
Background: Romiplostim, a thrombopoietin (TPO) receptor agonist approved for children and adults with chronic ITP, was evaluated in children with ITP in a ≤3-year open-label trial. Interim results were previously reported (Grainger et al., Blood 2017 130:2334). Here we present updated results as of 27 Mar 2019.
Methods: Eligible children from 17 countries with ITP for ≥6 months and screening platelet count ≤30×109/L (or uncontrolled bleeding) received SC romiplostim (1 μg/kg titrated to 10 μg/kg to maintain platelet counts of 50-200×109/L). In Europe, bone marrow was evaluated at baseline and after 1 (day 365 ± 4 weeks) or 2 (day 730 ± 4 weeks) years. The primary endpoint was % time with a platelet response (platelet count ≥50×109/L, no rescue therapy in preceding 4 weeks) in months 0-6.
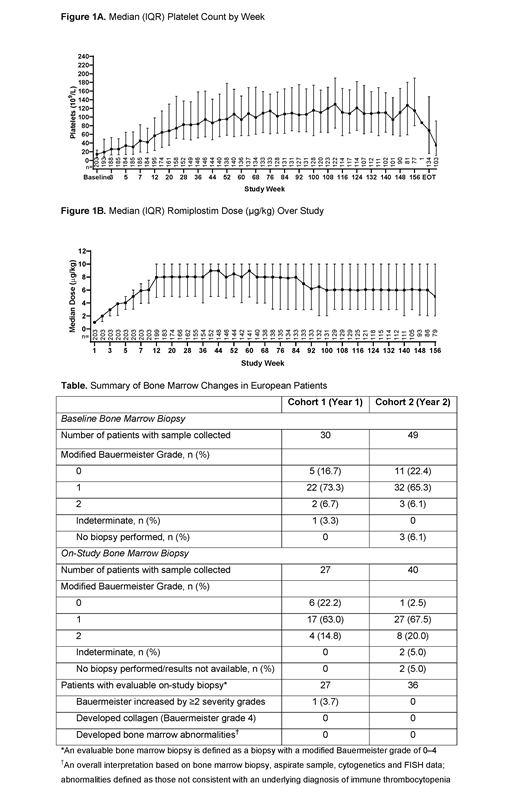
Results: A total of 203 patients (pts) received ≥1 dose; the median (interquartile range [IQR]) age was 10 (6-13) and median (IQR) platelet count 14 (7-23.5×109/L). The median (IQR) duration of treatment was 145 (39-156) weeks, median (IQR) % of time with a platelet response in months 0-6 was 50% (17-83%), with 88% (179/203) of pts having a platelet response at least once (Fig. 1A). In all pts, the median (IQR) % of time with an increase in platelet counts ≥20×109/L above baseline from week 2 until the end of treatment was 79% (39-92%). Median and lower quartile platelet counts were both consistently >50×109/L from week 12 and 48, respectively, and did not vary by age. Eleven pts maintained platelet counts ≥50×109/L without ITP medications (including romiplostim) for ≥24 weeks; median (IQR) time to onset was 50 (24-80) weeks after starting romiplostim. During the study, 60 (30%) pts received rescue therapy, typically within weeks 1-36, and 3 underwent splenectomy.
With a total exposure of 428.7 patient-years, median (IQR) average weekly romiplostim dose over the entire study was 6.9 (4.6-8.9) µg/kg; 8.5 (5.0-10.0) μg/kg at 1 year (n=144) and 6.0 (3.0-10.0) μg/kg at 2 years (n=129; Fig. 1B). Self-administration was initiated in 68% of pts. Ninety-five pts (46.8%) discontinued treatment: reasons included lack of efficacy (n=43 [21.2%]), patient request (n=15 [7.4%]), adverse event (AE; n=9 [4.4%]), and neutralizing antibodies (NAb; n=7 [3.4%]). AEs occurred in 192 pts (94.6%); the most frequent were epistaxis (38.4%), headache (37.9%), and nasopharyngitis (36.9%). Serious AEs (SAEs) occurred in 59 (29.1%) pts, including epistaxis (5.9%), decreased platelet count (4.4%), and thrombocytopenia and NAb (2% each); 8 pts had treatment-related SAEs (NAbs [n=4], headache and abdominal pain [each n=2], and presyncope [n=1]). Bleeding occurred in 69% of pts over the study, decreasing over time, with bleeding in 18% of pts from week 144 to the end of treatment. Bleeding-related AEs occurring in >10% of pts were epistaxis (38.4%), petechiae (23.6%), hematoma (20.7%), contusion (19.2%) and gingival bleeding (10.3%). CTCAE grade ≥3 bleeding events occurred in 20 pts (9.9%) and included epistaxis (n=9 [4.4%]), and persistent ITP (n=3 [1.5%]). In pts with no evidence of NAbs at baseline, there were 7 cases of NAbs to romiplostim (2 were transient) and 1 transient NAbs to TPO; only 1 pt, with NAbs to romiplostim, had a reduced therapeutic effect.
Of 75 European pts with evaluable baseline bone marrow biopsies [modified Bauermeister scores: grade 0 (no reticulin, n=16), 1 (fine fibers, n=54), or 2 (fine fiber network, n=5)], 27 had evaluable on-study biopsies after 1 year and 36 after 2 years (Table). Of these, 5 pts developed increased reticulin at year 1 and 17 at year 2. One pt had an increase in modified Bauermeister score from baseline of ≥2 grades (increase from grade 0 to 2), 4 pts had an increase in 1 grade, 1 a decrease in 2 grades, and 3 a decrease in 1 grade in year 1. In year 2, 15 pts had an increase in 1 grade and 3 pts a decrease in 1 grade. No pts developed collagen and no bone marrow abnormalities were detected.
Conclusion: Over the course of the study with >30 months of treatment, on a median dose of 6.9 μg/kg, 88% of children had a platelet response, median platelet counts were ≥20×109/L above baseline 79% of the time and >50×109/L from week 12. An important new safety signal over 429 patient-years was the 3.4% of NAbs to romiplostim; children develop NAbs with romiplostim more frequently than adults. Bone marrow findings showed that children, like adults, did not develop clinically important fibrosis.
Grainger:Amgen: Consultancy, Speakers Bureau; Ono: Consultancy; Alexion: Consultancy; ITP Support Association: Other: medical advisor; Octapharma: Consultancy; Biotest: Consultancy; Novartis: Consultancy, Speakers Bureau. Bussel:Tranquil: Honoraria, Membership on an entity's Board of Directors or advisory committees; Novartis: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Momenta Pharmaceuticals: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees; Dova Pharmaceuticals: Consultancy, Membership on an entity's Board of Directors or advisory committees; UCB: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees; argenx: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees; Regeneron: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees; RallyBio: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees; Kezar Life Sciences: Consultancy, Membership on an entity's Board of Directors or advisory committees; Physician Education Resource: Speakers Bureau; 3S Bio: Speakers Bureau; Rigel: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees; GSK: Honoraria, Membership on an entity's Board of Directors or advisory committees; Amgen: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees. Tarantino:Novo Nordisk: Consultancy, Membership on an entity's Board of Directors or advisory committees, Other: Investigator initiated study, Speakers Bureau; Bleeding and Clotting Disorders Institute: Employment; Pfizer: Other: PI for program grant; Amgen: Consultancy, Membership on an entity's Board of Directors or advisory committees, Other: Clinical Trial PI; Grifols: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding, Speakers Bureau; Takeda: Consultancy, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Michael Tarantino, MD SC: Other: President, Owner- Private Practice ; Genentech: Consultancy, Membership on an entity's Board of Directors or advisory committees. Cooper:Novartis: Honoraria, Other: clinical trial; Principia: Honoraria, Other: clinical trial; UCB: Other: clinical trial; Rigel: Honoraria, Other: clinical trial; Amgen: Honoraria, Other: clinical trial. Despotovic:Novartis: Research Funding; Dova: Honoraria. Wang:Amgen: Employment. Eisen:Amgen: Employment, Other: stock ownership. Bowers:Amgen: Employment, Other: stock ownership.
Author notes
Asterisk with author names denotes non-ASH members.

This icon denotes a clinically relevant abstract

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal