-cell maintenance in the case of other hematopoietic malignancies (eg, acute myeloid leukemia),8 and it is possible that similar findings might apply to MM. This possibility could also be tested by determining whether HS syndecan targeting might deplete more primitive MM progenitors purportedly enriched for stem cell–like cells (eg, CD138−, CD19+, CD20+,CD27+).
An additional question critical for translation of this strategy is whether and how the HS syndecan-1 pathway could be targeted therapeutically. One possibility would be to target HS biosynthesis or catabolism; another might involve antibodies directed against HS chains of glypican-3.9 As with any therapy, questions about the selectivity of these approaches for MM cells would be essential to resolve. In addition, inasmuch as targeting HS syndecan-1 seems to operate by antagonizing Wnt/β-catenin signaling, the superiority of this approach compared to the use of a direct Wnt/β-catenin antagonist10 remains to be determined. The same issue could be raised in relation to agents that downregulate c-Myc (eg, BET inhibitors). In any case, further understanding of the mechanism(s) responsible for the biologic effects of disabling the HS/syndecan-1 axis could lay the groundwork for rational combination strategies involving these and other targeted agents, and particularly those specifically designed to overcome microenvironmental forms of drug resistance in MM.
Conflict-of-interest disclosure: The author declares no competing financial interests.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal