Background:
The treatment of multiple myeloma (MM) is optimized by use of combination regimens consisting of agents with different mechanisms of action. Panobinostat is a pan-inhibitor of histone deacetylases types I,II, and IV. Panobinostat, bortezomib, dexamethasone was shown to be an effective regimen (San Miguel et al Lancet Hematol 2016; Richardson et al Blood 2016), leading to the FDA approval of panobinostat for patients with relapsed/refractory MM. Carfilzomib is a proteasome inhibitor that was FDA approved in relapsed/refractory MM with the advantage of minimal neuropathy. Panobinostat and carfilzomib has also been shown to be a highly active regimen in relapsed/refractory MM with an overall response rate of up to 75% (Berdeja et al, Haematologica, 2015). With the heterogeneity of MM, individual patients exhibit wide variability in responses to drug combinations. A test that could predict patient responses to specific agents might enable clinicians to optimize therapy for patients, improving outcomes. We developed an in vitro high throughput drug sensitivity assay with formal synergy testing to predict response. In this ongoing trial, Panobinostat with Carfilzomib and Dexamethasone for Relapsed/Refractory Multiple Myeloma: Correlation with In Vitro Chemosensitivity Testing (NCT03256045), we will correlate individual patient in vitro sensitivity assay results with individual clinical response to the same triple drug regimen.
Study Design and Methods:
This study's objective is to directly demonstrate the utility of a high throughput drug sensitivity assay in determining biomarkers (e.g. individual IC50s, AUCs and/or synergy scores) to accurately predict response to combination therapy that was given prospectively to all enrolled patients.
We are enrolling patients with relapsed/refractory MM by IMWG criteria with measurable disease defined by the detection of a quantifiable monoclonal protein in the urine or serum or an abnormal serum free light chain ratio. Additionally, patients must have adequate blood counts and organ function. Patients who have had prior autologous or allogeneic transplants or CAR-T cell therapy are eligible. The regimen consists of panobinostat 20 mg orally on days 1,3,5,15,17,19; carfilzomib 20 mg/m2/dose IV on days 1,2 of cycle 1, then dose escalation up to 45 mg/m2/dose days 8,9,15,16 and all days for subsequent cycles; and dexamethasone 20 mg orally on days of carfilzomib. Dose reductions of all three drugs are permitted per patient tolerance to allow continuation on study treatment. Up to 12 cycles of treatment are permitted. Patients are monitored by serial electrocardiograms and assessments of cardiac function. Safety parameters including adverse events are recorded.
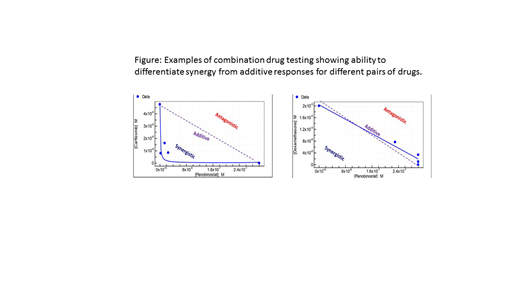
CD138+ plasma cells are procured from the patient bone marrow (aspiration and biopsy) and blood (when present) by magnetic bead separation. Cells are then added to 384-well plates and incubated overnight before the drugs are added. Cells are exposed to 8 concentrations (spanning 4 logs) of panobinostat, carfilzomib, or dexamethasone as singlet, doublet and triplet combinations for 72 hours. Cell viability is determined using CellTiter-Glo and IC50 and AUC values are are calculated by fitting data using least squares method to the standard four-parameter logistic model. Curve fitting is performed using IDBS XLFit software. The combination index is calculated by the method described by Chou and Talalay, Trends Pharmacol Sci 1983;4:450-4. Concentrations of Drug1 and Drug2 (that is, panobinostat and dexamethasone or panobinostat and carfilzomib) alone or in combinations are determined that give rise to 90% growth inhibition. At 90% Growth Inhibition, the Combination Index or CI = ([D1] in the combination / [D1] alone) + ([D2] in the combination / [D2] alone). All patients are treated with panobinostat, carfilzomib, and dexamethasone and evaluated for response using the IMWG response criteria. At the completion of enrollment at 35 patients, we plan to correlate the in vitro testing data with in vivo clinical response to determine appropriate biomarkers. This will be done by correlating the IC50s and AUCs for the individual drugs for responders vs. non-responders (including degree of response VGPR vs PR vs SD), as well as correlations of the synergy scores for each of the pairs of drugs in the responders vs. non-responders.
Enrollment was initiated in April 2018.
Becker:Accordant Health Services/Caremark: Consultancy; AbbVie, Amgen, Bristol-Myers Squibb, Glycomimetics, Invivoscribe, JW Pharmaceuticals, Novartis, Trovagene: Research Funding; The France Foundation: Honoraria. Libby:Abbvie: Consultancy; Pharmacyclics and Janssen: Consultancy; Akcea: Consultancy; Alnylam: Consultancy. Cowan:Juno: Research Funding; Abbvie: Research Funding; Sanofi: Consultancy; Janssen: Consultancy, Research Funding; Cellectar: Consultancy; Celgene: Consultancy, Research Funding. Hammer:Glycomimetics: Consultancy.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal